#NephJC Chat
Tuesday, March 10th 2026, 9 pm Eastern on Bluesky
Clin J Am Soc Nephrol. 2026 Jan 2. doi: 10.2215/CJN.0000000969. Online ahead of print.
A Randomized Clinical Trial of Kidney Autologous Cell Therapy in Diabetic Kidney Disease
PMID: 41481370
‘Failure is the simple opportunity to begin again, this time more intelligently’
Henry Ford
Introduction
Around 850 million people around the world suffer from CKD; among them, 50% is due to diabetic kidney disease (DKD). For many decades, the only proven available therapy was renin-angiotensin system inhibitors (RASi). However, there has been a recent renaissance of therapies to slow DKD progression with newer molecules like sodium glucose transport inhibitors (SGLT2i), non-steroidal mineralocorticoid blockers (ns-MRAs), and glucagon-like peptide-1 receptor antagonists (GLP-1 RAs), creating the four pillars of DKD goal-directed medical treatment (GDMT) (Neuen et al, NDT, 2025).
These drugs can slow the progression of kidney disease but not fully reverse it, indicating that there is still an unmet need in DKD. Recently, cellular therapy (i.e., regenerative medicine) has opened up new possibilities in CKD treatment, and this sci-fi-esque technology is now within reach. Mesenchymal stem cells (MSCs), present in bone marrow and other tissues, can generate any tissue depending on their surrounding milieu. MSCs are attracted to injured organs (due to cytokine signaling) and, therefore, are potential therapeutics for many chronic diseases (Farini et al, Stem Cells Int, 2014). Unfortunately, up until now, the ability of the kidney to repair glomerular injury was limited by the kidney’s low regenerative potential, and DKD led to an inevitable downward spiral to ESKD (Bussolati et al, Nat Rev Neph, 2016).
Although initial studies with renal progenitor cells gave positive results in animal models of CKD, their short life span and lower potential to regenerate nephrons have limited their use in kidney disease. As we are aware, there are many types of kidney cells, and some do regenerate. Clinical medicine tells us that ATN and tubule regeneration is possible over a period of days to months. Surviving renal epithelial cells, even in the absence of renal progenitor cells, can regenerate the entire tubular system following acute tubular injury (Lin et al, J Clin Invest, 2005). In addition, animal models showed that selective renal cell injections can decrease the progression of kidney disease, halting the NfKB and TGF-ꞵ progressive damage (Stenvinkel et al, KI Rep, 2016); hence, renal autologous cell therapy (REACT therapy) has attracted interest for clinical use. REACT cells are collected through renal biopsy samples and grown in culture media before they are reinjected into the kidney directly (Stavas et al, Am J Neph, 2022).
If a kidney transplant (whole organ) is the best renal replacement therapy, then is REACT (partial liquid transplant) the best preventative strategy for progressive DKD?
The Study
Methods
The REGEN-007 trial was a phase 2, multicenter, randomized, open-label trial designed to evaluate the safety and efficacy of rilparencel in adults with type 1 or type 2 diabetes mellitus and advanced CKD. The trial enrolled individuals aged 30 to 80 with an eGFR of 20 to 50 ml/min/1.73 m², a UACR between 30 and 5000 mg/g, and an HbA1c below 10%.
Figure 1. Study design from Cizman et al.CJASN 2025
Inclusion and exclusion criteria
Preparation of Autologous Cells (Rilparencel)
An ultrasound-guided or CT-guided renal biopsy specimen was taken and transported to the prokidney manufacturing unit. The harvested tissue undergoes enzymatic digestion and density gradient separation to isolate a heterogeneous population of renal epithelial lineage cells (SRCs). These cells are expanded ex vivo for approximately four to six weeks under “good manufacturing practice” conditions before formulation into the final product rilparencel. The cell suspension is embedded in a thermolabile hydrogel carrier to facilitate cortical retention following injection.
Unlike most investigational regenerative approaches in nephrology, which rely on systemically administered mesenchymal cells, rilparencel represents a locoregional autologous cell therapy. The therapeutic premise is that injection of renal lineage cells directly into the kidney cortex may exert regenerative or reparative effects through paracrine signaling, modulation of local inflammation, and antifibrotic pathways. Preclinical models have suggested that these SRC populations may contain epithelial progenitor-like cells derived from multiple nephron lineages, including tubular epithelial and glomerular epithelial cells, although the exact functional contribution of these cell populations in human kidneys remains uncertain.
