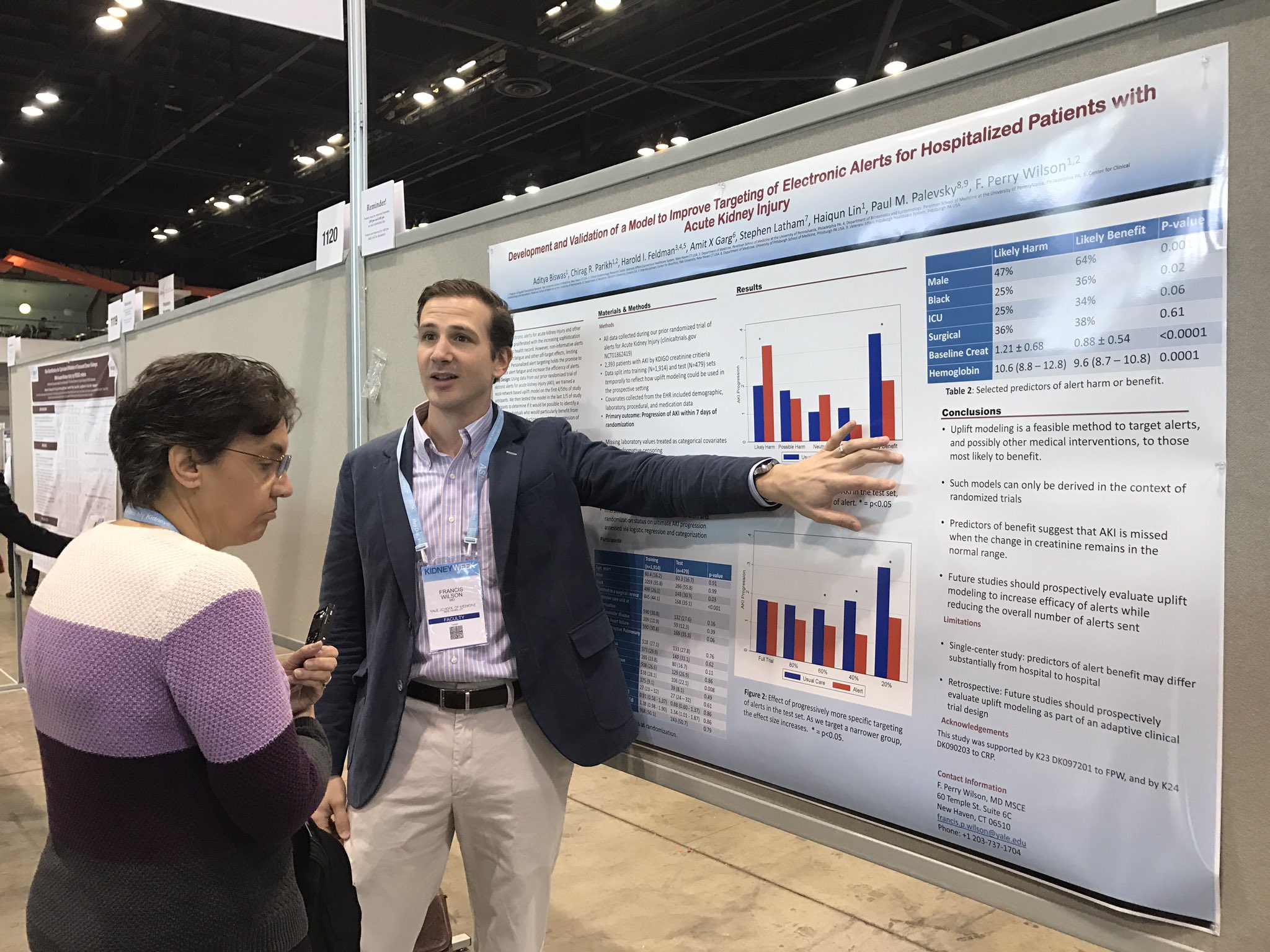
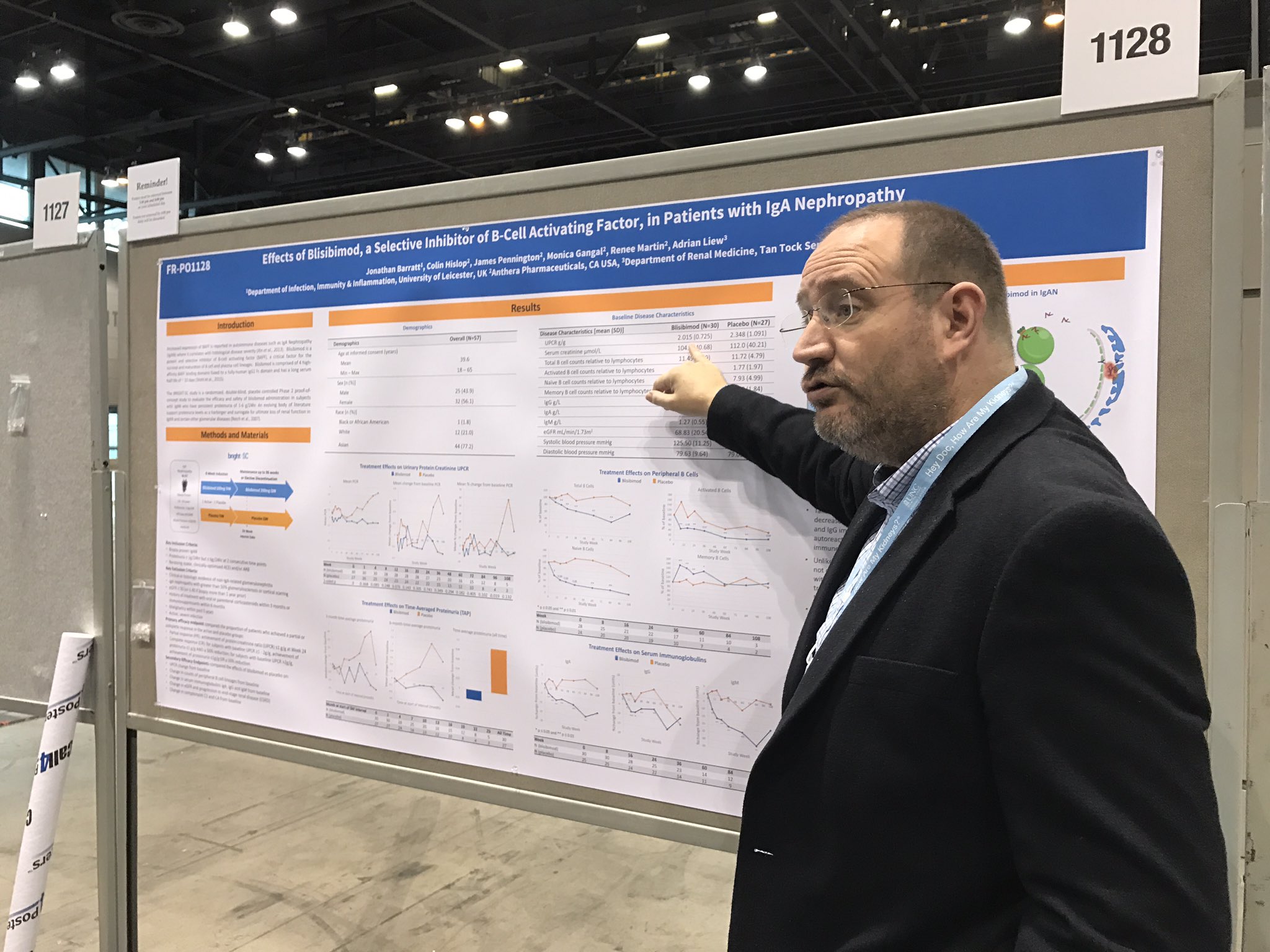
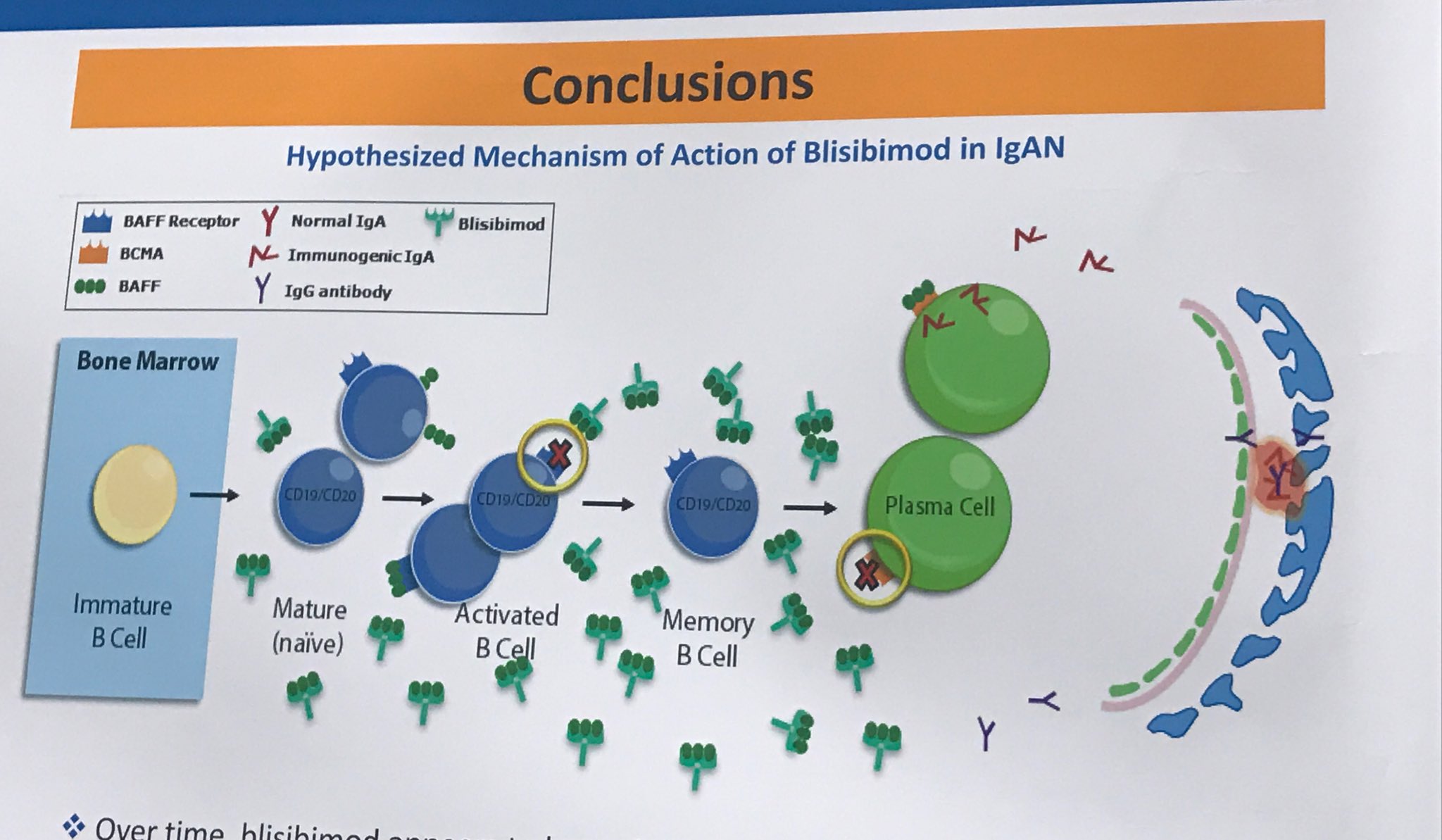
On Friday morning Michelle Rheault, Swapnil Hiremath and Joel Topf guided a tour of about a dozen people to some of the best posters of the morning session. It was delightful.
Here are some of the posters we saw:
FR-PO402
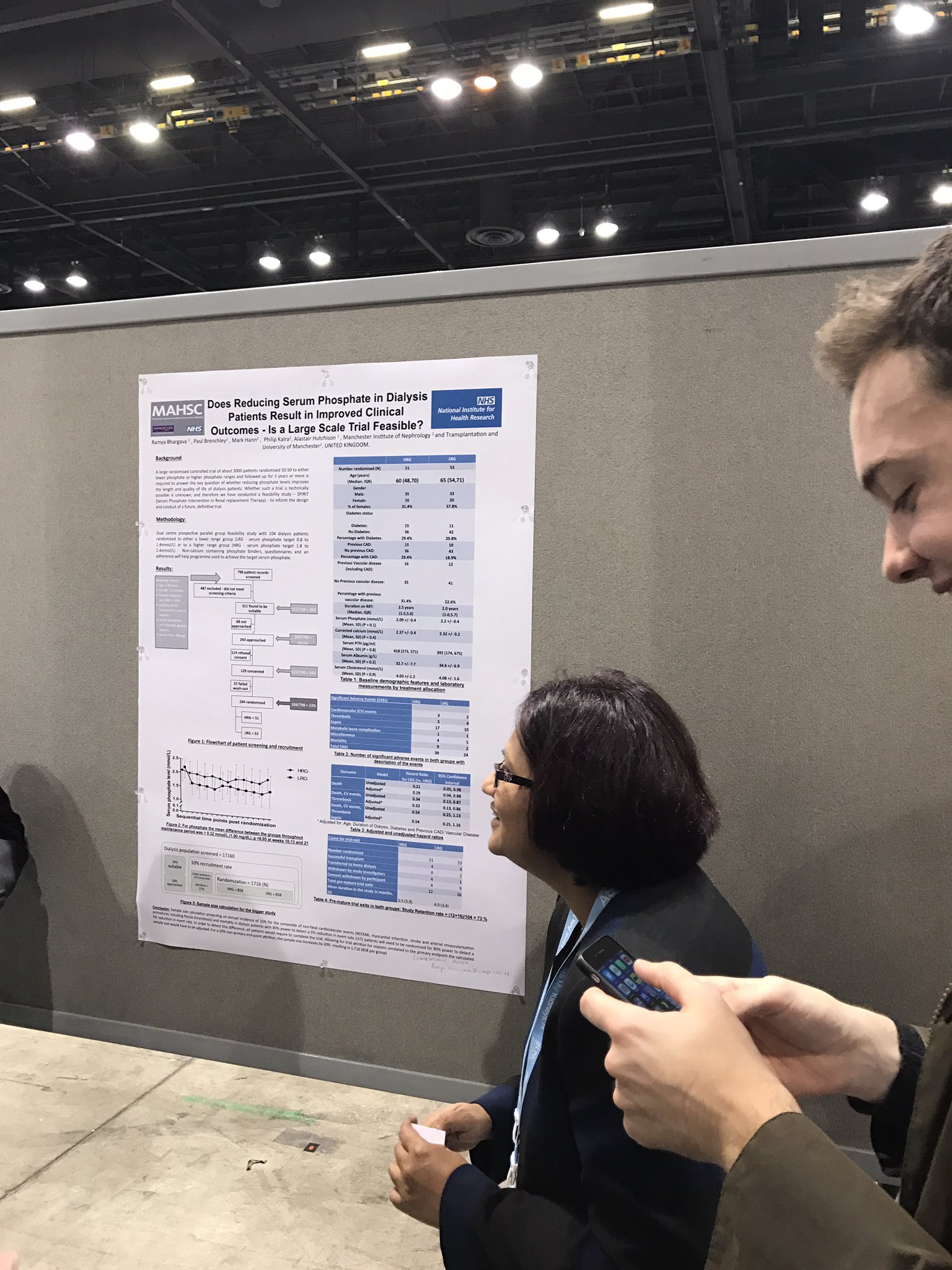
Does Reducing Serum Phosphate in Dialysis Patients Result in Improved Clinical Outcomes - Is a Large Scale Trial Feasible? Ramya Bhargava, Philip A. Kalra, Paul E. Brenchley,1 Alastair J. Hutchison. Manchester Royal In rmary; Salford Royal Hospital.
Background: High phosphate is associated with increased mortality in dialysis patients. However no RCTs have demonstrated that reducing serum phosphate improves quality or length of life. The required size and scope of such a trial is unknown; therefore we conducted a prospective, randomised, feasibility study to inform the design of a definitive trial.
