Lancet Respir Med. 2026 May;14(5):443-452.doi: 10.1016/S2213-2600(26)00047-0. Epub 2026 Mar 23.
Polymyxin B haemoadsorption in endotoxic septic shock (Tigris): a multicentre, open-label, Bayesian, randomised, controlled, phase 3 trial
Javier A Neyra, Matthieu Legrand, Mark A Tidswell, Ali Al-Khafaji, Claude Galphin, Ronald Rains, Danielle Davison, Ashita Tolwani, Jen-Ting Chen, William S Bender, Laurence W Busse, Nikhil K Meena, Jeffrey DellaVolpe, George W Williams, Kianoush B Kashani, Kyle J Gunnerson, Blaithin A McMahon, Jonathan Eaton, Sobia Khan, Roopa Kohli-Seth, Sugeet Jagpal, David Klein, Esha Kamaluddin, Debra M Foster, Paul M Walker, George Tomlinson, John A Kellum
PMID: 41887242
What is polymyxin B hemoperfusion?
Polymyxin B (PMX) is an antibiotic that binds strongly to endotoxin (lipopolysaccharide or LPS)- a component of the outer membrane of gram-negative bacteria that triggers overwhelming inflammation in septic shock. The catch, of course, is that systemic polymixin B is not a therapy we can casually give to crashing ICU patients, because nephrotoxicity and neurotoxicity make intravenous use untenable.
The workaround was elegant on paper and deeply attractive to device logic: immobilize polymixin B on polystyrene fibers inside a cartridge, run blood through it for 90 to 120 minutes, and claim endotoxin is captured as the blood is “cleansed” and returned. The cartridge has been around for decades, used in Japan since 1994, in Europe since 1998, and by more than 100,000 patients worldwide (Shoji H, Ther Apher Dial, 2003). Thus, it is established enough to persist and controversial enough to still need justification.
Why was this study needed?
The evidence before Tigris was deeply inconsistent. EUPHAS reported a mortality reduction from 53% to 32% but enrolled only 65 patients and stopped early, most likely overestimating treatment effects (Cruz DN, et al, JAMA, 2009). ABDOMIX found no benefit; 28-day mortality was 27.7% with PMX versus 19.5% without, and one third of the patients never received the second treatment session (Payen DM et al, Intensive Care Med, 2015). EUPHRATES, the largest and most rigorous trial (N=450, double blind, sham-controlled), showed no mortality difference (37.7% vs 34.5%) (Dellinger RP et al, JAMA, 2018).
Then came a post-hoc analysis of EUPHRATES. The Endotoxin Activity Assay (EAA) measures circulating endotoxin on a 0 to 1 scale. Values above 0.9 cannot be accurately quantified and may represent endotoxin burdens exceeding the cartridge’s adsorption capacity. When patients with EAA ≥ 0.9 (thus possibly the most severe sepsis and arguably the ones who might benefit the most) were removed, the analysis suggested a 10.7% absolute mortality reduction in those with EAA 0.60-0.89 (Klein DJ et al, Intensive Care Med, 2018). This was hypothesis-generating, non-confirmatory- yet it became the foundation for Tigris (Neyra JA et al, Lancet Respir Med, 2026). The study was needed because the US Food and Drug Administration required a prospective trial targeting this specific subgroup before considering approval.
How was the study done?
