Nephrol Dial Transplant. 2025 Dec 23;41(1):149-160. doi: 10.1093/ndt/gfaf154.
Cost-effectiveness of protein-restricted diets in advanced CKD
Vincenzo Bellizzi, Mario Fordellone, Luca De Nicola, Roberto Minutolo, Simona Signoriello, Giuseppe Quintaliani, Biagio Di Iorio, Paolo Chiodini
PMID: 40815268
DOI: 10.1093/ndt/gfaf154
What do we actually know about protein restriction?
For decades, nephrology has kept dietary protein restriction (DPR) on life support: never quite dead, never convincingly alive. Physiologically, the case is neat: less protein means less glomerular hyperfiltration, lower nitrogenous waste, less acidosis, and phosphate load, and theoretically slower scarring of nephrons (Sallstrom et al, Am J Physiol Regul Integr Comp Physiol, 2010). Experimental models and tortured MDRD secondary analyses based on achieved (and not intention-to-treat) have suggested that lower achieved protein intake is associated with slower GFR decline and longer time to kidney failure. (Levey et al, AJKD, 1996)
Clinically, things are murkier. Large RCTs of low-protein diets (LPD) versus “normal” protein intakes did not deliver the sort of clean, hard outcome wins we now expect from drug trials. Meta-analyses show that very low-protein diets (VLPD 0.3-0.4g/kd/day) supplemented with ketoacids may reduce the risk of kidney failure in non-diabetic CKD, but have uncertain effects on mortality, GFR slope, and quality of life (Jiang et al, Int Urol Nephrol. 2016| Li et al, Nutrients. 2019). In contrast, standard “moderate” LPDs (~0.55-0.6 g/kg/day) have shown at best modest or inconsistent effects on progression (Hahn et al, Cochrane Database Syst Rev, 2020| Bellizzi et al, Am J Clin Nutr, 2022 | Bawazir et al, J Braz Neph, 2025).
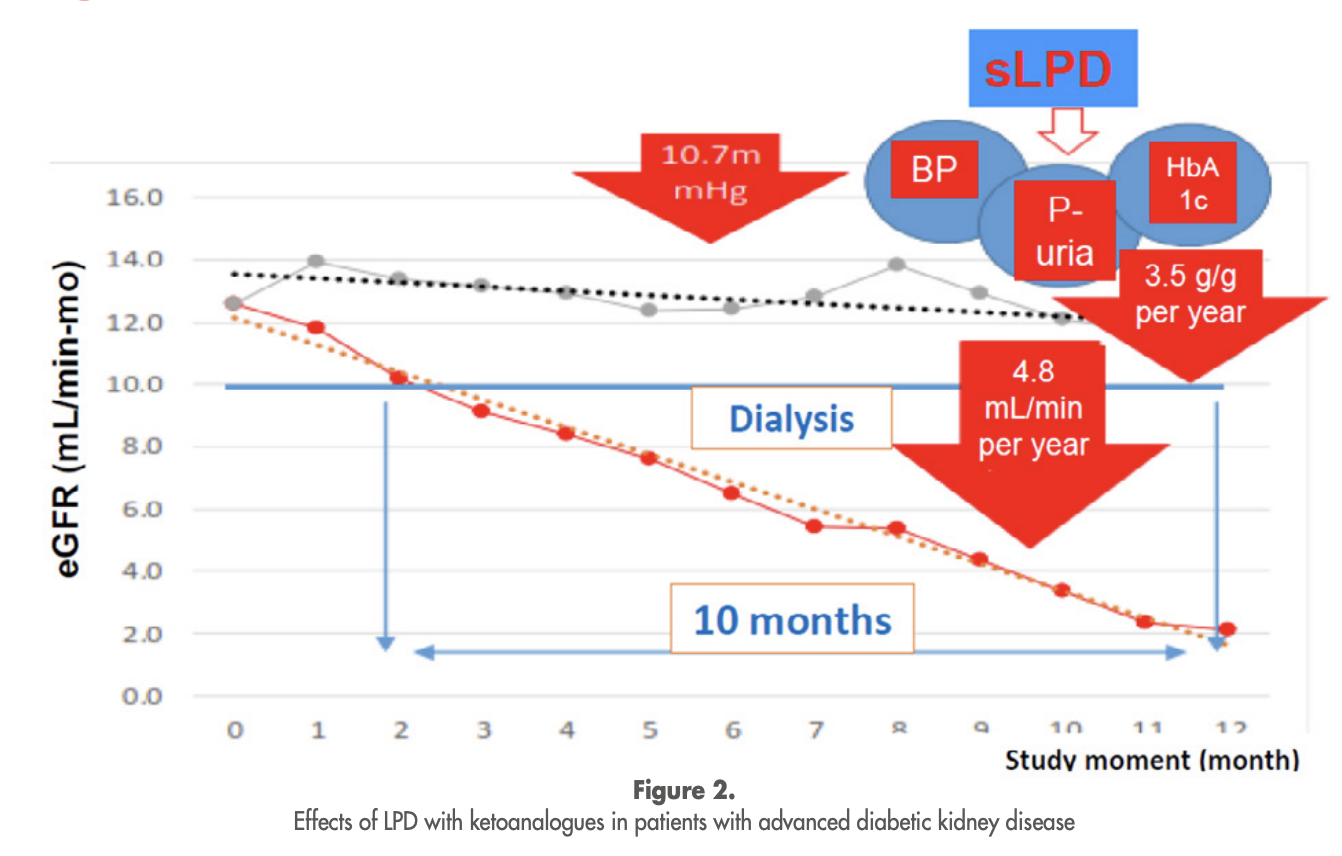
Figure 2. Effects of low protein diet in patients with advanced CKD, from European Renal Association e-seminar summary
Meanwhile, the practical obstacles are enormous and well known to anyone who has tried to run a serious renal diet program:
Adherence is the Achilles’ heel. In real-life cohorts, only about one-third of patients prescribed an LPD and one-quarter on a VLPD hit their protein targets (Bellizzi et al, Am J Clin Nutr, 2022)