Methods: Dual centre, prospective, parallel group, RCT feasibility study. 104 HD patients were randomized to lower range (LRG phosphate target 2.5 to 4.3mg/dL) or higher range group (HRG 5.6 to 7.4mg/dL). Non-calcium containing binders, questionnaires & an adherence self-help programme were used to achieve target phosphate. End points included - number titrated to required range & maintained in range over the maintenance period, consent rates, pill burden, drop-out rates & cardiovascular events.
Results: For phosphate the mean difference between the groups throughout maintenance period was >1.00 mg/dL, p <0.05 at weeks 10,13 and 21. 9 patients died in HRG vs only 2 in LRG. Consent withdrawal 8 LRG vs 4 HRG. Pill burden was higher in LRG. Dialysis access problems 9 in HRG vs 3 in LRG.
Conclusions: 65% retention rate is similar to other interventional RCTs in dialysis patients. 10% mortality is as expected for this cohort, but was higher in HRG. With a 13% randomization rate and 65% annual retention rate, 3 - 4,000 patients would need to be recruited for 1000 to complete 2 years follow-up. This suggests a target dialysis population of about 25,000, necessitating a multi-national approach.
FR-PO406
Low Serum Magnesium Is Associated with an Increased Risk of Prediabetes
Brenda C.T. Kieboom, Symen Ligthart, Abbas Dehghan, Oscar Franco, AlbertHofman, RobertZietse, BrunoH. Stricker, EwoutJ.Hoorn. 1Internal Medicine, Erasmus MC, Rotterdam, Netherlands; Epidemiology, Erasmus MC, Rotterdam, Netherlands.
Background: Previous studies identi ed an association between serum magnesium and incident diabetes mellitus. However, this association may be explained by reverse causality, as hyperglycemia can lower serum magnesium through urinary magnesium loss. Prediabetes is less likely to cause urinary magnesium loss, because hyperglycemia is less severe. Therefore, we studied, for the rst time, the association between serum magnesium and prediabetes.
Methods: The association was analyzed in the population-based Rotterdam Study using Cox proportional hazard models adjusted for age, sex, lifestyle factors, comorbidities, kidney function, other electrolytes and diuretic use. In addition, a mediation analysis was performed to study if the risk is mediated through insulin resistance (HOMA-IR levels) or in uenced by common genetic variation in eight magnesium transporter genes.
Results: 8555 participants (mean age 64.7 years) with normal glucose levels at baseline were included; the median follow-up was 7.0 years. A 0.1 mmol/L decrease in serum magnesium level was associated with an increase in diabetes risk (HR:1.15, 95%CI 1.02- 1.31), con rming previous studies. Of interest, a similar association was found between serum magnesium and prediabetes (HR:1.12, 95%CI 1.01-1.26). The effect of serum magnesium on prediabetes was mediated for 34.6% through HOMA-IR levels. Genetic variation in CNNM2 decreased prediabetes risk, which was completely mediated by increasing serum magnesium levels (indirect effect: OR:0.96, 95%CI 0.94-0.98), whereas genetic variation in FXYD2 increased prediabetes risk, which was completely mediated by decreasing serum magnesium levels (indirect effect: OR:1.03, 95%CI 1.01-1.05).