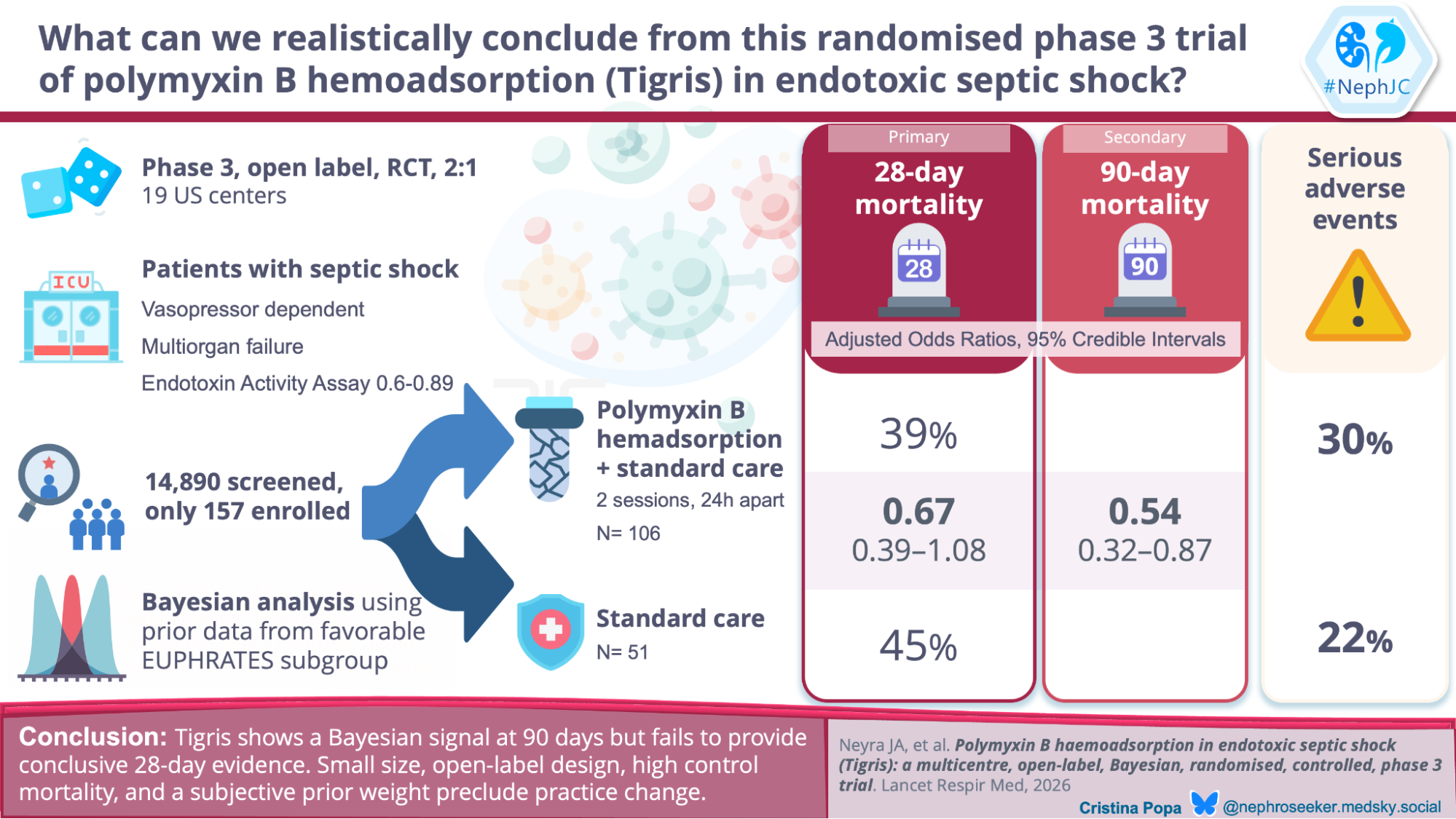
Tigris (Neyra JA et al, Lancet Respir Med, 2026) was a prospective, multicenter, open-label, randomized controlled phase 3 trial at 19 US hospitals between September 2019 and APRIL 2025. Eligible adults had septic shock requiring vasopressors for ≥2 hours, multiple organ dysfunction (MODS>9, SOFA>11), and EAA between 0.60 and 0.89 units. The EAA is a rapid (30-minute) whole blood immunoassay that quantifies endotoxin by measuring neutrophil chemiluminescent response- essentially, how strongly a patient’s white blood cells react to the endotoxin in their own blood. Key exclusion criteria included inability to maintain MAP ≥65 mmHg despite vasopressors and end-stage renal disease.
Randomization was 2:1 (106 PMX, 51 control), stratified by site. The intervention consisted of two PMX hemoperfusion sessions (Toraymyxin PMX-20R cartridge) of 90-120 minutes each, approximately 24 hours apart, at a blood flow rate of 80-120 mL/min. Controls received standard medical care alone. The primary outcome was 28-day mortality; the key secondary outcome was 90-day mortality.
