#NephJC Chat
Tuesday Feb 20 9 pm Eastern
Wednesday Feb 21 8 pm BST, 12 noon Pacific
N Engl J Med. 2018 Feb 1;378(5):428-438. doi: 10.1056/NEJMoa1700993.
History of Childhood Kidney Disease and Risk of Adult End-Stage Renal Disease.
Calderon-Margalit R, Golan E, Twig G, Leiba A, Tzur D, Afek A, Skorecki K, Vivante A.
PMID: 29385364 Full Text at NEJM (click here for full free text)
Also see related Editorial by Julie Ingelfinger and Video Summary
Introduction
Imagine you’re in your pediatric nephrology clinic seeing a 17 year old patient that you’ve been following for years. He had a UPJ obstruction that was repaired in infancy with residual hydronephrosis on that side and now has normal kidney function and blood pressure. What do you tell him about his long term risk of kidney disease? Do you refer him to a nephrologist or internist to transition to adult care? What recommendations should you give for monitoring? Now what if he had a single episode of pyelonephritis as a child instead of UPJ obstruction? Or Henoch-Schonlein purpura (HSP) nephritis (I’ll never call it IgA vasculitis!) that completely resolved? What is his long term risk of ESKD in those situations? This large population-based cohort study from Israel attempts to answer some of these questions, however I think more questions may be generated from this publication than answered.
A number of kidney disorders affect children yet may leave a child with no visible signs or symptoms of kidney disease including congenital anomalies of the kidney and urinary tract (CAKUT), pyelonephritis, and glomerular disorders. CAKUT are common in childhood affecting 0.4 to 4.0 children per 1,\000 and include congenital single kidney, unilateral hypoplasia/dysplasia, ectopic kidneys, horseshoe kidney, hydronephrosis, hydroureter, UPJ stenosis, UVJ stenosis, among others. The incidence of pyelonephritis in childhood is challenging to determine due to the difficulty in distinguishing between upper and lower tract disease.
The prevalence of upper or lower tract UTI is about 1% in school age girls and 0.03% in school age boys.
Glomerular diseases are rare and affect 0.1 to 2 children per 100,000 annually and may include minimal change nephrotic syndrome, post-infectious glomerulonephritis, HSP nephritis, and others.
The long-term outcomes of children with each of these categories of kidney disorders are unclear, making counselling about long-term risks of chronic kidney disease and ESKD difficult. This study was designed to determine the long term kidney outcomes in each of these categories of childhood kidney disease.
Methods
Israeli military health screening records combined with the Israeli ESKD database are a treasure trove of population health information. The authors previously published a study in JAMA in 2011 showing that adolescents with persistent microscopic hematuria at military screening (0.3% of overall cohort) had increased risk of ESKD over 22 years of follow up. They expanded their study this time to examine risk of ESKD in potential military conscripts with a history of childhood kidney disease that had resolved.
They examined records of 1,521,501 conscripts between 1967-1997.
They excluded 22,596 individuals with a known increased risk of ESKD (diabetes, lupus, vasculitis, cancer, hypertension, or impaired GFR).
Then they identified cohorts of participants with a history of:
CAKUT (N=3198)
Pyelonephritis (N=7231) defined as a single or recurrent episode with or without evidence of scarring
Glomerular disease (N=8611). They had to be disease free for 1 year prior to evaluation
Members of the cohort had to have a normal blood pressure, no proteinuria, and normal serum creatinine at the time of military screening examination. All diagnoses were confirmed by a board certified nephrologist.
The military conscript records were then linked with the Israeli ESKD registry between 1980 and 2014, and life tables were constructed to show the cumulative incidence of ESKD in each of these categories. Cox proportional-hazard models were used to determine the hazard ratios for ESRD in each of the categories and adjusted for factors that can affect ESRD risk.
Results
The cumulative incidence of ESKD in adults with a history of any childhood kidney disease (CAKUT, pyelonephritis, glomerular disease) was greater than those with no history of childhood kidney disease.