Conclusions: The observation that lower serum magnesium levels are not only associated with diabetes but also with prediabetes makes reverse causation less likely. The effect of serum magnesium on prediabetes is partially mediated through insulin resistance. Common variants in magnesium transporter genes modify prediabetes risk by in uencing serum magnesium. Hence, serum magnesium may be a modi able risk factor for diabetes.
FR-PO418
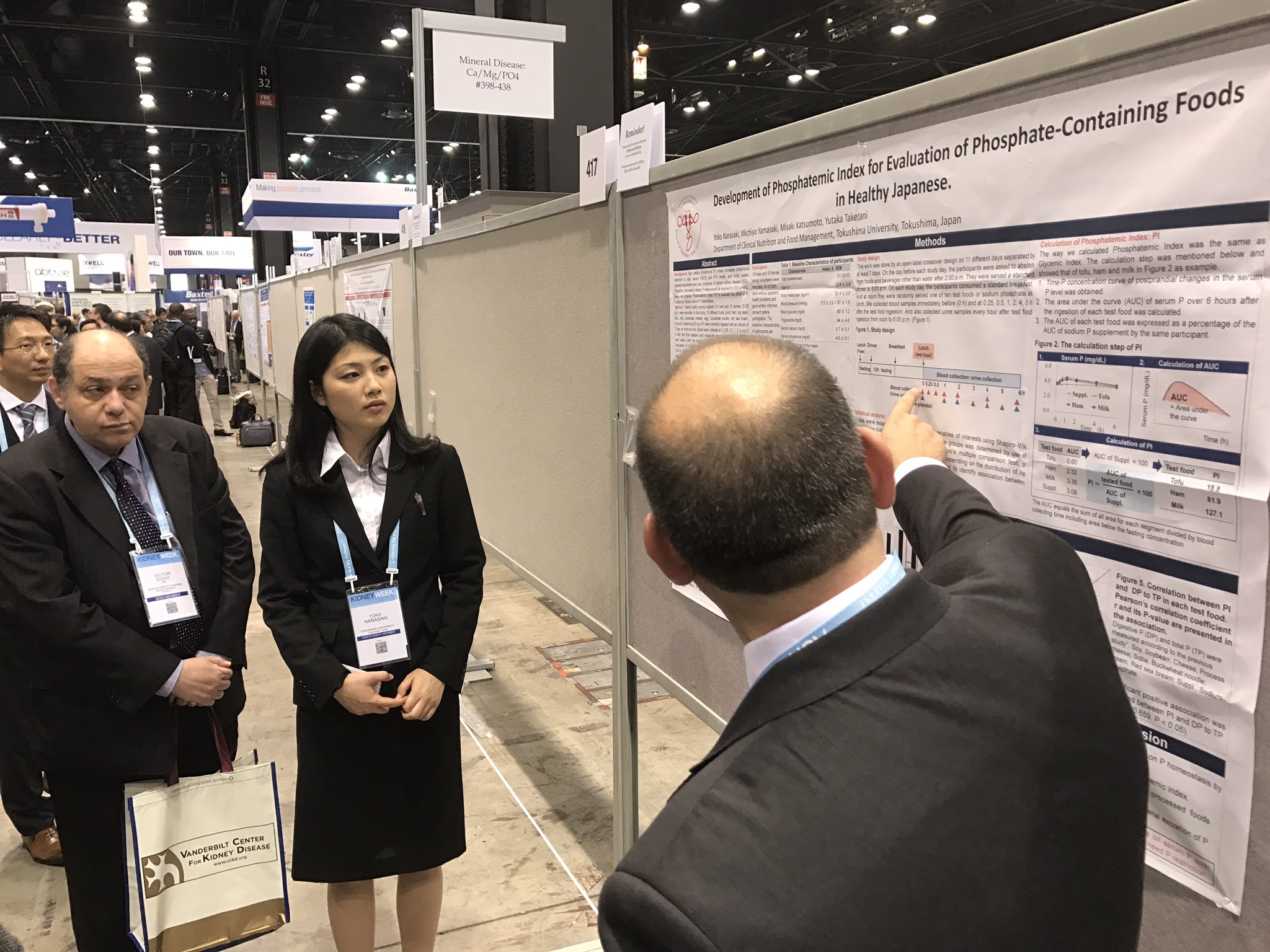
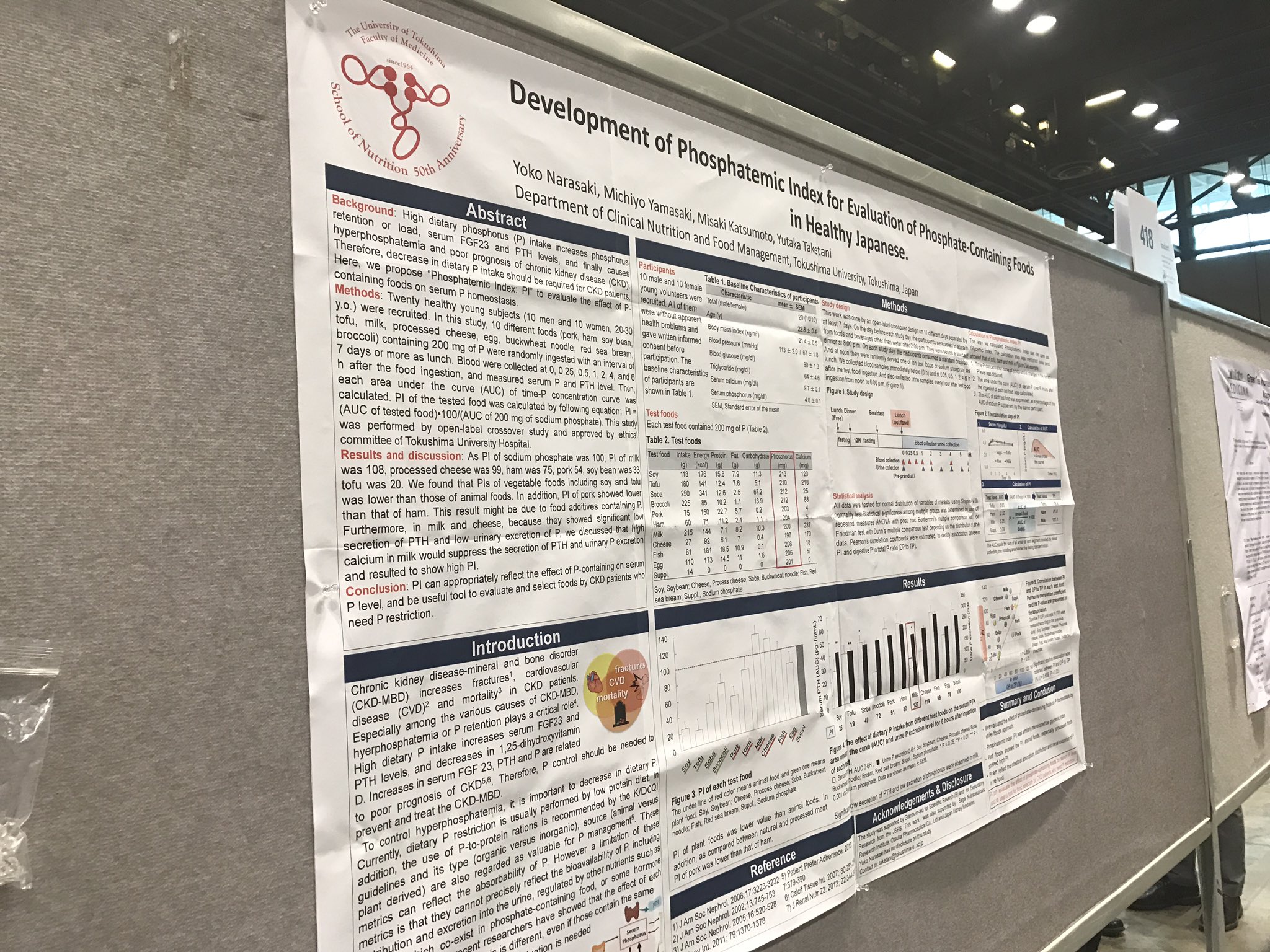
Development of Phosphatemic Index for Evaluation of Phosphate- Containing Foods in Healthy Japanese Yoko Narasaki, Michiyo Yamasaki, Misaki Katsumoto, Yutaka Taketani. Clinical Nutrition and Food Management, Tokushima Univ, Tokushima, Japan.
Background: High dietary phosphorus (P) intake increases phosphorus retention or load, serum FGF23 and PTH levels, and nally causes hyperphosphatemia and poor prognosis of CKD. Therefore, decrease in dietary P intake should be required for CKD patients. Here, we propose “Phosphatemic Index: PI” to evaluate the effect of P-containing foods on serum P homeostasis.
Methods: Twenty healthy young subjects (10 men and 10 women, 20-30 y.o.) were recruited. In this study, 10 different foods (pork, ham, soy bean, tofu, milk, processed cheese, egg, buckwheat noodle, red sea bream, broccoli) containing 200 mg of P were randomly ingested with an interval of 7 days or more as lunch. Blood were collected at 0, 0.25, 0.5, 1, 2, 4, and 6 h after the food ingestion, and measured serum P and PTH level. Then, each area under the curve (AUC) of time-P concentration curve was calculated. PI of the tested food was calculated by following equation: PI = (AUC of tested food) 100/(AUC of 200 mg of sodium phosphate). This study was performed by open-label crossover study and approved by ethical committee of Tokushima University Hospital.
