#NephJC 10 post discussion
Tuesday, April 10th 2026, 9 pm Eastern on X and Bluesky
JAMA Netw Open. 2026 Mar 16;9(3):e261943. doi: 10.1001/jamanetworkopen.2026.1943
Hydralazine Use and Risk of Vasculitis
D Fremont, S Dhaliwal, M Canney, A Akbari, G L Hundemer, V K Derebail, M M Sood, D Massicotte-Azarniouch
PMID: 41838000
Introduction
Hydralazine, a direct-acting vasodilator introduced in the 1950s, occupies a curious paradox in contemporary medicine. Though the therapeutic armamentarium for hypertension, pregnancy-related hypertension, and heart failure has expanded dramatically, this modest (thrice daily), inexpensive drug remains as an alternative choice when first-line agents are ineffective, contraindicated, or poorly tolerated. Its continued widespread use is thought to reflect a combination of availability, low cost, and a niche role at the periphery of guideline-directed care (McEvoy et al, Eur Heart J, 2024 | Jones et al, Hypertension, 2025 | Heidenreich et al, Circulation, 2022). It is more likely to reflect misguided choice for its fast action, unfounded fear of RASi in advancing CKD, hydralazine’s perceived metabolic neutrality, and race-tinged RASi science. More about that in the discussion.
The story of hydralazine's modern relevance, as with many drug-safety narratives, begins with a single patient. Case reports in the 1980’s described an unexpected constellation of immune phenomena- drug-induced lupus, and more ominously, antineutrophil cytoplasmic antibodies (ANCA)-associated vasculitis- often accompanied by rapidly progressive glomerulonephritis (Peacock, Br Med J (Clin Res Ed), 1981| Mason, J Clin Lab Immunol, 1986). Over the ensuing decades, a growing tapestry of observational signals reinforced the concern that the hemodynamic benefits of hydralazine may be overshadowed by immune dysregulation and devastating vasculitis (Choi et al, Arthritis Rheum, 2000| Kumar et al, Semin Arthritis Rheum, 2018| Santoriello et al, Kidney Int, 2021). These reports are largely derived from small case series and mechanistic conjecture rather than definitive causal trials, a limitation that is intrinsic to the study of rare adverse drug events.
Drug-induced vasculitis presents a methodological impasse: low incidence, heterogeneous clinical presentation, and a reliance on imperfect coding or reporting systems can obscure true associations. Consequently, clinicians must navigate uncertainty between a faint “signal” of harm and the surrounding “noise” of background disease, all while the potential outcomes- small-vessel vasculitis, crescentic glomerulonephritis, pulmonary-renal syndromes are severe (Kumar et al, Semin Arthritis Rheum, 2018| Santoriello et al, Kidney Int, 2021).