Figure 2 from Calderon-Margalit et al, NEJM 2018
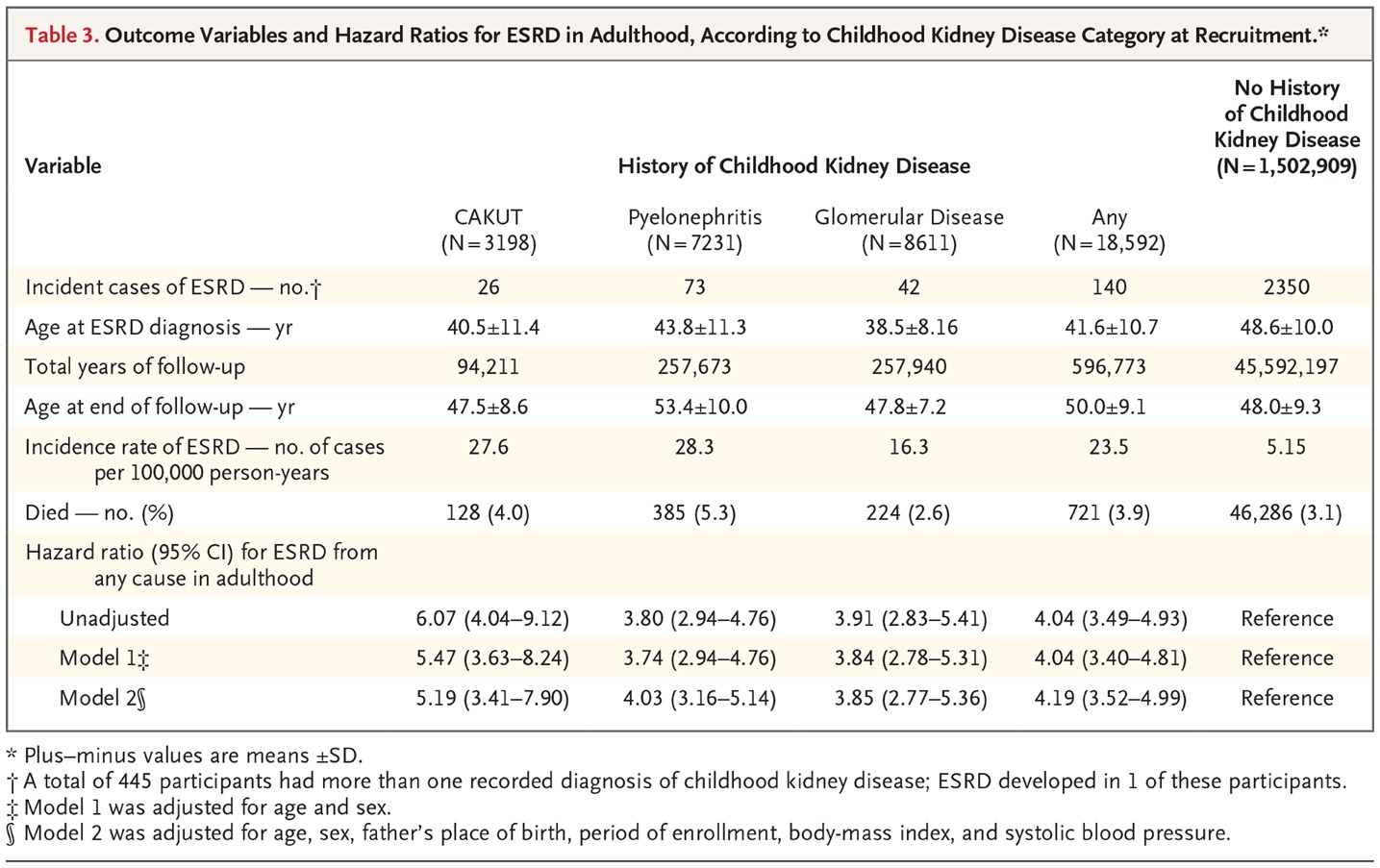
Having a history of any of the three categories of childhood kidney disease conferred an increased hazard ratio for ESRD of 4.19 (95% CI 3.52-4.99) even if blood pressure, creatinine, and urine protein were normal in late adolescence. Risk was similar in magnitude when each group was analyzed separately. The authors demonstrate increased adjusted hazard ratio for ESRD of 5.19 (95% CI 3.41-7.90) for CAKUT, 4.03 (95% CI 3.16-5.14) for pyelonephritis, and 3.85 (95% CI 2.77-5.36) for glomerular disease.
Table 3 from Calderon-Margalit et al, NEJM 2018
Discussion
Many pediatric nephrologists will not be surprised by these findings. We have long recognized that decreased nephron mass at birth due to prematurity leads to increased risk of hypertension and CKD later in life (the Brenner hypothesis). Thus, decreased nephron mass due to CAKUT or due to scarring from pyelonephritis or glomerular inflammation should also increase risk of ESRD long term. These results provide us some backing for our usual recommendations for yearly follow up after transition out of the pediatric clinic. Is primary care follow up adequate for these patients? How can we educate primary care physicians to ask about childhood kidney disease and to monitor appropriately? I can imagine a lot of blank stares when a provider asks a 23 year old new patient if they ever had a history of pyelonephritis in their life. This is another reason why universal medical records would be useful. A new provider shouldn’t have to page through 180 pages of printed EPIC records to find that one time a patient had PSGN or to find their ultrasound report from when they were 3 years old.
One study weakness is that specific details about diagnoses within these broad umbrella categories of childhood kidney disease are lacking, leaving me wanting more information and with concerns about generalizability of results. A conscript with 1-2 episodes of steroid sensitive minimal change disease 10 years ago is analyzed in the same “glomerular disease” category as a child with PSGN or HSP nephritis who may be expected to have more permanent injury in some glomeruli depending on the severity of the initial episode. I recognize that absolute numbers with ESRD are small, precluding any sort of meaningful subgroup analysis, however at least a listing of primary diagnoses for each subgroup would have been useful in interpreting the results. Was the glomerular group comprised of 90% minimal change? 90% PSGN? How many patients with a single episode of pyelonephritis were included in that cohort?
The authors should be commended for their work. It’s not every day a pediatric nephrology topic appears in the New England Journal of Medicine. They provide important population risk information for children with seemingly resolved childhood kidney disease. The next step is to move toward understanding risk on an individual level.