#NephTrials
Friday, April 10th 2026, 12 pm Eastern- Space on X
Contemp Clin Trials. 2025 Jun:153:107911. doi: 10.1016/j.cct.2025.107911. Epub 2025 Apr 6.
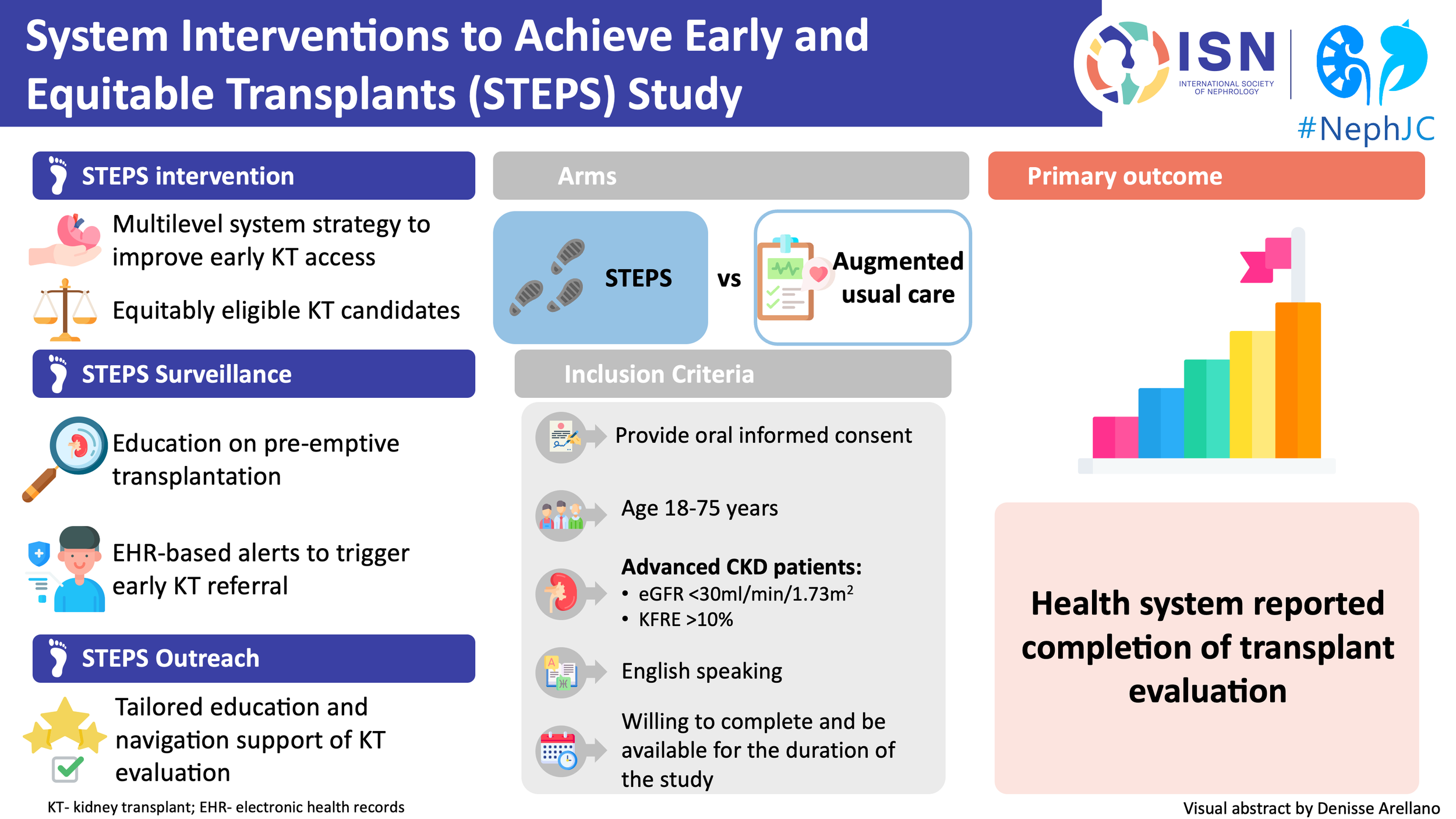
System Interventions to Achieve Early and Equitable Kidney Transplants (STEPS): Protocol for STEPS, a randomized comparative effectiveness clinical trial
PMID: 40199386
#NephTrials is an ongoing initiative between #NephJC and the ISN-ACT group. The ISN-ACT (Advancing Clinical Trials) is an International Society of Nephrology (ISN) initiative to encourage existing infrastructures within ISN to improve the global nephrology community’s participation in clinical trial research.
