#NephJC Chat
Tuesday, February 25th 2026, 9 pm Eastern on Bluesky
Kidney Int 2026 Jan;109(1S):S1-S99. doi: 10.1016/j.kint.2025.06.006.
KDIGO 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease (CKD)
Kidney Disease: Improving Global Outcomes (KDIGO) Anemia Work Group: Marcello Tonelli, Jeffery S Berns, Biykem Bozkurt, Rebecca S Cheung, Yarieli Cuevas, Emmanuel E Effa, Michele F Eisenga, Steven Fishbane, Yelena Z Ginzburg, Volker H Haase, S Susan Hedayati, Siah Kim, José A Moura-Neto, Evi V Nagler, Patrick Rossignol, Manisha Sahay, Tetsuhiro Tanaka, Angela Yee-Moon Wang, David C Wheeler, Jodie L Babitt
PMID: 41485812
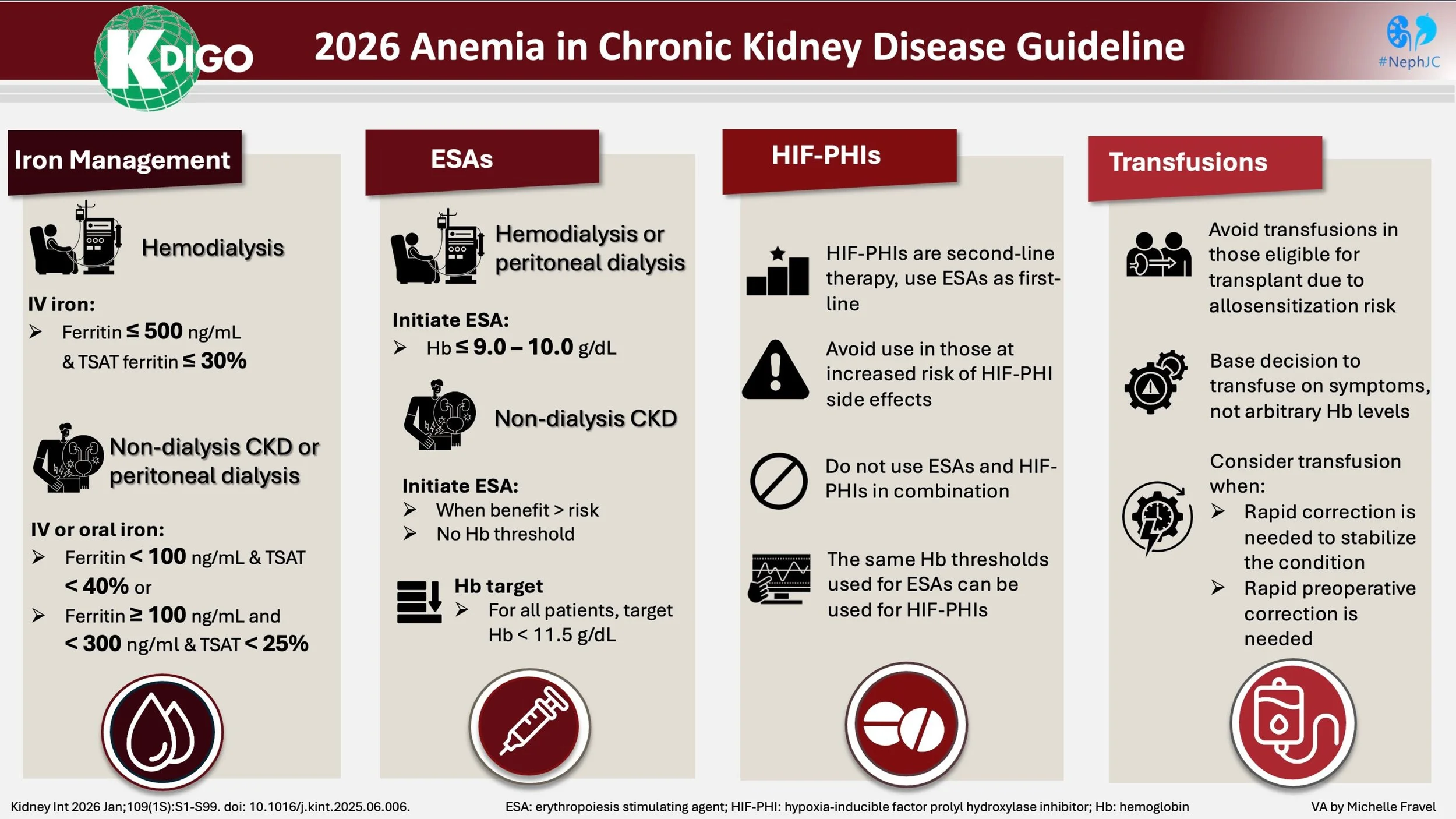
Introduction
Anemia is one of the most common and troubling issues faced by people living with chronic kidney disease (CKD). Anemia is associated with higher cardiovascular risk and mortality, and drives healthcare utilisation plus erodes quality of life. Patients describe it in concrete terms: exhaustion that sleep does not repair, breathlessness with minor exertion, dizziness, headaches, cognitive slowing, loss of appetite, and low mood. These symptoms overlap with CKD itself, blurring attribution and often delaying targeted treatment. The result is a functional decline that feels disproportionate to laboratory values.