The statistical analysis was Bayesian, which is sensible when the sample is tiny, but it also means the result is shaped by prior beliefs, not just by the new trial.
In Tigris, that mattered because only 157 patients were enrolled out of 14,890 screened, so the authors leaned on the EUPHRATES treatable cohort and then discounted it by 25%, formalized as a 75% weighted prior on the log-odds ratio for treatment effect. This means the analysis started with 94.2% posterior probability that PMX was beneficial before seeing any Tigris data (see table 2 below). Success was defined as a posterior probability p(OR<1) ≥ 95%.
The problem? The choice of 75% weighting is not derived from any objective criterion, and current data equally suggest a reasonable heuristic, but it is still heuristic. The sensitivity (eTable 5a) shows the consequences: at 50% weight, the posterior probability of benefit falls to 89.9% (below the 95% threshold); at 72%, it crosses 95%. The primary analysis, therefore, succeeds only in a negative trial with a post-hoc subgroup finding, which should be discounted more heavily, and would reasonably reject the 28-day conclusion.
eTable 5a. Results for 28-day Mortality using alternative weighing on the prior, from Neyra JA et al, Lancet Respir Med, 2026
Funding came from Spectral Medical, the manufacturer of the EAA assay and the trial sponsor. Sponsor employees participated in the study design, data interpretation, and manuscript writing. The senior author is a full-time Spectral employee holding company stock.
Results
Among 157 patients (106 PMX, 51 control), 100 patients in the PMX group and 51 in the control group received treatment and comprised the safety cohort.
Figure 1. Study flow of patients in the Tigris trial, from Neyra JA et al, Lancet Respir Med, 2026
Baseline characteristics were balanced. Mean APACHE II scores were 32.8 (PMX) and 31.7 (control); mean SOFA ~14, and 92% on norepinephrine >0.1 mcg/kg/min (Table 1). Endotoxin activity was balanced. The Tigris control group had a higher proportion of documented gram-negative infections (29%) than the EUPHRATES prior cohort (11%) and higher norepinephrine requirements (92% vs 84%).
Primary outcome (28-day mortality). Observed mortality was 38.7% in the PMX group versus 45.1% in controls- an absolute risk reduction of 6.4%. The adjusted odds ratio was 0.67 (95% credible interval 0.39-1.08). The posterior probability of benefit was 95.3%, meeting the prespecified threshold. A two-sided 95% credible interval excluded 1.0 only when the posterior probability of benefit exceeds 97.5%. At 95.3%, the analysis cannot rule out no effect or even harm.
Key secondary outcome (90-day mortality). Observed mortality was 43.4% versus 60.8% - an absolute reduction of 17.4%. The adjusted OR was 0.54 (95% credible interval 0.32-0.87), with a posterior probability of benefit >99%. The result seems robust. Even with an uninformative prior (eTable 5c), the posterior probability at 90 days is 98.4%.
Table 2. Mortality analysis by endpoint and source, from Neyra JA et al, Lancet Respir Med, 2026
The divergence between 28 and 90-day mortality raises a question: why would a two-session intervention given in the first 24 hours have a larger effect three months later? The author suggests that early shock resolution prevents persistent organ dysfunction that drives late deaths. This might sound plausible to the gullible readers, but the open-label design means clinicians know who received PMX, which could influence withdrawal of life-sustaining therapy and post-discharge care, potentially affecting survival trajectories beyond the acute phase.
Sensitivity analyses reveal fragility at the 28-day finding. As shown in eTable 5a, the posterior probability of benefit at 28 days exceeds 95% only when the prior weight is ≥ 72%. At 50% weight, the probability drops 89.9%- below the success thresholds. The 28-day results depend heavily on borrowing information from EUPHRATES. At 90 days, by contrast, the findings hold across all weights.