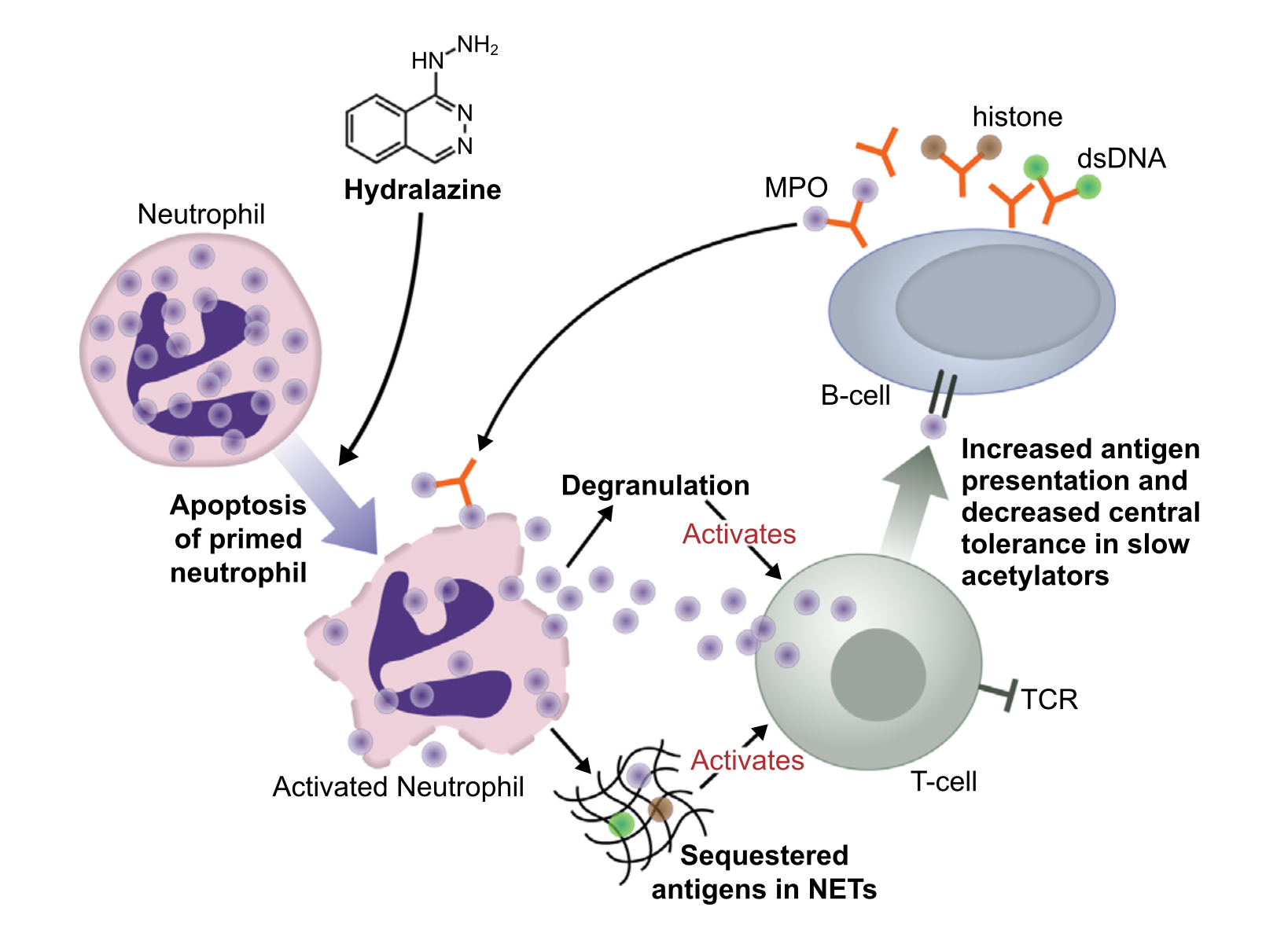
A coherent biological explanation has been proposed. Hydralazine is hypothesized to react with carbonyl derivatives on myeloperoxidase (MPO) or to bind directly to MPO, inducing conformational changes that generate neo-epitopes. These altered MPO molecules can stimulate autoantibody formation, particularly anti-MPO antibodies, setting off a cascade that culminates in small-vessel vasculitis and glomerular injury (Xi et al, J Clin Invest, 2025). Although not proven definitively, this mechanistic pathway, along with many case studies, justifies clinical vigilance.
Fig 2. Proposed hypotheses for how hydralazine promotes autoimmunity, from Kumar et al, Semin Arthritis Rheum, 2018
Medicine, by its nature, operates in realms of uncertainty; even therapies that have stood the test of time may harbor latent risks that surface only through fragmented evidence (Quizon et al, Clin Kidney J, 2025| Bomback, CJASN, 2017). Our prologue serves as an entry point into a deeper examination of how clinicians should interpret emerging population-level data on hydralazine-associated vasculitis, balancing the drug’s value against the rare but serious possibility of immune-mediated harm. Will Freemont et al study offer a rigorous epidemiologic lens through which to view this enduring paradox?
The Study
Methods
The investigators conducted a population-based retrospective cohort study using linked administrative health databases from ICES (Institute for Clinical Evaluative Sciences), covering Ontario’s universal single-payer healthcare system. The study period spanned January 2008 to December 2021, with follow-up extended to December 2022.
Population
The authors enrolled adults ≥66 years receiving a new outpatient prescription for either hydralazine or an ACE inhibitor/ARB. They set a lower age bound at 66 rather than 65 (the age of drug benefit eligibility) specifically to allow for a 1-year lookback window confirming true incident drug use.
Exclusions: The study excluded individuals with a prior vasculitis diagnosis, prior hydralazine use, prior kidney transplant, age >105, non-Ontario residency, and missing eGFR at the index date.
Exposure and comparator
The author defined the index date as the date of first dispensing. They employed a new-user active comparator design- comparing hydralazine users to new ACEi/ARB users rather than non-users. This choice reduces healthy-user bias by ensuring both groups share the act of initiating a cardiovascular medication for overlapping indications. In sensitivity analyses, the authors also tested new alpha-blocker users as a secondary comparator, representing an alternative 4th/5th line antihypertensive, and censoring when the drug was discontinued.