Anemia advances quietly as kidney function recedes, following an insidious arithmetic. Overall, anemia is twice as prevalent as in the general population, with rates rising progressively as kidney function declines (Stauffer et al. PLOSOne, 2014). In the earliest stages, it is present in roughly 8-15% of the patients, by CKD stages 3-4, prevalence climbs to 30-50%, and in CKD stage 5 often 50-80% of patients are anemic. Within dialysis units, it is nearly universal, exceeding 90% in many cohorts (Stauffer et al, PLOS One, 2014; Hashimi et al, StatPearls Publishing, 2025; Kim et al, Nephrology (Carlton), 2025). Regional variability in anemia rates reflects demographic structure, socioeconomic gradients, nutritional status, comorbidity burden, and access to screening (Gardner et al, Lancet Global Health, 2023). Treatment intensity mirrors these disparities. Some health systems deploy iron supplementation and erythropoiesis-stimulating agents (ESAs) early and systematically; others struggle with delayed recognition and therapeutic inertia. Therefore, anemia in CKD is also shaped by the systems of care, as well as a myriad of other biologic factors. These challenges underscore the critical importance of evidence-based management strategies to mitigate anemia's impact on patient outcomes. The Kidney Disease: Improving Global Outcomes (KDIGO) has recently released the 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease, the first comprehensive update to the KDIGO guidelines since 2012. This guideline incorporates over a decade of new evidence on anemia pathophysiology, diagnostic approaches, iron therapy, erythropoiesis-stimulating agents (ESAs), hypoxia-inducible factor–prolyl hydroxylase inhibitors (HIF-PHIs), and red blood cell transfusions. It emphasizes individualized, patient-centered care—balancing benefits, risks, symptoms, and preferences—while providing practical recommendations and practice points applicable across CKD stages, including dialysis and transplant populations.

And let's give a massive shoutout to the science flex behind those HIF-PHIs. Back in 2019, the Nobel Prize in Physiology or Medicine went to Gregg Semenza, Sir Peter Ratcliffe (a nephrologist), and William Kaelin Jr. for cracking the code on how cells sense and adapt to oxygen levels—straight-up discovering and decoding the hypoxia-inducible factor (HIF) pathway. It’s not just decreased erythropoietin production, but rather a lack of localized hypoxia due to loss of kidney cellular activity, driving anemia.
Chapter 1
Diagnosis and evaluation of anemia in people with CKD
Anemia definition
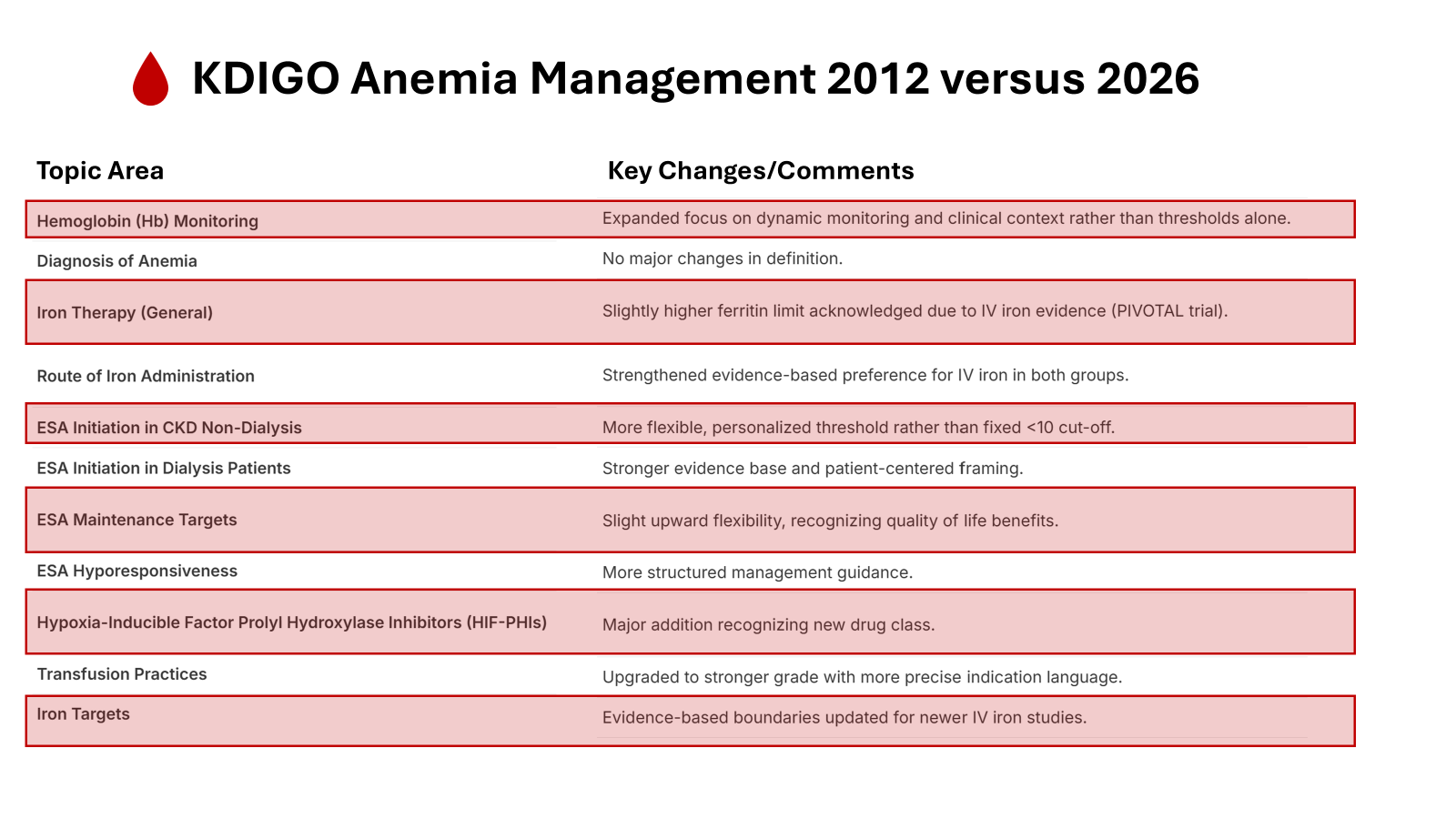
KDIGO 2026 retains the WHO-derived hemoglobin thresholds Hb <13 g/dl in men, and <12g/dl in women (recommendation 1.1.1). The definition is explicitly not graded. The work group states that there’s no new evidence to justify the revision. This preserves epidemiologic continuity with KDIGO 2012, and NICE 2021.