The frequentist analysis of Tigris alone (Figure 3) doesn’t support the conclusion of benefit at conventional levels of statistical significance. The hazard ratio is 0.68 (95% CI 0.43-1.07), with a confidence interval that crosses 1.0. The Bayesian analysis (Table 2) reaches significance only by incorporating prior data from EUPHRATES, the trial that had its primary analysis negative and whose positive subgroup was post-hoc. Does the prior deserve 75% weight? If prior discounts were more heavily discounted, the 90-day result might also vanish.
Figure 3. Kaplan–Meier graph of the cumulative survival at 90 days, from Neyra JA et al, Lancet Respir Med, 2026
Survival and safety. A Bayesian Cox model for survival to 90 days (eTable 6) gave a hazard ratio of 0.68 (95% credible interval 0.47-0.95) with a posterior probability of benefit of 98.8%.
Serious adverse events occurred in 30% of PMX patients and 22% of controls. Two treatment-related serious adverse events: one hypotension probably related to PMX, and one bleeding event definitely related to the dialysis catheter (eTable 7b). The safety profile may be considered acceptable for an extracorporeal therapy in critically ill patients, but it is difficult to be confident given small numbers.
eTable 7a. Serious adverse events (SAE) among safety population| eTable 7b. Listing of Treatment-related SAE, from Neyra JA et al, Lancet Respir Med, 2026
Does it change anything?
The 28-day result is fragile, at best. The credible interval includes 1.0. The observed 6.4% absolute risk reduction would not be statistically significant by conventional standards. The analysis crosses its Bayesian threshold only through a 75% weighted prior, possibly justifiable but clearly an arbitrary choice. A reader who believes EUPHRATES should be discounted more heavily (say 50%) would not consider the 28-day results convincing.
The 90-day result is stronger but raises a different concern. Control mortality was 60.8%, higher than the 43.9% in the EUPHRATES’ MODS >9 subgroup (Dellinger RP et al, JAMA, 2018) and higher than most contemporary septic shock trials. Why? The authors do not fully explain. In the controlled group mortality, which is atypically high, the 17.4% absolute risk reduction may be thus inflated. If this is the true natural history of endotoxic shock, then prior trials underestimated the baseline risk. Either way, generalizability is not guaranteed.
The open-label design also introduces potential bias. Mortality is objective, but knowledge of allocation can influence decisions about withdrawal of life support and discharge planning. In a disease where treatment limitations are common, unblinded status matters. The authors note that research teams were distinct from clinical teams, but complete separation is impossible.
What changes for clinical practice?
In the US, Tigris will likely nudge the FDA toward approval. Spectral Medical probably already has its hand on the submit button. In Europe and Japan, where PMX has been available for decades, proponents will feel vindicated; skeptics will shrug. The full catalog- EUPHAS (positive but flawed), ABDOMIX (negative), EUPHRATES (negative), and Tigris (positive only if you accept the Bayesian terms)- remains underwhelming, unimpressive and deeply debatable. The next version of the KDIGO AKI guidelines might endorse its use, but, like its endorsement of NAC for CIN, these recs would be mostly ignored by sensible peeps. Inserting a dialysis catheter when there may not be AKI, measuring something which most centers do not have access to make dialysis decisions, for uncertain benefit dependent on Bayesian voodoo and prejudiced priors? Really?
What changes for sepsis research?
Fifteen thousand screens. One hundred fifty-seven patients. That is a one percent capture rate. Tigris did not validate the elegant δ-phenotype of liver failure, shock, and coagulopathy (Seymour CW et al, JAMA, 2019); it validated an EAA test that most ICUs do not run. Bayesian methods are clever, but regulatory usefulness is not clinical truth. The real lesson is darker: after 30 years and tens of thousands of screens, we have a signal in 157 patients that still depends on how much faith you place in prior.
Conclusion
The 28-day result only reaches certainty because the authors chose a prior weight that guaranteed it. Adjust that assumption even slightly, and the benefit evaporates. The 90-day finding is complicated by a control group whose mortality was higher than the contemporary benchmark, leaving the true treatment effect uncertain. Strip away the borrowed confidence from a prior trial that never proved a positive outcome, and what remains? A signal too fragile to trust. This is not practice-changing evidence. It is a post-hoc hypothesis clothed in Bayesian formality.