Introduction
Kidney transplantation remains the optimal therapy for kidney failure, yet access is neither early nor equitable. Despite decades of progress, only a minority of patients reach the transplant pathway before dialysis, and even fewer complete the steps required to receive a transplant. This gap reflects a fragmented process: patients are often identified late, inadequately informed, inconsistently referred, and infrequently able to complete the complex evaluation processes. As highlighted in the STEPS protocol, transplantation is not a single event but a multistep pathway, and failure at any single step can prevent patients from ever reaching the waitlist (Boulware et al,.Contemp Clin Trials, 2025).
Non-medical barriers, including health literacy, provider communication, system capacity, and logistical constraints, play a central role in limiting access to early transplant evaluation (Harding et al, Transplant Rev, 2021). At the same time, structural inequities remain deeply embedded in the system. Community-level vulnerability independently reduces access to living donor kidney transplantation (LDKT) and only partially explains racial disparities, with disproportionately worse effects among Black recipients (Killian et al, JAMA Surg, 2021). Meanwhile, these observations reinforce the need to move beyond simply describing inequities toward actively addressing them, as emphasized by Patzer, who highlighted bridging the gap between improved outcomes and unequal access to transplantation (Patzer R, JASN, 2024). More recently, these disparities have been reframed through an intersectional lens, recognizing how overlapping systems—race, geography, socioeconomic status, and healthcare infrastructure—interact to shape access long before transplant listing is even considered (Nonterah CW, Transpl Int, 2024).
Interventions targeting single components of the pathway have produced modest and inconsistent gains. Even multicomponent strategies, such as the pragmatic EnAKT LKD cluster randomized trial, improved selected steps but did not increase overall progression towards transplantation, underscoring the cumulative nature of barriers (Garg et al, JAMA Internal Medicine, 2023| NephJC summary | FF episode).
The STEPS trial (System Interventions to Achieve Equitable and Early Transplants) emerges in response to this gap. The trial tests a coordinated system-level intervention that combines automated electronic health record surveillance with proactive outreach and longitudinal patient navigation. Rather than improving isolated components, STEPS evaluated whether redesigning the transplant pathway itself can increase completion of the transplant evaluation and reduce disparities in access to kidney transplantation.
Adapted table 1 and 4 of STEPS protocol, from Boulware et al, Contemp Clin Trials, 2025. The critical roadblocks and features of STEPS to early kidney transplants
How do you test a system-level intervention? The STEPS Trial Design
STEPS is a pragmatic, randomized comparative effectiveness trial designed to evaluate whether a coordinated health system intervention can improve access to kidney transplantation. The study examines whether redesigning how patients are identified, referred, educated, and navigated can change progression through the transplant pathway. The unit of intervention is the system, but the unit of inference is the patient, achieved through individual randomization embedded in routine care.
The STEPS intervention combines active electronic health record (EHR)–based surveillance with patient-centered outreach delivered by transplant social workers and coordinators. The surveillance registry was updated nightly within the EPIC electronic health record, identifying adults aged 18-74 years with CKD using objective criteria (two eGFR values < 60 ml/min/1.73 m²), followed by risk-based triggers: alerts at eGFR <30 ml/min/1.73 m² or ≥ 10% two-year risk using the Kidney Failure Risk Equation, and transplant referral prompts at eGFR ≤ 20 ml/min/1.73 m². This approach standardizes candidate identifications and reduces reliance on clinician recognition- one of the major sources of variability in previous studies. This multilevel approach targets key barriers across the transplant pathway- particularly among Black and rural individuals with advanced chronic kidney disease who are not yet on dialysis.