The retained thresholds are age-agnostic and largely statistical rather than mechanistic. A hemoglobin of 12.5 g/dL in a young woman is biologically different from the same value in an octogenarian with multimorbidity, yet KDIGO applies a single cut-off. Nor does the guideline address the conceptual tension between diagnosis and treatment: a patient with CKD stage G3 and one with G5D with identical hemoglobin carry the same diagnostic label, but therapeutic implications differ radically. Pregnancy-specific thresholds are not incorporated (see table 1 from Popa, Piccoli, Kidney Int Rep, 2024).
Diagnostic framework
Practice points 1.2.1-1.2.3 expand the initial work-up to include CBC, with reticulocyte count, ferritin, TSAT, CRP, vitamin B12, folate, and TSH, with peripheral smear when unrevealing. This is an upgrade to a much narrower 2012 focus on iron and EPO deficiency. The 2026 approach recognizes anemia in CKD as layered: inflammatory, nutritional, endocrine, hemorrhagic, and marrow-related.
Figure 6 Flowchart of different steps to follow when anemia and CKD, from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
Yet, the algorithm remains underspecified. The sequence of testing is not evidence-derived. CRP is listed conditionally, although inflammation is central to hepcidin-mediated iron sequestration. Reticulocyte hemoglobin content and percentage of hypochromic red cells are acknowledged as mechanistically superior to ferritin and TSAT for assessing iron availability, but are not recommended for routine use due to cost and availability. Hepcidin, the biological pivot of the chapter, is not measured. The guideline invokes a mechanism without equipping clinicians to confirm it.
Updated KDIGO Nomenclature for Iron-Deficient States
Anemia in CKD is more than simply limited iron availability for red blood cell (RBC) production. This arises from multiple factors: ongoing blood loss, nutritional shortfalls, drugs impairing iron absorption (e.g., PPIs), elevated hepcidin (the iron-regulatory hormone that blocks dietary iron uptake and sequesters iron in stores), and increased iron demand during ESA-driven erythropoiesis. The complexities of iron absorption and utilization, EPO production and bone marrow output are visualized below.
Figure 2 Mechanisms underlying anemia of chronic kidney disease from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
These processes lead to two distinct iron-deficient states in CKD. The KDIGO Work Group updates the terminology for greater pathophysiological precision:
Previously “absolute iron deficiency” → now systemic iron deficiency (low TSAT + low ferritin; true depletion of circulating and storage iron).
Previously “functional iron deficiency” → now iron-restricted erythropoiesis (low TSAT despite normal/high ferritin; iron trapped in stores, often hepcidin-driven, limiting erythroid use despite apparent adequacy).
This shift better explains why iron supplementation can boost hemoglobin and reduce ESA needs, even when stores appear replete.
Figure 4 Systemic movement of iron in different iron-related states from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
Monitoring
KDIGO 2026 recommends annual hemoglobin assessment in CKD G3, biannual in G4-G5 non-dialysis, and every 1-3 months in G5D. Iron parameters should be reassessed during therapy, with withholding considered when ferritin exceeds 700 ng/mL or TSAT ≥40%. These intervals reflect clinical logic: anemia accelerates as kidney function declines. No randomized data define these frequencies. The “1-3 months” range leaves room for practice variation. The guideline specifies absolute thresholds but doesn’t address rate-of-change or lab variability. Monitoring is sensible and empirically derived.
Chapter 2
Use of iron to treat iron deficiency and anemia in people with CKD
Two issues dominate this chapter: when to start iron and how to give it. KDIGO 2026 answers both, but with explicit acknowledgment that much of the evidence remains of low certainty.