Monitoring is intensive. Achieving and maintaining restriction without protein-energy wasting (PEW) needs regular dietitian input, frequent dietary assessment, and biochemical monitoring- luxuries in many clinics.
Nutritional risk is real. Patients with CKD spontaneously reduce energy and protein intake as the disease progresses; overzealous restriction risks accelerating sarcopenia and PEW, particularly in older or frail patients (Kopple et al, Kidney Int, 2000). Longer term follow up of the MDRD trial reported twice the mortality with LPD (Menon et al, AJKD, 2009)
Outdated Evidence: The LPD trials were run when we had no effective interventions for CKD progression, as we do now (see below).
Adapted from Obied W et al, Kidney360, 2022
Guidelines reflect this tension:
KDOQI (Ikizler et al, Am J Kidney Dis, 2020) takes a relatively assertive stance: for metabolically stable CKD G3-G5 (non-dialysis), it recommends an LPD of 0.55-0.6 g/kg/day, or a VLPD of 0.28-0.43g/kg/day plus ketoacid/amino acid analogues, under close supervision, to reduce ESKD/death risk and possibly improve quality of life (1A).
KDIGO is notably more cautious. It does not support low-protein diets alone (0.4-0.6g/kg/day) as a strategy to slow progression (KDIGO, Kidney Int, 2024| NephJC summary). Meta-analyses show little or no effect on kidney failure or eGFR change. Instead, KDIGO recommends 0.8 g/kg/day for most CKD G3-G5, and only “considers” very low, protein keto-supplemented diets for selected, motivated patients at high risk of kidney failure and under strict supervision. It explicitly warns against low/very-low protein diets in metabolically unstable patients or those with sarcopenia, cachexia, or undernutrition.
All of this plays out in an era where guideline-directed medical therapy (GDMT) for cardiorenal protection (maximal RAS inhibition, flozins, non-steroidal MRAs, and GLP1RAs) delivers large, reproducible reductions in kidney failure, cardiovascular events, and mortality across multiple RCTs. EMPA-KIDNEY (The EMPA-KIDNEY Collaborative Group, NEJM, 2023| NephJC summary), DAPA-CKD (Heerspink et al, NEJM, 2020| NephJC summary), and CREDENCE (Perkovic et al, NEJM, 2019| NephJC summary) alone show ~30-40% relative risk reductions in kidney disease progression and kidney failure, with benefits independent of diabetes in many patients (Nuffield Department of Population Health Renal Studies Group, Lancet, 2022).