Results: As PI of sodium phosphate was 100, PI of milk was 108, processed cheese was 99, ham was 75, pork 54, soy bean was 33, tofu was 20. We found that PIs of vegetable foods including soy and tofu was lower than those of animal foods. In addition, PI of pork showed lower than that of ham. This result might be due to food additives containing P. Furthermore, in milk and cheese, because they showed signi cant low secretion of PTH and low urinary excretion of P, we discussed that high calcium in milk would suppress the secretion of PTH and urinary P excretion and resulted to show high PI.
Conclusions: PI can appropriately re ect the effect of P-containing foods on serum P level, and be useful tool to evaluate and select foods by CKD patients who need P restriction.
Funding: Pharmaceutical Company Support - Otsuka Pharmaceutical Co., Ltd, Government Support - Non-U.S.
FR-PO433
Acid-Base and Phosphorus Homeostasis in Chronic Kidney Disease: Results from the Chronic Renal Insufficiency Cohort (CRIC) Study
Pascale Khairallah, Tamara Isakova, John R. Asplin, L. Lee Hamm,4 Mirela A. Dobre, Mahboob Rahman, Kumar Sharma, Mary B. Leonard, Edgar R. Miller,8 Bernard G. Jaar, Carolyn S. Brecklin, Wei Yang,10 Harold I. Feldman, Myles S. Wolf, Julia J. Scialla.
Duke Univ; Northwestern Univ; Litholink Corporation; Tulane Univ; 5Case Western Reserve Univ; Univ of California San Diego; Stanford Univ; Johns Hopkins Univ; Univ of Illinois; Univ of Pennsylvania.
Background: Kidneys excrete acid either as ammonium or as titratable acids (TA) that use phosphate as a buffer. In CKD, impaired ammoniagenesis promotes metabolic acidosis. In vitro, acidosis stimulates phosphaturic hormones, parathyroid hormone (PTH) and broblast growth factor 23 (FGF23), possibly to increase buffer for TA. These in vitro ndings have not been con rmed in humans.
Methods: In 980 CRIC participants with CKD (eGFR 44±14 ml/min/1.73m2), we measured net acid excretion (NAE) in 24h urine, potential renal acid load (PRAL) by food questionnaire, and serum bicarbonate. Using adjusted linear and log-linear regression, we modeled associations between acid parameters and 24h urine phosphorus, serum phosphorus, FGF23, and PTH.
Results: 24h urine phosphorus was higher at higher NAE, higher PRAL, and lower bicarbonate (all p-trends<0.05). Serum phosphorus was higher with higher NAE and lower bicarbonate (both p-trends=0.001). Higher NAE or PRAL were not associated with FGF23 or PTH. PTH, but not FGF23 (p=0.2), was 26% higher (P<0.001) when serum bicarbonate was < vs. ≥22meq/L. Results were similar if strati ed by eGFR categories, or if adjusted for iothalamate GFR, energy intake, urine urea nitrogen, or dietary phosphorus, where available.
Conclusions: Acid loading may augment phosphaturia in CKD to maintain NAE. Mechanisms may include higher serum phosphorus and not primary increase in FGF23 or PTH. Effect of acid load on phosphorus homeostasis must be tested in trials.