This is the updated threshold from recent KDIGO anemia guidelines—a higher ferritin cutoff than older versions, reflecting real-world data on iron repletion in dialysis without excessive risk of overload. The 2D grade means a weak recommendation (benefits likely outweigh risks/burdens in most, but evidence quality is low-moderate), so individualization remains key: think symptoms, ESA responsiveness, inflammation, infection history, etc.
KDIGO’s 2.1 adopts the same ferritin ≤500 ng/mL/ TSAT ≤30% cut-off that existed in the 2012 guideline, but now couples it with a proactive IV-iron regimen (400 mg monthly, unless ferritin >700 ng/mL). The logic rests almost entirely on the PIVOTAL trial (Macdougall et al, NEJM 2019| NephJC summary). PIVOTAL (2141 patients with incident HD) compared proactive IV iron with a reactive protocol that gave iron only when ferritin fell <200ng/mL or TSAT <20%. The proactive arm used less ESA (~25% lower) and achieved a modest hemoglobin advantage. The primary composite cardiovascular endpoint showed an HR of 0.85, but the 95% CI (0.73-1.00) demonstrating a CV benefit with proactive iron approach (whether driven by more iron, less ESA, or higher Hgb will remain a topic of debate). KDIGO extrapolates the trial’s protocol (withholding iron only when ferritin >700ng/mL), which is reasonable given that PIVOTAL demonstrated it is safe to go beyond the arbitrary 500 to 700. Interestingly, the smaller and shorter DRIVE trial is ignored (Coyne et al, JASN 2007), which demonstrated the safety of IV iron to a ferritin of 1200, though without any CV outcome data.
The 2.2 recommendation that IV iron is superior to oral in HD is supported by small RCTs showing superior hemoglobin response and lower ESA doses with IV iron. Yet, those studies also share KDIGO’s limitation: they measure surrogate outcomes (Hb, ESA dose) and are short-term; no trial has shown a mortality or hard cardiovascular benefit from IV versus oral iron in dialysis.
Adapted table S6. RCT evidence informing iron recommendations in HD, from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
The guidelines prefer the intravenous over the oral route of administration among patients receiving hemodialysis.
Table 2. IV versus oral iron, from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
The permissive non-dialysis thresholds
Recommendation 2.3 dramatically lowers the ferritin bar for non-dialysis CKD (to <100 ng/mL) or 100-299 ng/mL with a tighter TSAT<25%. The change is driven by the FIND-CKD (Macdougall et al, NDT, 2014) and REVOKE (Agarwal et al, Kidney Int, 2015| NephJC summary) trials, both tiny. FIND-CKD showed that IV ferric carboxymaltose raised Hb by ~0.7 g/dL more than oral iron over 12 weeks; REVOKE showed a similar modest benefit, but reported more adverse events with IV Iron. Neither trial tested the specific thresholds that KDIGO now recommends, and both were underpowered to detect hard outcomes. The 2D grade, therefore, reflects that KDIGO took the inclusion criteria of these trials (ferritin < 300ng/mL) and turned them into treatment cut-offs.
This recommendation acknowledges the uncertainty by urging shared decision-making. It correctly notes that oral iron is cheaper, less invasive, and often sufficient, while IV iron gives a bigger Hb boost. The recommendation’s strength (2D) mirrors the modest efficacy gap (~0.3-0.7g/dL) and the lack of mortality data. In practice, many clinicians will try oral iron first, reserving IV for intolerance or failure after 1-3 months- exactly what KDIGO suggests, albeit in a more verbose, less decisive fashion.
Upper safety limits- where KDIGO’s caution may be too cautious
KDIGO’s practice point to stop iron when ferritin is >700 ng/mL or TSAT≥ 40% is taken straight from the PIVOTAL protocol. Observational dialysis cohorts do show higher infection and mortality rates at ferritin >700-800 ng/mL, but ferritin is an acute-phase reactant; the association might be confounded by inflammation. The DRIVE trials (Coyne et al, JASN, 2007), including patients with ferritin 500-1200 ng/mL, demonstrated no excess serious adverse events with short-term high-dose iron, hinting that the 700 ng/mL ceiling might be arbitrary.
What do other guidelines say?
NICE (London: National Institute for Health and Care Excellence (NICE); 2021) avoids numeric cut-offs altogether, simply recommending high-dose iv iron to spare ESA.
ERBP (Locatelli et al, NDT, 2013; Stoumpos et al, NDT, 2024) prefers the older, more conservative thresholds (absolute deficiency: ferritin <100ng/mL, TSAT <20%). The 2026 commentary (Del Vecchio et al NDT 2026) endorses KDIGO’s proactive HD approach but cautions against liberal iron in patients with high CRP, effectively re-introducing inflammation-adjusted decision-making that KDIGO 2026 glosses over.
UKKA is the most conservative: iron only for absolute deficiency or when ESA is already required, and it sets the upper ferritin limit at 600 ng/mL (Bhandari et al, BMC Nephrol, 2025).
Chapter 3
ESA/HIF-PHI Initiation, Targets & Management: Personalized & Cautious Approach.