The required dose is calculated depending on the kidneys’ volume by MRI. For each gram of kidney, 3×106 cells are required for transfusion. The rilparencel concentration of 100×106 per 3 ml is sufficient for 100 g of kidney tissue. The final rilparencel product was injected percutaneously under CT guidance back into the patient’s kidney cortex.
Renal autologous cell therapy- REACT figure from Salybekov et al, Front Cell Dev Biol, 2024
Randomization and endpoints
The study’s primary efficacy endpoint was the change in the total (acute and chronic) slope of eGFR, calculated with the CKD-EPI 2009 equation, comparing the pre-injection period to the post-last injection period. Secondary efficacy endpoints included time to ≥40% eGFR decline, dialysis, renal or cardiovascular death, and composite renal outcomes. Safety endpoints encompassed biopsy-related complications, injection-related AEs, and product-related AEs. Analyses employed linear mixed-effects models for slope comparison and Kaplan-Meier methods for time-to-event outcomes.
Patients were randomized 1:1 into two treatment groups.
Cohort 1 (scheduled dosing): received two REACT injections—the first into 1 kidney and the second into the contralateral kidney approximately 3 months later.
Cohort 2 (triggered dosing): received one initial injection for the biopsied kidney and then evaluations every 3 months. A second injection was only administered if the patient met a “redose trigger," defined as an eGFR decline of ≥ 20% or a sustained UACR increase of ≥ 30%. If there were no triggers over a 15-month observation period following the first injection, then those patients would only receive a single injection.
Outcome Measurements, Safety Assessments, and Statistical Analysis
Efficacy analyses were performed on the modified intent-to-treat population and safety analyses on the safety set. The primary efficacy endpoint—fall in eGFR slope—was made from a linear mixed model.
Various subgroups
Primary safety endpoint: The percentage of participants with procedure-related and investigational product-related treatment-emergent adverse events (AEs and TEAEs). Secondary safety endpoint: The percentage of participants with procedure-related death.
Determinants of 5-year and 2-year risk of ESRD
Funding Source
Funding done by ProKidney (NASDAQ: PROK), a late-clinical-stage biotechnology company involved in autologous cell therapy which makes the product, and many of the authors (including first/last) are employees of the company. The company people did all the analysis and wrote the manuscript.
Results
Between July 2021 and March 2023, 77 participants were screened across five clinical sites in the United States. Of these, 53 participants underwent randomization, with 27 assigned to Cohort 1 and 26 to Cohort 2. Overall, 45 participants completed study treatment (23 in Cohort 1 and 22 in Cohort 2).
Figure 2. Participants screened and treated in study REGEN-007 (CONSORT Flow Diagram) from Čižman et al. CJASN 2026
The study population reflected a typical cohort of patients with advanced diabetic kidney disease. The mean age was 60 years, and approximately 2/3rd of the trial participants were male. The majority had type 2 diabetes (78%), and the mean baseline eGFR was 33 ml/min/1.73 m², indicating moderate to advanced CKD. Median albuminuria across the cohort was 421 mg/g. Most patients were on a RASi, around 30-40% were flozinated, with smaller numbers on a GLP1RA or an MRA.
Table 1. Demographics and baseline characteristics from Čižman et al. CJASN 2026
In the first cohort, most patients (24) received the two doses of rilparencel, while in the second cohort, 15/25 (~ 67%) needed a second injection because of eGFR decline of ≥ 20% or a sustained UACR increase of ≥ 30%.