Funding: NIDDK Support
FR-PO458
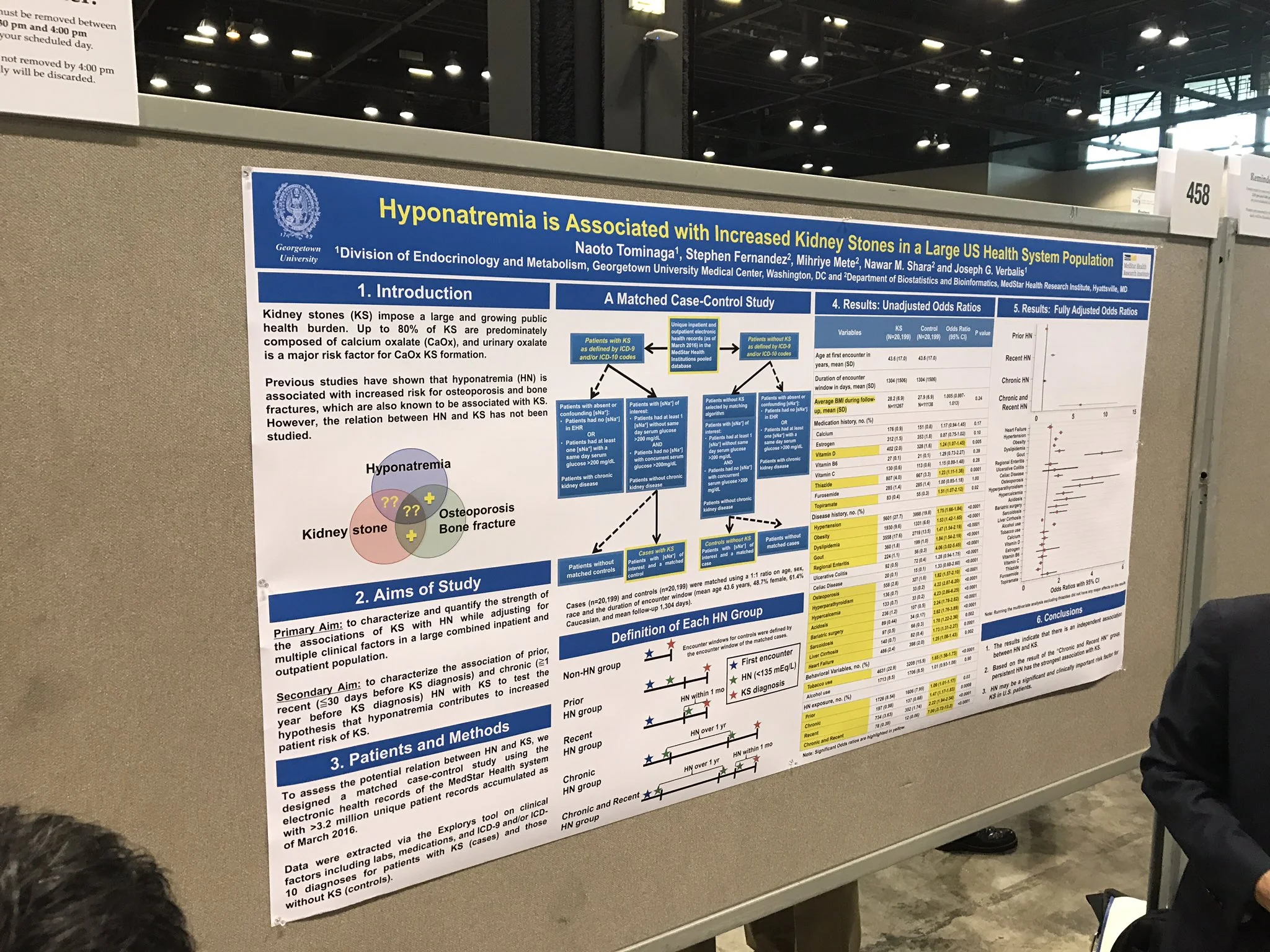
Hyponatremia Is Associated with Increased Kidney Stones in a Large U.S. Health System Population Naoto Tominaga, Stephen Fernandez, Mihriye Mete, Nawar M. Shara, Joseph G. Verbalis.
Div of Endocrinology and Metabolism, Georgetown Univ Medical Center; Dept of Biostatistics and Bioinformatics, MedStar Health Research Inst.
Background: Kidney stones (KS) impose a large and growing public health burden. Up to 80% of KS is predominately composed of calcium oxalate (CaOx), and urinary oxalate is a major risk factor for CaOx KS formation. Previous studies have shown that hyponatremia (HN) is associated with increased risk for osteoporosis and bone fractures, which are also known to be associated with KS. It is therefore reasonable to hypothesize that HN may be related to the occurrence of KS.
Methods: To assess the potential relationship between HN and KS, we designed a matched case-control study using the electronic health records of the MedStar Health system with more than 3.2 million unique patient records accumulated as of March 2016. Data were extracted via the Explorys tool on clinical factors including labs, medications, and ICD-9 and/or ICD-10 diagnoses for patients with KS (case) and those without KS (control).
Results: Cases (n=21,232) and controls (n=21,232) were matched with a 1:1 ratio on age, sex, race and the duration of encounter window (mean age 44.3 years, 48.4% female, 61.1% Caucasian and mean follow-up days 1345 days). Case and control exposures for each of the HN variables were de ned by serum [Na+] laboratory measurements reported within the encounter windows, and divided into 4 categories: prior HN, chronic HN, recent HN, and chronic and recent HN. Bivariate analyses using conditional logistic regression models con rmed increased risk of KS among patients with risk factors such as hypertension (OR=1.77, 95% CI 1.68-1.86). In addition, the risk of KS increased in all of HN categories: prior HN OR, 1.10 [95% CI, 1.03-1.18]; chronic HN OR, 1.43 [95% CI, 1.17-1.75]; recent HN OR, 2.30 [95% CI, 2.02-2.62]; chronic and recent HN OR, 5.18 [95% CI, 3.08-8.70]. Preliminary results of multivariate analyses adjusted for potential confounders con rm independent associations between HN and KS.
Conclusions: These results therefore support the hypothesis that HN is a signi cant and clinically important risk factor for KS in both inpatients and outpatients in the U.S.