For four decades, Erythropoiesis-Stimulating Agents (ESAs) have dominated CKD anemia management. They reliably raise hemoglobin, reduce transfusion need, and improve quality of life, outcomes demonstrated in many trials. Then came the hypoxia-Inducible Factor–Prolyl Hydroxylase Inhibitors (HIF-PHIs), oral agents that stabilize HIF- ⍺, upregulate endogenous erythropoietin, and reduce hepcidin-driven iron sequestration. They promise convenience, mechanistic elegance, and potentially reduced iron requirements. Yet their long-term safety remains uncertain. The 2026 KDIGO update attempts to reconcile the long-term safety data of ESAs with the newer, oral, but safety-uncertain HIF-PHIs. The basic clinical dictum of correcting potentially reversible causes of anemia before initiation of these agents remains constant and has been well emphasized by the guidelines. Many patients won't need additional medications if iron is optimized. Discuss symptoms, transfusion risk, and medication risks (stroke, thrombosis, cancer progression) with patients when trying to make an anemia plan.
Figure 8. Potentially reversible causes of anemia in chronic kidney disease in addition to decreased erythropoietin production from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
ESA recommendations
KDIGO 2026 maintains the same hemoglobin threshold for ESA initiation in dialysis (HD or PD) patients as did the 2012 guideline: start therapy when hemoglobin falls to or below 9-10 g/dL. This recommendation carries grade 2D (weak recommendation/very low certainty), signaling that while evidence supports starting ESA in this range, the certainty is limited. The rationale draws from observational and trial data showing that untreated dialysis patients rapidly progress to Hb <8g/dl, with associated transfusion requirement and poor functional status. An early landmark Canadian trial randomised patients on dialysis with Hb <9 g/dL to placebo, low-target ESA (Hb 9.5-11 g/dL), or high-target ESA (Hb >11 g/dL) and found that 58% of placebo recipients required transfusion within 8 weeks, compared with 2.5% in each ESA arm (Canadian Erythropoietin Study Group, BMJ, 1990). This trial established the principle that ESA prevents transfusion dependency, but it doesn’t answer whether Hb 9.5%g/dL is superior to 10.5g/dL as an initiation threshold, nor does it validate the exact 9-10g/dL window.
For non-dialysis CKD, KDIGO 2026 takes a different posture, abandoning a fixed threshold in favor of shared decision-making. The guideline suggests considering the presence of symptoms attributable to anemia, the potential benefits of higher hemoglobin, and the potential harms of RBC transfusion or ESA therapy. This echoes the KDIGO 2012 recommendation, but is now graded 2D (vs 1B in the 2012 recommendation 3.2, and 3.4.2- 2C), making explicit the very low certainty. In practice, this means ESA initiation might occur at Hb 8.5 g/dl in high-risk patients (recent stroke, thrombosis history, active cancer with curative intent) or at Hb 10 g/dL in a symptomatic patient pursuing a kidney transplant. The guideline does not specify exact thresholds for these subgroups; instead, it defers to clinician judgement informed by shared discussion. This transparency about uncertainty is commendable, but creates ambiguity. Two nephrologists facing a non-dialysis patient with a Hb of 9.5 g/dL, dyspnea, or prior stroke could reach opposite decisions under this framework, and both could be guideline-concordant.
KDIGO 2026 recommends maintaining Hb below 11.5 g/dl in adults on ESA therapy. This is the guideline’s strongest recommendation (1D- strong recommendation/very low certainty), a rare combination reflecting both mechanistic conviction and explicit acknowledgment of weak evidence. It’s very logical: ESA trials targeting higher hemoglobin (≥12.6-14 g/dL) consistently increased stroke, vascular access thrombosis, and cardiovascular events compared with lower targets (10-11 g/dL). The CHOIR trial randomized 1432 patients with non-dialysis CKD to Hb targets of 13.5 vs 11.3 g/dL and was terminated early after detecting a hazard ratio of 1.3 for the composite of death, MI, heart failure hospitalization, or stroke in the high-target arm (Singh et al, NEJM, 2006). The TREAT trial in diabetic CKD similarly found increased stroke with the darbepoetin targeting Hb ≥ 13g/dL (Pfeffer et al, NEJM, 2009). Notably, CHOIR and TREAT were trials of ESA dosing strategies, in addition to Hb targets (TREAT required rescue doses of darbepoetin if Hb < 9 g/dL). The achieved hemoglobin in the “high target” arms was often lower than intended (e.g. CHOIR achieved 12.6 g/dL in the high target group, despite aiming for 13.5), and one still cannot help but ask: was it the ESA or the “high” hemoglobin target that was problematic? Why does it matter? If the culprit is ESA dose rather than Hb level, then a non-dialysis patient who achieves Hb 13 g/dL on very low ESA doses might face less risk than current evidence suggests. This was also the era of lower iron targets (see above) so that also should be taken into considerations. With more (proactive) iron use, ESA doses are typically lower.
The guidelines also suggest that pediatric and transplant populations may tolerate higher Hb targets without equivalent toxicity, citing lower baseline cardiovascular risk and distinct clinical priorities (avoiding allosensitization, optimizing growth) (Portoles et al, Clin Kidney J, 2025). However, no pediatric RCTs have tested ESA safety at different Hb targets, so the recommended range for children (implicitly similar to adults, but individualized) rests on observational data and extrapolation.