Yet, in practice, both arms of this story underperform: strict protein restriction is rarely implemented to specifications, and GDMT uptake remains anemic (flozin eligible CKD cohort use often sits <30%) (Luyckx et al, Kidney Int, 2024). The question then, is not just “does a low-protein diet work?”, but “what is the most rational comparator and where should we spend our limited implementation capital?”
The study: a polished narrative constructed on an over-selected, hand-picked cohort
The cost-effectiveness article in question (Bellizzi et al, NDT, 2025) is, in many ways, the best-case scenario for protein-restricted diets.
The study was a prospective observational economic analysis built on previously established cohorts. It included outpatients with CKD stages 4-5 from Italian nephrology clinics. Patients were divided into:
Intervention (PRD): 62 patients with proven adherence to prescribed protein restriction: either LPD (0.6g/kg/day) or a VLPD (0.3g/kg/day plus ketoanalogues and protein-free special foods)
Controls: 123 matched patients on a “free” diet who had never been prescribed restriction.
All were followed for 5 years. Outcomes included time to ESKD, time to death, and a detailed accounting of direct and indirect costs during both non-dialysis CKD and dialysis.
The study declared no industry funding. However, one author reports consultancy/ speaker roles with Fresenius Kabi, the manufacturer of ketoanalogues (ketosteril), a central therapeutic component of supplemented very-low protein diets. Additional author ties include companies active in CKD, dialysis, anemia, and metabolic therapeutics.
The results follow a sequential structure that mirrors cohort construction, clinical outcomes, and downstream economic modelling.
The study initially included 1471 patients with CKD: 223 exposed to protein-restricted diets and 1248 controls on a free diet. Only patients with proven dietary adherence were retained in the intervention arm. This reduced the analytic PRD cohort to 63 individuals; after propensity matching, the final comparison consisted of 62 PRD and 123 controls.
Figure 1. Patient flow according to CKD PRD and free diet (control) groups, from Bellizzi et al, NDT 2025
Baseline characteristics in the matched sample were balanced:~67 years, ~60% male, diabetes ~29%, cardiovascular disease ~35%, BMI ~26kg/m2, and eGFR of ~16-17mL/min/1.73 m2, with roughly 45% on CKD stage 5 (table 1).
Follow-up duration was longer in the PRD cohort (~106 vs 89 months). Median time to kidney failure was longer with PRD 48.6 months vs 28.8 months (log-rank P=0.017).
Figure 2. Renal survival.Estimated time to ESKD for CKD patients on a PRD and a free diet, from Bellizzi et al, NDT 2025
Mortality outcomes followed a similar pattern. Median survival reached 107 months in PRD vs 86.6 months in control (P=0.004). Event proportions reflected these trajectories, with fewer deaths and later ESKD onset in the diet-adherent cohort.
Figure 3. Patient survival.Estimated time to overall survival for CKD patients on a PRD and a free diet, from Bellizzi et al, NDT 2025
Economic analyses were anchored in phase-specific cost structures. During the non-dialysis CKD phase, monthly costs were higher with dietary restriction: €383 for free diet, €507 for LPD, and €767 for VLPD. The incremental expenditure derived from protein-free foods, ketoanalogues supplementation, more frequent visits, and monitoring.
Once dialysis began, costs rose sharply to approximately €4150 (~$4900) per patient per month, exceeding conservative-phase costs by several fold.
Table 3. Comprehensive monthly costs in euros for the management of a CKD patient according to the Conservative (type of dietary treatment) and Substitutive (dialysis) treatments, from Bellizzi et al, NDT 2025
Two economic models were applied. In the theoretical model, which assumes survival to dialysis thresholds without accounting for attrition, protein restriction increased short-term costs but reduced long-term expenditures. Annual costs were ~80% higher early in CKD, but ~64% lower over longer horizons, producing an overall cumulative saving of ~43%.