Outcome
The primary outcome was defined as any new vasculitis diagnosis after drug initiation, capturing it via ICD-10 codes from hospital and emergency department encounters. The authors chose a broad vasculitis code definition over AAV-specific codes because the latter yielded fewer than 6 events in hydralazine users- insufficient for analysis. This approach broadens sensitivity at the cost of specificity and cannot capture vasculitis that clinicians diagnosed and managed entirely in the outpatient setting.
Confounding control
To overcome baseline differences between groups, the authors applied overlap propensity score weighting across 21 covariates. This method weights patients in the zone of clinical equipoise, supposedly producing more stable estimates than conventional inverse probability weighting.
Statistical analysis
The authors generated the primary hazard ratio using overlap-weighted Cox proportional hazards regression and pre-specified six sensitivity analyses:
Competing risk adjustment via Fine-Gray subdistribution hazards is important in the elderly, comorbid population, where death often precludes vasculitis
Censoring at drug discontinuation: estimating on-treatment risk
Positive ANCA serology as an alternative, more specific outcome
Dose-response analysis: comparing above vs. below median daily does (40 mg)
Alpha-blocker comparator- replacing ACE/ARB with a similarly positioned drug class
Latency period analyses at 90, 180, and 365 days- accounting for potential delayed disease onset.
Funding
The study received institutional funding from the Ontario Ministry of Health via ICES and the Vasculitis Foundation Young Investigator Grant held by the lead author. Neither funder had any role in the conduct or reporting of the study.
Results
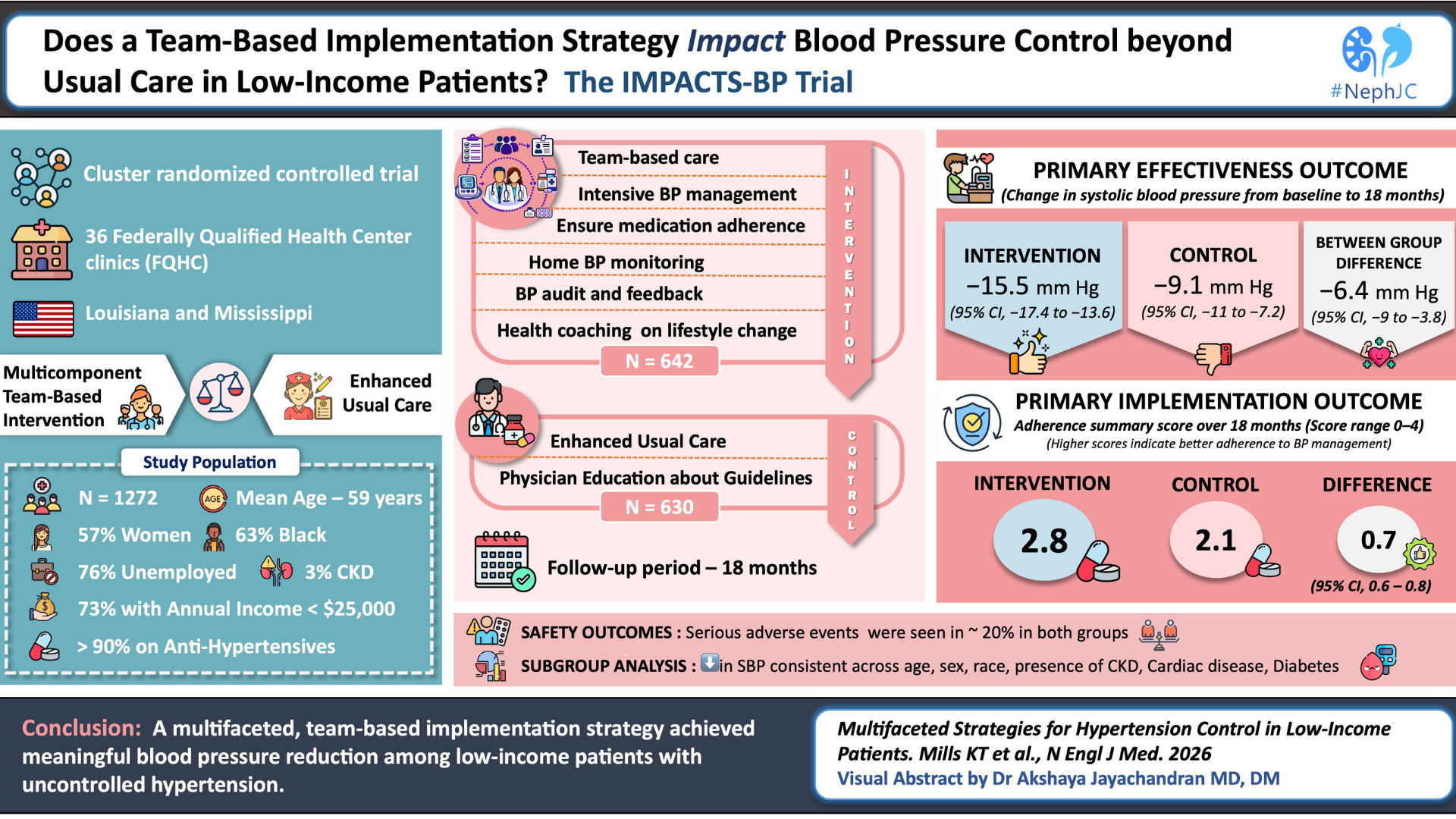
The final cohort included 583,136 older adults, of whom 40,748 initiated hydralazine and 542,388 initiated an ACE inhibitor or ARB. The mean age was 73 years, and over half were female. Mean follow-up was approximately 5.9 years.