Primary endpoint
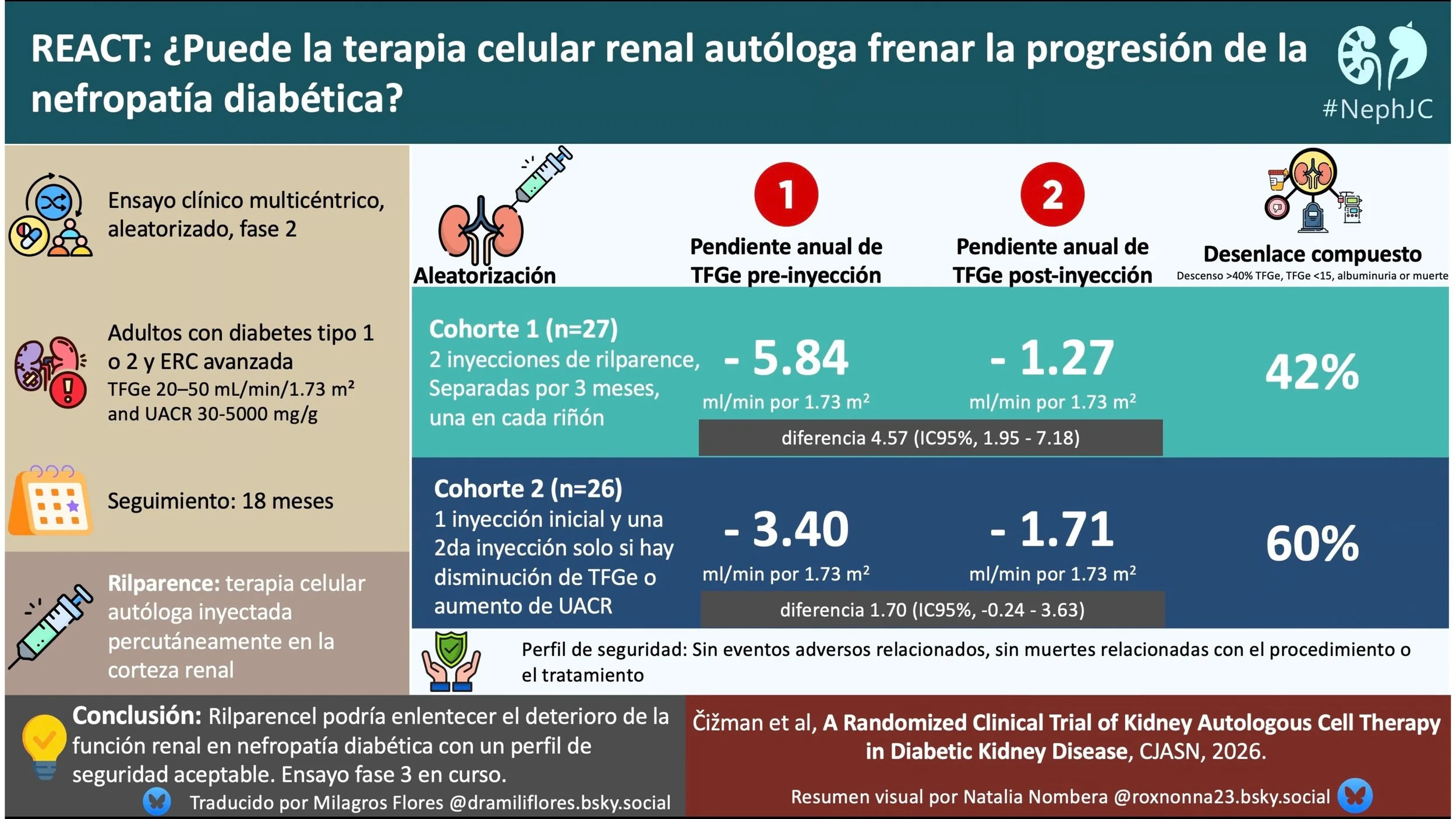
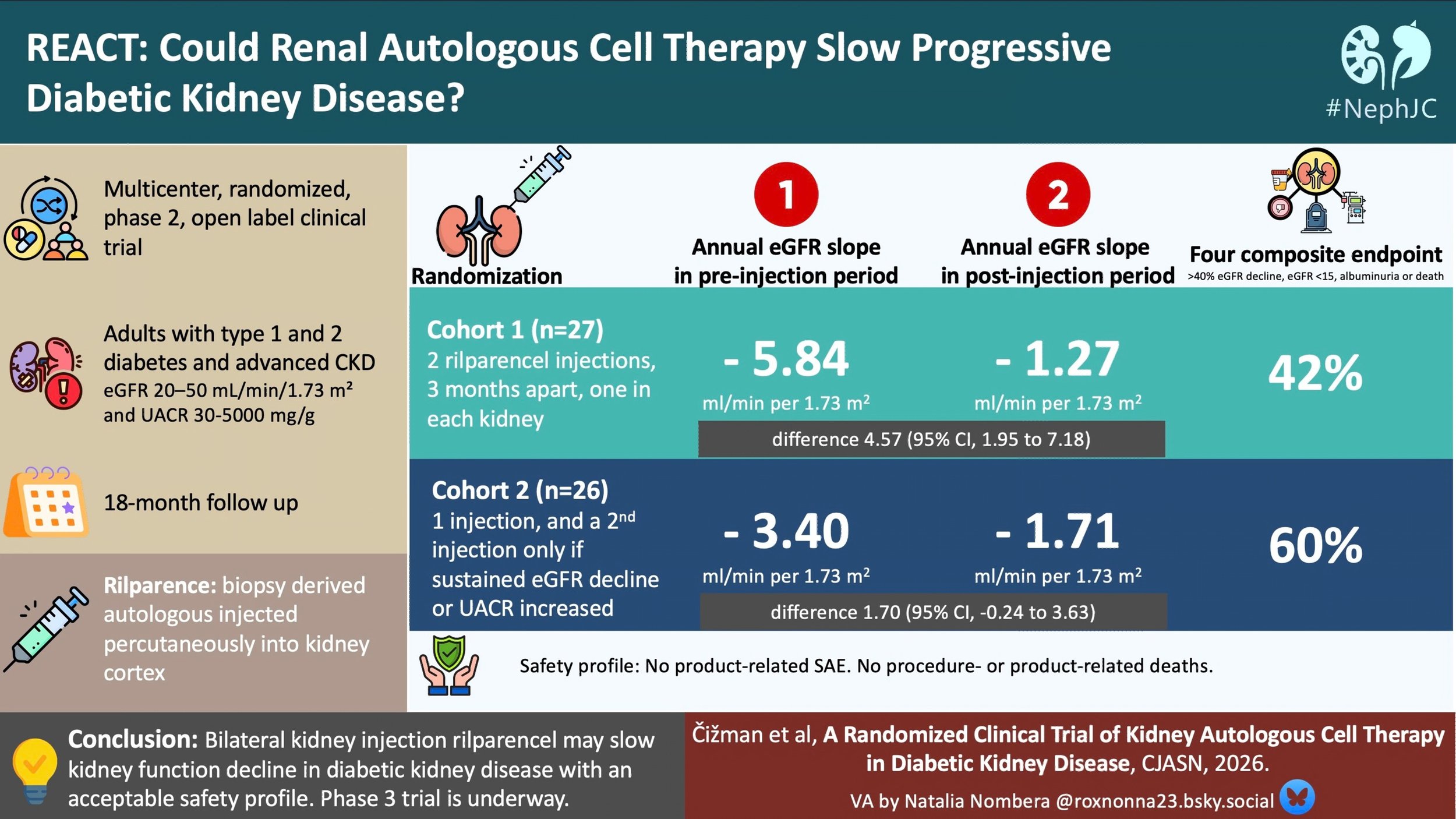
In Cohort 1, the annual change in kidney function in the preinjection period, as measured by the slope of eGFR, was −5.84 ml/min per 1.73 m² (95% CI, −7.97 to −3.70). In the period after the last injection, the annual change was −1.27 ml/min per 1.73 m² (95% CI, −3.97 to 1.43). The difference in the slope of eGFR between treatment periods was 4.57 ml/min per 1.73 m² (95% CI, 1.95 to 7.18). This represents a 78% improvement in the rate of eGFR decline for this group.
Figure 3. Change in GFR slope, preinjection to post last infusion from Čižman et al. CJASN 2026
In Cohort 2, the annual change in kidney function in the preinjection period, as measured by the slope of eGFR, was −3.40 ml/min per 1.73 m2 (95% CI, −5.03 to −1.77). In the period after the last injection, the annual change was −1.71 ml/min per 1.73 m2 (95% CI, −3.78 to 0.36). The difference in the slope of eGFR between treatment periods was 1.70 ml/min per 1.73 m2 (95% CI, −0.24 to 3.63).