Table 4. Theoretical and observed costs for therapies per patient, and outcomes differences between PRD and free diet groups, during early, long, and overall follow-up periods, from Bellizzi et al, NDT 2025
The observed model, incorporating deaths, dialysis initiation, and follow-up losses, showed smaller savings. Annual costs were reduced by ~30% in the short follow-up and ~28% in the longer follow-up, with overall reductions of ~25%. Cost divergence emerged as more control patients entered dialysis earlier and remained longer in the high expenditure phase.
Figure 6. Observed cumulative costs of CKD therapies (including both conservative and dialysis treatments) for a CKD patient on a PRD and a free diet, from Bellizzi et al, NDT 2025
Across analyses, the economic signal tracked dialysis exposure. Delayed kidney failure translated into a prolonged time in the lower-cost conservative phase and reduced cumulative dialysis spending.
Discussions: 21st-century view on protein restriction in CKD
Bellizzi and colleagues have produced an impressive piece of work concerning the economics of a treatment that belongs to another century. They take a small, exquisitely selected subgroup of patients with stage 4-5 CKD who are willing to live for years inside a narrow biochemical corridor of protein intake, surround them with dieticians, protein-free foods, and ketoanalogues, and document what happens: dialysis comes later, remarkably death comes later, and the health system spends less on machines and membranes per unit of survival. Within that closed system, the logic is flawless. The unease comes from recognizing that the system itself is an artifact of a therapeutic worldview that has already been displaced.
The lineage is not in doubt by far. Addis’s early experiments with severe nitrogen restrictions made it clear that, if one supply can be coaxed in a state of “minimum endogenous nitrogen metabolism” and held there, it will be at the cost of “mounting aversion and eventual loss of courage on the part of the subject” (Smith M, J Biol Chem, 1926| Obeid et al, Kidney360, 2022). The Giordano-Giovannetti diet translated into clinical practice 18-20 g of protein a day, carefully balanced amino acids, and in return a striking disappearance of classic gastrointestinal torments of uremia (vomiting, hiccups, anorexia, diarrhea), while blood urea levels fell and patients enjoyed a somewhat longer "comfortable life”…… before inexorable death (Berlyne et al, Lancet, 1965). The promise was never that the kidney would scar less; it was that the dying would be biochemically tidier. Barry Brenner’s hyperfiltration hypothesis then conferred upon this ascetic tradition a physiologist’s elegance. High protein intake was shown in animals to increase single nephron GFR and intraglomerular pressure; protein restriction mitigated these surges and delayed the development of glomerulosclerosis (Brenner et al, NEJM, 1982| Brenner et al, Kidney Int, 1996). In the absence of dialysis and any drug capable of altering glomerular hemodynamics, it was rational and almost inevitable, to turn the plate.
What happened when this physiological conviction was finally submitted to the discipline of the randomized human trials is, retrospectively, not flattering. Across Rosman (Lancet, 1984), Ihle (NEJM, 1989), Locatelli (Lancet, 1991), Hansen (Kidney Int, 2002), Cianciaruso (AJKD, 2009), and, most decisively MDRD, the pre-specified endpoints are remarkably stubborn. Low-protein diets do not reliably reduce the rate of dialysis initiation, nor do they produce a sustained, clinically important slowing of GFR decline when GFR is measured directly rather than inferred from creatinine (Mehrotra, Miner Electrolyte Metab, 1999| Bawazir et al, J Braz Neph, 2025). In MDRD, low versus “usual” protein in patients with moderate CKD (0.58 vs 1.3 g/kg/day) and very low vs low protein in advanced CKD (0.28 vs 0.58 g/kg/day, with ketoacid supplements) failed to reduce time to ESKD or death in intention-to-treat analysis (Levey et al, AJKD, 1996). The methodological innovation of MDRD (measured iothalamate clearance) had the unfortunate virtue of showing that much of the apparent improvement seen in earlier creatinine-based studies was not a slowing of nephron loss at all, but a biochemical illusion created by reduced creatinine generation under protein restriction (Levey et al, AJKD, 1996| Bawazir et al, J Braz Neph 2025).