Figure 1. Patient inclusion flow diagram. Fremont D et al, JAMA Netw Open, 2026
At baseline, hydralazine users were generally older and had more comorbidities and prior rheumatology visits. However, after overlap propensity score weighting, baseline characteristics seem balanced between groups, supporting comparability of groups.
Table 1. Patient characteristics, Fremont D et al, JAMA Netw Open, 2026
Primary Outcome
During follow-up, vasculitis diagnoses occurred in 328 of hydralazine patients (absolute risk 0.8%) versus 2712 of ACE or ARB patients (absolute risk 0.5%). In weighted analyses, hydralazine use was associated with an increased risk of vasculitis of 1.19 (95% CI 1.04 to 1.37) when compared to ACE or ARB users. This corresponded to an absolute risk difference of 0.3% and higher crude incidence rates in the hydralazine group (234.7 vs 81.6 per 100,000 person-years). This three-times aka ~300% higher risk translates into only a ~ 20% higher risk through the magic of propensity score weighted analysis.
Table 2. Risk of any vasculitis with hydralazine vs ACE or ARB. Fremont D et al, JAMA Netw Open, 2026
Time-to-event analysis showed that vasculitis occurred earlier in hydralazine users (median 545 days versus 1200 days, p<0.001), with separation of cumulative incidence curves over time.
Figure 2. Cumulative incidence of vasculitis by group. Fremont D et al, JAMA Netw Open, 2026
Additional Analyses
Diagnoses of AAV (using AAV codes) were very rare in hydralazine users to calculate reliable risk estimation. When the competing risk of death was considered, the association was not statistically significant (1.01; 95% CI, 0.88 to 1.16). Analyses censoring at treatment discontinuation yielded a higher estimated risk (HR 1.36), while a dose–response signal was observed, with higher doses (>40 mg/day) associated with increased risk (HR 1.28). However, when comparing with α-blockers, no significant difference was detected (HR 1.16, 95% CI, 0.98 to 1.37), and latency analyses at 90, 180, and 365 days showed non-significant associations.
Table 3. Sensitivity analyses of vasculitis risk with hydralazine use. Fremont D et al, JAMA Netw Open, 2026
Discussion
Since no prior studies directly compared hydralazine with ACEi or ARBs, this cohort provides one of the first population-level estimates of vasculitis risk in a real-world setting. In this cohort, hydralazine was associated with a risk for vasculitis in the weighted Cox model of 1.19, although the absolute risk difference was small (0.3%), and the association was no longer significant when accounting for additional analyses like the competing risk of death. These findings suggest that hospital-coded vasculitis following hydralazine exposure is uncommon, but they are less definitive in excluding under-recognized or incompletely captured cases (Fremont D et al, JAMA Netw Open, 2026).
This is particularly important when considered alongside prior clinicopathologic studies. In a biopsy-based series, hydralazine-associated ANCA glomerulonephritis accounted for 4.3% of all ANCA-GN cases and was characterized by a distinctive overlap phenotype: high MPO positivity, dual ANCA seropositivity, anti-histone antibodies, hypocomplementemia, and immune-complex deposition (Santoriello et al, Kidney Int, 2021). Similar findings have been reported in other cohorts, reinforcing that hydralazine-associated disease frequently diverges from classic pauci-immune AAV and instead reflects a broader clinical spectrum of immune dysregulation with multiple phenotypes. This heterogeneity raises the possibility that reliance on administrative coding may underestimate the true burden of disease (Santoriello et al, Kidney Int, 2021| Kumar et al, Semin Arthritis Rheum, 2018 | Choi HK et al, Arthritis Rheum, 2000). More recent case-based evidence further supports the ongoing clinical relevance of this entity. Contemporary reports of biopsy-proven hydralazine-associated ANCA-GN continue to describe severe renal presentations requiring immunosuppressive therapy (stopping the offending medication is insufficient), with consistent serologic and histopathologic overlap between drug-induced lupus and ANCA-associated vasculitis. Together, these suggest that although rare at the population level, hydralazine-associated vasculitis remains a reproducible and clinically meaningful syndrome (Quizon MR et al, CKJ, 2025).