Subgroup analyses evaluated the primary endpoint across baseline characteristics (including CKD stage, body mass index, HbA1c level, albuminuria category, and background therapy with SGLT2i or GLP-1RA). No consistent interaction between baseline characteristics and treatment response was identified, although subgroup sample sizes were small.
Supplement figure 1. Difference in slope of eGFR between pre and post injection from Čižman et al. CJASN 2026
Secondary outcomes
Clinical events were relatively infrequent during follow-up. For the 3-component composite outcome (≥40% decline in eGFR, eGFR <15 ml/min/1.73 m², or kidney/cardiovascular death), events occurred in 7 participants (29%) in cohort 1 versus 5 participants (20%) in cohort 2.
When a fourth component (increase in albuminuria ≥30%) was included, the four-component composite outcome occurred in 10 participants (42%) in Cohort 1 and 15 participants (60%) in Cohort 2. The median event-free time for this composite outcome in Cohort 2 was 17 months.
Table 3. Secondary Outcomes from Čižman et al. CJASN 2026
Risk prediction using the Kidney Failure Risk Equation (KFRE, 8 variable equation) suggested that kidney failure risk stabilized or improved in a proportion of participants following treatment. This was a prespecified endpoint of the study.
At 12 months after the first injection: 29% (7 patients) Cohort 1 and 16% ( 4 patients) Cohort 2 had the same or a lower predicted risk of ESKD compared with baseline. At 18 months, this was unchanged for Cohort 1 and had increased further to 28% in Cohort 2 showing stabilization or reduction in their predicted risk of kidney failure.
Table 2. Summary of 2-year and 5-year ESKD risk reduction. from Čižman et al. CJASN 2026
Four patients in cohort 1 and two patients in cohort 2 experienced a > 40% sustained drop in eGFR at 30 days. Additionally, six patients in cohort 1 and twelve patients in cohort 2 had a > 30% increase in UACR sustained at 90 days. The actual change in UACR is not found in the paper or supplement.
Safety
A total of 87 rilparencel injections were performed during the trial. Safety outcomes included adverse events related to 3 distinct components of the intervention: the kidney biopsy, the injection procedure, and the rilparencel itself. Procedure-related treatment-emergent adverse events occurred in 16 participants (33%). The most frequently reported events included injection site pain, renal hematoma, chills, headache, nausea. Six participants (12%) experienced product-related treatment-emergent adverse events, most of which were mild systemic symptoms such as nausea, dizziness, fatigue, or headache.
Supplement table 3. Adverse events related to rilparencel, from Čižman et al. CJASN 2026
Biopsy-related serious adverse events were reported in three participants and included subcapsular renal hematoma, acute kidney injury, and hematuria with hydronephrosis. One participant experienced a procedure-related subcapsular hematoma following injection.
Supplement table 1. Adverse events related to kidney biopsy, from Čižman et al. CJASN 2026
Importantly, no product-related serious adverse events and no procedure-related deaths were reported during the study.
Discussion
In this phase 2, clinical trial, the use of the rilparencel was associated with a slower decline of eGFR compared to the previous trend, in patients with advanced DKD (GFR ~ 30s, UACR ~ 400 mg/g) and a pre-infusion eGFR loss of ~ 3.4 to 5.8 ml/min/1.73m2/year. This suggests that even in such advanced disease when we usually think fibrosis and a cycle of inexorably declining kidney function has set in, injecting renal autologous cells may promote recovery or suppress further damage. In this limited sample size and follow up, rilparencel also seems to be well tolerated and safe.
Strengths
For a phase 2 RCT, this was quite well done to demonstrate that the investigational agent has some promise of efficacy and is relatively safe.