Meta-analysis has not rescued the paradigm. Kasiske et al (AJKD, 1998) combined more than 1900 patients and found that protein restriction slowed GFR decline by roughly 0.5 ml/min/year on average, a statistically significant but clinically modest effect that did not translate into a robust reduction in kidney failure. Hahn’s Cochrane review concluded that low-protein diets probably have little or no effect on the composite of kidney failure or death in non-diabetic CKD, and that the seemingly beneficial effect of VLPD over-simplifies avoiding high protein and is fragile and confounded (Hahn et al, Cochrane Database Syst Rev, 2020). Trials in diabetic nephropathy are even less forgiving: a small signal here, an equivocal slope there, none of it rising to the standard we routinely apply to therapies that claim to modify renal prognosis. The hyperfiltration story is not disproven so much as rendered insufficient: glomerular physics is not destiny when one is constrained by human behavior and the ethics of long-term semi-starvation.
Landmark randomized, controlled trials of protein restriction in chronic kidney disease, table adapted from Bawazir et al, J Braz Neph 2025
Even this would be tolerable if the intervention itself were benign. From the earliest trials onward, the cost of sustained protein restriction has been paid in acceptance and flesh. In Rosman’s multicenter study, a third of patients rated the low-protein diet bad at 3 and 6 months, and substantial numbers withdrew for intolerance or non-cooperation (Rosman et al, Lancet, 1984). Locatelli’s comparison of 0.6 vs 1 g/kg/day reported 64 withdrawals, the majority for inability or unwillingness to comply with low-protein food (Locatelli et al, Lancet, 1991). Even with a carefully curated population, both protein and energy intake drifted downward, accompanied by reductions in body weight, arm muscle area, and transferrin, even as albumin remained in the normal range and gave the comforting appearance of safety (Chauveau et al, AJKD, 1999). Mehrotra’s review of these data is blunt: in the best hands, in non-diabetic, non-frail patients, long-term protein diets produce small but consistent signals of subclinical malnutrition, and there is every reason to believe that the margin of error is narrower in older and more comorbid patients who now populate our CKD clinics (Mehrotra, Miner Electrolyte Metab, 1999). To persist in calling such regimens conservative care as patients involuntarily lose lean mass feels, at some point, more like a euphemism than medicine.
It is into this already crowded and ambiguous landscape that Bellizzi’s cost-effectiveness analysis arrives. The authors do something few have had the patience or infrastructure to do: they identify only those patients who actually adhere to a prescribed low- or very-low protein diet, achieve intake within 0.2 g/kg/day of the target, and follow them over five years, comparing propensity- matched patients who never received dietary restriction. Roughly a quarter of those originally prescribed the diet qualify as “adherent” by this definition. Under the care of Italian nephrology units with specialist dietitians and access to protein-free medical foods, these adherent patients enjoy significantly longer times to ESKD (48.6 vs 28.8 months) and death (107 vs 86.6 months) than their free eating counterparts. Their monthly costs during the non-dialysis phase are higher (€507 for LPD and €767 for supplemented VLPD vs €383 with no diet), but once dialysis begins at roughly €4150 per month, the arithmetic becomes trivial (Bellizzi et al, NDT, 2025). This kind of analysis is picking low protein cherries to the extreme. Data obtained from only adherent patients (a kind of per-protocol, not intention to treat), and even that from an observational study, should not satiate anyone. It only provides biased estimates. Patients adherent to a specific diet usually have other underlying characteristics (education, socioeconomic status, adherence to other therapeutic interventions) that make for the real mechanistic effect.
If one’s worldview is constructed around dialysis as the unavoidable abyss, and grams of protein as one of the few adjustable variables, this is triumphant. But it’s very coherence exposes how far removed it is from what now constitutes contemporary CKD care. The comparator is not “modern GDMT with or without serious dietary restrictions”. It is “diet plus a largely pre-GDMT standard of care” versus “no diet, plus same”. The physiological problem the diet is solving - hyperfiltration and its hemodynamic sequelae- is the same problem now addressed, more potently and reproducibly, by RAAS inhibitors and flozins (Brenner et al, Kidney Int, 1996| Iman et al, J Ren Nutr, 2025| Wanner et al, Adv Ther, 2025). ACE inhibitors and ARBs lower efferent arteriolar resistance, reducing intraglomerular pressure, proteinuria, and progression. Flozins enhance distal sodium delivery, engage tubulo-glomerular feedback, and constrict the afferent arteriole, producing an immediate drop in GFR followed by a slower decline- a kinetic pattern eerily reminiscent of the hoped-for effect of low protein diets, but achieved without asking the patient to consider every morsel of food. Large trials like EMPA-KIDNEY (The EMPA-KIDNEY Collaborative Group, NEJM, 2023) and DAPA-CKD (Heerspink et al, NEJM, 2020) show a 30-40% relative risk reduction in kidney failure and sustained GFR decline with parallel reductions in heart failure and cardio-vascular death, on top of RAS blockade, in diabetic and non-diabetic CKD alike (Nuffield Department of Population Health Renal Studies Group, Lancet, 2022). Ns-MRAs and GLP1RA add further, if more population-specific, renal and cardiovascular protection.