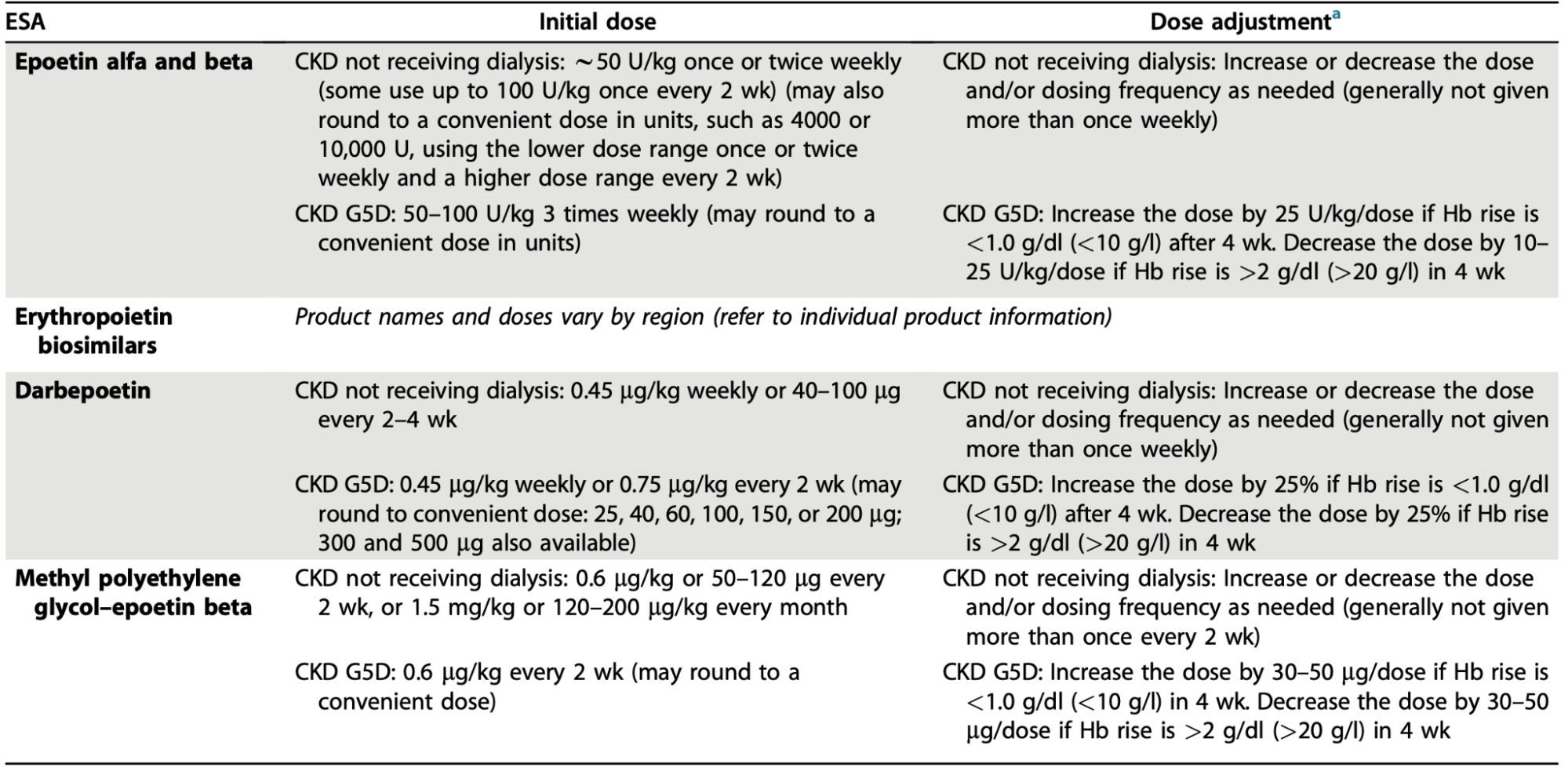
Table 7. Dosing of ESAs from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
ESA hyporesponsiveness
In clinical practice, ESA hyporesponsiveness is defined by the inability to achieve target hemoglobin levels despite significant dose escalations or the requirement of persistently high doses to maintain stability. This condition affects approximately 12.5% to 30.3% of the CKD population, though prevalence varies globally (Macdougall et al, Am J Nephrol, 2023).
Crucially, hyporesponsiveness is not merely a laboratory hurdle; it is a potent clinical marker. Clinically, it creates a significant testing burden of invasive testing for patients. Additionally, patients who exhibit a poor response to ESAs face significantly higher risks of cardiovascular events, progression to kidney failure, and all-cause mortality.
Key characteristics of ESA hyporesponsiveness:
Temporal nature: It can be acute or chronic (defined as lasting >4 months). It is often dynamic and transient, making it a difficult clinical target to treat.
Etiology: The underlying causes are complex and multifactorial. Common drivers include absolute or functional iron deficiency, chronic inflammation (the "anemia of inflammation"), and secondary hyperparathyroidism.
The "unexplained" gap: Despite thorough investigation, an evident cause may not be identified in approximately 30% of cases, highlighting the limitations of current management strategies.
Managing these patients requires a shift from simply "chasing the number" with higher doses, which may itself increase the risk of toxicity. Potentially reversible causes of anemia—particularly iron deficiency—should first be identified and treated.