Limitations
No placebo or sham procedure arm was included, a decision driven largely by the procedural nature of the intervention. Because all participants required an initial kidney biopsy to manufacture the cellular product, the investigators elected to compare outcomes against each participant’s historical disease trajectory rather than against a concurrently treated control group. Consequently, the trial’s primary efficacy analysis relied on within-subject slope comparison, contrasting the pre-intervention decline in kidney function- derived from historical eGFR measurements spanning up to 24 months- with the slope observed after treatment. While this design increases statistical efficacy in small exploratory studies, it introduces important interpretive challenges, including susceptibility to regression to the mean, the Hawthorne effect, variability in historical laboratory measurements, and changes in concomitant therapies over time. Though the authors report changes in KFRE, they do not report actual pre/post UACR to allow us to understand what drove this change.
Rilparencel or GDMT?
The present study allowed all baseline GDMT for DKD. During the study 80% of participants were on ACE inhibitors or ARBs, however only 37% were receiving SGLT2 inhibitors and 39% were on GLP-1 receptor agonists at baseline. This is actually better than average for most patients with DKD in the US. So the change in slope of eGFR attributed to rilparencel injections is not the full story. Even in 2021-2023, our optimal use of DKD medications remains low, and less than 10% of patients who would qualify for such GDMT are actually receiving all four medications.
Hence, this was not a comparison that allows us to pit rilparencel against GDMT - since GDMT is already approved, one cannot do such a trial, and it is likely that in a phase 3 RCT many (or most) patients will be on GDMT as baseline standard of care. Nevertheless, as we have seen with reported data, it is almost possible to achieve remission - or bring eGFR down slope to a level expected for age-related decline - with full GDMT. GDMT also has other systemic benefits: reduction in blood sugars, blood pressure, obesity, and cardiovascular outcomes. It is unsure (and unlikely) that rilparencel would have such benefits. However, it represents a one or two shot intervention, with no ongoing pill burden. As uptake of GDMT medications increases, the need and effects of invasive procedures like REACT may be reserved for specific high risk DKD patients. Ultimately, the placebo-controlled phase 3 trial will determine whether rilparencel represents a new treatment option for patients with advanced type 2 diabetes mellitus and CKD.
What do other studies in this area show?
Various studies done with different cell lines in patients with AKI and CKD have been attempted, including stem cells and progenitor cells (Salybekov et al, Front Cell Dev Biol, 2024).
These studies are not without controversy, and unfortunately incorporate many elements that may lead to bias in observations. Current research is hindered by several common limitations including: selection of patients at different CKD stages and different underlying etiologies of the CKD. Only a few trials, such as the REACT studies, specifically target diabetes-related cases. This is problematic because different etiologies can lead to distinct types of kidney damage, which may affect the efficacy of the treatment. In addition, there remains no consensus on the optimal cell dosage. The absence of a standardized dosing protocol may lead to inconsistent results across studies, difficulties in conducting meta-analyses and systematic reviews, and challenges in clinical implementation. Finally, the small sample sizes in many studies contribute to a lack of randomization, low statistical power, limited generalizability, a higher risk of false positives, and potential biases.
Studies on autologous renal cells
Of course there are many potential advantages of autologous cell therapy over RRT and even transplantation. Being able to regenerate one’s own tissue to prevent organ failure and/or exposure to lifelong immunosuppression would be preferable to many patients. Infusions are also less stressful than organ surgery on elderly patients who might not otherwise qualify for kidney transplantation. Various studies are in the pipeline; some are completed, some are recruiting, and the majority of the studies are in the US.
Conclusion
The REGEN-007 phase 2 trial suggests that autologous renal cell therapy may alter the trajectory of kidney function in patients with diabetic CKD. The magnitude of the reported change in eGFR slope is notable, although interpretation is limited by the open-label design, reliance on historical controls, and small sample size. Whether rilparencel ultimately becomes part of the DKD therapeutic armamentarium will depend on the results of the ongoing placebo-controlled phase 3 trial, which are needed to determine whether the observed signal translates into meaningful clinical outcomes such as delayed dialysis or improved survival.
Summary by
Dr Sai Vani Yellampalli,
Consultant nephrologist
Kurnool Kidney Care
Dr Akshaya Jayachandran
Assistant Professor, Nephrology
Christian Medical College & Hospital
Vellore, Tamilnadu, India
NephJC Interns, Class of 2025
Reviewed by
Cristina Popa, Brian Rifkin, Swapnil Hiremath
Header Image created by AI, based on prompts by Akshaya Jayachandran