KDIGO 2024 recognizes this hierarchy explicitly. It recommends protein intake around 0.8 g/kg/day for most adults with CKD, warns against low- or very low protein diets in metabolically unstable patients, and judges the certainty of evidence for such diets on hard outcomes to be low and very low (KDIGO, Kidney Int, 2024| NephJC summary).
The same guideline confers its highest grade of recommendation (1A) to flozins as kidney-protective therapy and frames RAASi, SGLT2i, ns-MRA, and GLP1RA as the backbone of disease-modifying treatment in cardio-renal disease. KDOQI nutrition guidelines, by contrast, emerge from a different epistemic culture (Ikizler et al, AJKD, 2020). It grants strong recommendations to LPD and VLPD with or without ketoanalogues in CKD stages GA3 and G5, leaning heavily on small RCTs with surrogate endpoints, and embeds these diets as central tools of kidney protection rather than futile adjuncts. The divergence is not about data, is about what one wrongfully counts as a convincing signal, and how one weighs mechanistic plausibility against existence of therapies that have already cleared much higher evidentiary thresholds. A recent meta-analysis shows that adding ketoanalogues to LPD or VLPD improves GFR trajectories, reduces urea and phosphate, and maintains albumin and muscle indices over a median of 13 months (Chen et al, J Nephrol, 2024). There’s, however, no significant reduction in all-cause mortality and no statistically robust reductions in ESKD across the full CKD population. The trials are small, short, and methodologically heterogeneous. Safety is conditional on adequate caloric intake and careful monitoring, and the supplements themselves are expensive, in some settings approaching the cost of a year of dialysis when all inputs are counted (pills, dietitian time, repeat visits). It is telling that an expert RAND/UCLA panel, faced with this evidence, declared ketoanalogue supplementation “appropriate” in many scenarios on the basis of consensus rather than outcome data (Saavedra-Fuentes et al, Nutrients, 2024). This is not the profile of a foundational therapy, is the profile of an intricate scaffold built on still-damp concrete.
What Bellizzi et al therefore show, perhaps more starkly than they intended, is not that protein restriction should reclaim the center of CKD management, but that it can still perform impressively under highly contrived conditions. If one is willing to build and fund an infrastructure of specialist dietitians, subsidized protein-free products, monthly visits, and detailed intake monitoring; if one is content to compare this against a backdrop in which RAASi and flozins are neither universal nor always optimized, and you choose to include data from patients who follow through all this - then yes, a few years of dialysis may be bought back and the balance sheets will look better. The absurdity, viewed from the vantage point of 2025, is that we continue to invest experimental and policy energy into perfecting this apparatus of controlled deprivation while simultaneously failing to deliver once-daily pills with high-certainty, trial-proven benefits to vast numbers of eligible patients. It’s easier to model the economics of a gram of protein than to solve the organizational problem of why so many patients with significant albuminuria never see a flozin.
LPD and VLPD are less marginal than outdated. In advanced CKD, asking patients to metabolically restrict themselves makes little sense when the same hemodynamic targets can be addressed pharmacologically. The so-called “ideal candidate”- stable, motivated, dietitian-supported- highlights how narrow, and not how relevant the strategy remains. At scale, it doesn’t hold. Adherence fails, infrastructure varies, and patients with advanced CKD are already nutritionally vulnerable. Dialysis delay that comes with muscle loss or frailty is a questionable trade. From that angle, Bellizzi et al reads like a detailed documentation of what dietary deprivation could achieve before pharmacological protection became routine.
Summary by
Cristina Popa
Reviewed by
Swapnil Hiremath and Brian Rifkin