If hyporesponsiveness persists despite evaluation and correction, management depends on anemia severity and symptoms: continue monitoring if mild and asymptomatic, or consider a 3–4 month trial of a HIF-PHI (stop HIF-PHI if Hb unchanged, ungraded) and/or transfusion as clinically indicated.
Table 10 Definitions of Hyporesponsiveness to ESA from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
Table 9. Causes of Hyporesponsiveness to ESA from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
Figure 10. Treatment algorithm for sustained erythropoiesis-stimulating agent (ESA) hyporesponsiveness from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
ESAs as first line, HIF-PHI as alternative
This is a weak recommendation (2D) that places higher value on ESA’s long track record of safety and efficacy than on HIF-PHIs’ putative advantages. The rationale acknowledges that heat-to-head RCTs have shown non-inferiority of HIF-PHIs may carry increased thrombotic risk, particularly vascular access thrombosis and acute coronary syndrome, in certain populations. The very-low-certainty grade reflects genuine uncertainty: the data do not conclusively show that ESAs are better than HIF-PHIs for hard outcomes, only that they have more extensive safety tracking. This is a conservative but defensible position in the face of incomplete information.
HIF-PHI role
If ESAs are the reliable "old guard," Hypoxia-Inducible Factor–Prolyl Hydroxylase Inhibitors (HIF-PHIs) are the shiny new oral disruptors. But with new mechanisms come new rules. Here’s the "CliffsNotes" version of how to use them without getting burned.
HIF-PHIs serve as a strategic pivot rather than a first-line default, offering a vital oral alternative for patients with ESA hyporesponsiveness, intolerance, or logistical barriers to injections. When switching, clinicians should utilize the same hemoglobin triggers and targets established for ESAs, following a "start low, go slow" titration to the minimum dose required for symptom relief and transfusion avoidance. Crucially, these agents must be used as a monotherapy; combining ESAs and HIF-PHIs is strictly avoided due to a lack of safety data.
Vigilance is required regarding off-target effects, necessitating extreme caution in patients with Polycystic Kidney Disease, proliferative retinopathy, or pulmonary arterial hypertension, and a definitive avoidance during pregnancy. Monitoring extends beyond monthly hemoglobin checks; if a meaningful response is not achieved within 3–4 months, the drug should be discontinued rather than escalating to potentially toxic doses. Finally, Roxadustat specifically requires thyroid monitoring during the first trimester of treatment to screen for central hypothyroidism. Ultimately, success with HIF-PHIs relies on a clear exit strategy and strict adherence to safety protocols. As the global availability of HIF-PHI molecules varies significantly by region, ongoing local post-marketing surveillance will be essential to clarifying their long-term safety and efficacy profiles across diverse populations.
Table 6. Considerations for HIF-PHI in CKD 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
Table 8. HIF-PHI agents available as of October 2024 from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
Some of the shiny new drugs image has been tarnished with the rocky Roxadustat rollercoaster (See NephJC summary and Podcast). Subsequent -dustats have demonstrated non-inferiority quite well, but not clear superiority hence these measured suggestions.
Chapter 4
Red blood cell transfusions to treat anemia in people with CKD
While modern erythropoietic agents and iron therapy have reduced blood bank reliance, RBC transfusions remain a vital but risky necessity in CKD. A restrictive strategy is now favored, moving away from arbitrary hemoglobin triggers toward clinical necessity. This shift balances immediate stability against significant risks like circulatory overload lung injury (TRALI), and—most critically for CKD—immunologic sensitization. This alloimmunization can devastate transplant prospects by increasing antibody levels, narrowing donor pools, and raising rejection risks. Therefore, clinicians must weigh acute life-saving benefits against long-term transplant-threatening harms through shared decision-making (practice point 4.1).
Transfusions are clearly indicated for life-threatening crises, such as acute hemorrhage or unstable coronary disease. In chronic settings, they are reserved for cases where ESAs or HIF-PHIs are ineffective—due to bone marrow failure—or contraindicated, such as active malignancy. Crucially, the decision should be symptom driven rather than numerical. While guideposts like <7 g/dL for asymptomatic inpatients or <8 g/dL for those with cardiovascular disease exist, the patient's actual clinical trend and symptoms, like dyspnea or angina, take precedence.
Practice point 4.5 specifies that transfusion is indicated when the benefit is clearly outweighed by the risk in acute crises (massive hemorrhage, unstable coronary syndromes, or perioperative anemia correction). The wording mirrors the older KDIGO 2012 stance (2C), but was now downgraded to a practice point. Minimizing transfusion requirements involves proactive systemic and bedside strategies. This includes standardized iron deficiency correction, consistent use of maintenance therapies during hospitalization, and limiting unnecessary phlebotomy. By educating patients and utilizing decision aids, clinicians ensure that every transfusion is a targeted, value-aligned choice that protects both current stability and future transplant eligibility.
Figure 13. Treatment algorithm for RBC transfusions in patients with CKD from 2026 Clinical Practice Guideline for the Management of Anemia in Chronic Kidney Disease
Discussion
We stand, as ever, on the narrow ledge peering into the dim recesses of CKD anemia. The 2026 KDIGO compass points us toward familiar landmarks (Hb thresholds inherited from a bygone WHO decree, ferritin and TSAT as the only lanterns we trust, and the old names of ESAs and iron as the twin pillars that have steadied practice for decades. Yet the markers, like the warning moon, are dimmed by inflammation, the hidden hand of hepcidin, and the shadows of age, gender, and pregnancy that the guideline refuses to illuminate.