Historical data suggest a stronger signal when broader autoimmune outcomes are considered. Earlier prospective studies demonstrated a dose-dependent relationship, with incidence rates of hydralazine-induced autoimmune syndromes increasing substantially at higher doses and with prolonged exposure, mean cumulative dose 146 grams & mean time of three months. This aligns with the current cohort, in which events occurred after prolonged exposure and dosing. Differences in clinical descriptors, ranging from lupus-like to biopsy-proven vasculitis, likely explain the apparent discrepancy across studies (Cameron HA & Ramsay LE, BMJ, 1984 | Timlin H et al, Cureus, 2019 | Kumar et al, Semin Arthritis Rheum, 2018, Fremont D et al, JAMA Netw Open, 2026).
The divergence between analytic approaches further highlights the complexity of interpretation. While the Cox model suggests association, the competing risk model attenuates this signal. However, this should not be interpreted as definitive reassurance. In an older population with significant comorbidity, death may not only compete with vasculitis as an outcome but may also preclude its recognition (Fremont D et al, JAMA Netw Open, 2026). It is unclear how the much higher crude ratio (~ 3 times) gets attenuated to only 20% after propensity score matching. The censored analysis (with HR 1.36) after drug discontinuation might even be the more important result.
Mechanistic data provide additional support for a causal relationship between hydralazine and autoimmune vasculitis. Hydralazine has been shown to alter immune tolerance through multiple pathways, including modulation of neutrophil antigens such as MPO and PR3, promotion of autoantibody formation, and induction of immune-complex–mediated injury. These mechanisms align closely with the clinical and histopathologic features described in biopsy-based studies, reinforcing the biological plausibility of hydralazine-induced vasculitis despite its low observed incidence in population studies. (Xi G et al , JCI, 2025 | Santambrogio L, JCI, 2025 | Drouzas K et al, Life (Basel), 2025 | Santoriello et al, Kidney Int, 2021).
Taken together, these findings suggest that hydralazine-associated vasculitis is a rare but biologically and clinically meaningful entity.
The apparent discrepancy between epidemiologic and clinicopathologic data likely reflects differences in outcome definition, ascertainment, and patient selection rather than true absence of effect.
Why use Hydralazine at all?
From a clinical perspective, these results emphasize the importance of vigilance for autoimmune complications, particularly in patients receiving higher doses or prolonged therapy. The authors suggest the risk of vasculitis is so low that it is not clinically meaningful given its risk-benefit. However, what benefit does it have? It is often used incorrectly for inpatient hypertension or hypertensive emergency. Inpatient hypertension does not need treatment and hydralazine use if associated with poor outcomes (eg Ghazi et al, J Hypertens 2023), and true hypertensive emergency has much more effective drugs (nitrates, nicardipine, clevidine, labetolol etc). In pregnancy, its use should follow labetolol, nifedipine, and alpha-methyldopa. Lastly, the heart failure trials (A-HeFT Taylor et al NEJM 2004) were done in the era when it was thought that skin color dictated RASi effect, and hydralazine was tested against placebo (not RASi, forget about flozins, MRAs, and beta-blockers, see Laurie Tomlinson rounds). There are no good indications for using hydralazine. When following a patient on hydralazine, we are left with the simple question, why-dralazine?
Strengths
Large, population-based cohort
First study including comparison groups
Overlap propensity score weighting could control confounding variables
Long follow-up, capturing delayed autoimmune events
Multiple sensitivity analyses (competing risk, dose-response, alternative comparators)
Limitations
Propensity score matched administrative database study with inherent limitations
Outcome based on ICD-10 codes, with risk of misclassification and under-ascertainment
Very low number of AAV-specific events, limiting phenotype-specific conclusions
Hydralazine-associated disease heterogeneity (AAV–lupus overlap) and may not be fully captured by coding
Restricted to older adults, limiting generalizability
Conclusions
Hydralazine-associated vasculitis appears to be rare at the population level but remains a biologically plausible and clinically significant condition. While this study helps address a long-standing evidence gap, it does not fully resolve whether hydralazine confers a clinically meaningful increased risk of AAV. For now, we should balance the risk, use when needed, but stay vigilant particularly in patients with prolonged exposure or higher dosing.