In the dialysis ward, the guideline urges a “proactive” infusion of iron when ferritin slips beneath 500ng/mL and TSAT beneath 30%. The edifice of this counsel is the PIVOTAL trial. In the non-dialysis world, the threshold is lowered still, to ferritin <100 ng/mL with TSAT <40% or the more tentative ferritin 100-299 ng/mL with TSAT <25%. Here, the numbers are not drawn from outcomes but from the very doors through which patients were allowed to enter the FIND-CKD study, a subtle reminder that we sometimes mistake the map for the territory.
The ESAs, those old companions, are summoned when hemoglobin sinks to 9-10 g/dL in the dialyzed and are left to the discretion of the clinicians in the non-dialyzed, a range that swells to 8.5-10 ng/mL for those who suffer the weight of symptoms or the dread of cardiovascular peril.
The ceiling (11.5g/dL) stands as a stark reminder of the CHOIR and TREAT tragedies: to chase a higher tide is to invite storms of stroke and hypertension. Yet, the recommendation to hold fast at this ceiling is itself a thin thread, graded only “strong” in name while the certainty beneath it is whisper-thin. ESA hyporesponsiveness is highlighted as a strong prognostic marker (2- to 3-fold higher cardiovascular risk). The guideline rightly urges correction of reversible causes (iron, inflammation, PTH, infection) before escalating ESA dose. However, the algorithm for managing refractory cases (switch to a HIF-PHI for 3-4 months or consider transfusion) relies on very limited head-to-head data.
HIF-PHI agents are placed as a “second-line” oral alternative. The 2D recommendation is cautious: use the same Hb thresholds as ESAs, start slow, titrate slowly, and stop after 3-4 months if the response is inadequate. Safety concerns (thrombotic events, VEGF-mediated angiogenesis, thyroid dysfunction) are flagged, but the guideline doesn’t differentiate between agents that have shown divergent safety signals in recent phase 3 trials. The overall confidence is low, reflecting the paucity of long-term data.
Transfusion is finally framed as a symptom-driven, transplant-protective decision rather than a numeric trigger. The guideline advises transfusion only in life-threatening bleeding, unstable coronary disease, or when ESA/HIF-PHI therapy has failed. Reference values (Hb <7 g/dL asymptomatic, <8 g/dL with cardiovascular disease) are retained as background, not mandates. The evidence for reducing allo-sensitisation by restricting transfusion comes from older observational studies; no contemporary RCT has proven that a restrictive strategy improves transplant outcomes.
Where should anemia management head?
Mechanistic biomarkers: replace ferritin/TSAT with hepcidin, reticulocyte-hemoglobin content, or % hypochromic RBCs, and validate these markers in CKD cohorts. Until we can reliably differentiate systemic iron deficiency from iron-restricted erythropoiesis, the current threshold will continue to be arbitrary.
Outcome-based thresholds- conduct RCTs that test the clinical impact of ferritin/TSAT cut-offs, not merely their ability to raise Hb. Likewise, evaluate whether a lower ESA dose to achieve the same Hb target reduces cardiovascular risk.
Clarify HIF-PHI role: head-to-head, long-term studies comparing HIF-PHI with ESAs in non-dialysis CKD, with explicit safety endpoints (thrombosis, cancer progression, thyroid dysfunction), are needed before HIF-PHIs can be recommended beyond a narrow rescue indication.
Population-specific guidance: develop separate algorithms for women of reproductive age, pregnancy, children, and the elderly. Current one-size-fits-all hemoglobin definitions ignore well-documented physiological differences.
Equity and implementation: address the documented 68% treatment gap in the US non-dialysis CKD (CKD-DOPPS data) by integrating decision-support tools into electronic health records, tracking ESA and iron utilization, and targeting resources to underserved populations.
Transfusion stewardship: build concrete decision aid calculators that incorporate symptom burden, CV risk, and allo-sensitisation probability, turning the guideline’s “shared decision-making” principle into an actionable process.
KDIGO 2026 is a methodologically transparent document: it grades most recommendations, openly acknowledges very low certainty, and emphasizes correction of reversible causes before adding pharmacologic therapy. This is honestly commendable. However, the substance of many recommendations is little more than a rebranding of old numeric thresholds, now couched in softer language. The guideline leans heavily on post-hoc interpretations of trials (PIVOTAL, FIND-CKD) and on observational data for transfusion safety, offering little in the way of new, high-quality evidence. In practice, clinicians will find a clearer philosophical framework: ”treat what you can, use minimal effective dose, involve the patient”, but will still be left to decide how to apply vague hemoglobin or iron cut-offs in the absence of solid outcome data. In short, KDIGO 2026 moves the conversation forward in tone and structure, yet the road ahead remains paved with uncertainty. Until the recommended thresholds and therapeutic hierarchies are proven by robust trials, clinicians must treat the guideline as a cautionary compass rather than a definitive map.
Summary by
Jeyakumar Meyyappan
Transplant Nephrologist
Reviewed by
Cristina Popa, Sayali Thakare, Pallavi Prasad,
Swapnil Hiremath, Brian Rifkin