Importantly, the trial was conducted across three large and diverse U.S. health systems: Duke University Health System (DUHS), Geisinger Health System (GHS), and the University of Mississippi Medical Center (UMMC)- selected to capture different structural barriers to transplant access. DUHS and UMMC care for a high proportion of Black patients and rural populations, groups historically underrepresented in transplant access. Black individuals accounted for over 50% of kidney transplant recipients at DUHS and more than 70% at UMMC. In contrast, GHS has a highly rural population, enabling the study to also evaluate geographic disparities independent of race. By embedding the intervention within health systems that reflect real-world inequities in race and geography, STEPS is positioned not only to evaluate effectiveness but also to generate insights into how system-level interventions perform across populations disproportionately affected by limited access to transplantation.
Can redesigning the health system improve access to kidney transplantation?
Following identification through the surveillance registry and enrollment, participants enter a structured pathway that reflects both methodological rigor and real-world applicability. Randomization was performed using a centralized, stratified block design, accounting for clinical site, race, and kidney function (eGFR ≤20 vs >20 ml/min/1.73m²), ensuring balance across factors closely linked to disparities in transplant access.
Figure 2. Eligibility assessments and randomization process from Boulware et al, Contemp Clin Trials, 2025
The trial successfully completed enrollment of 1,168 participants between March 2022 and March 2024. The trial was registered at ClinicalTrials.gov (NCT05014256). The planned sample size of approximately 1150 participants is expected to provide >80% power to detect a treatment effect for the primary outcome overall and specifically among Black participants, with conservative assumptions based on usual-care estimates across participating sites.
Once randomized, participants followed one of two care pathways. Participants assigned to arm 1, augmented usual care, continued to receive standard clinical management supported by EHR-based surveillance and best practice alerts, prompting clinicians to initiate nephrology referral and transplant evaluation when guideline thresholds are met. Participants in arm 2, STEPS intervention arm, receive a coordinated, multilevel intervention that integrated EHR-based identification, structured education, proactive outreach, and longitudinal patient navigation, designed to address barriers that traditional care does not systematically overcome.
Adapted from table 4, distinguishing features of STEPS compared to augmented usual kidney care from Boulware et al, Contemp Clin Trials, 2025
A central component of STEPS is the integration of patient-centered educational and behavioral support strategies delivered by transplant social workers and nurse coordinators. Participants receive the “Living with Kidney Disease: All the Facts” booklet, developed to provide accessible, literacy-sensitive information about CKD progression and treatment options (Boulware et al, The PREPARE NOW Study, 2023). This is complemented by the TALK Social Worker Program, a structured intervention grounded in behavioral theory, which facilitates discussions about kidney transplantation, particularly LDKTs, among patients, their families, and providers (Boulware et al, AJKD, 2013). Through individualized and group sessions, the program helps patients identify personal, social, and logistical barriers while promoting informed decision-making and engagement in the transplant process.
Beyond education, STEPS incorporates active patient navigation, a critical element often missing in traditional care models. Transplant social workers and nurse coordinators maintain longitudinal contact with participants, assisting with scheduling, coordinating appointments, addressing financial and transportation barriers, and guiding patients through each step of the transplant evaluation. This coordinated approach reduces reliance on patient self-navigation, a well-recognized contributor to dropout along the transplant pathway.
Data collection in STEPS reflects its hybrid nature as both a clinical and behavioral intervention. Outcomes are assessed using electronic health records, administrative data, and centralized patient-reported measures collected at baseline and at 6, 12, and 18 months. The recruitment was centralized, having a dedicated data coordinating center to standardize the outcome ascertainment and minimize loss of follow-up.
The primary outcome is completion of the kidney transplant evaluation, defined using objective health system documentation, including evaluation completion, time to completion, listing decisions, or receipt of transplantation. This endpoint targets a critical bottleneck in the transplant pathway while avoiding confounding from organ availability. Secondary outcomes expand this perspective by examining intermediate steps along the pathway, including patient discussions with providers and family members, initiation and progression of transplant evaluation, and identification of potential living donors. Additionally, measures of patient knowledge, empowerment, and behavioral activation are included to better understand how the intervention influences decision-making and engagement.
Together, this design allows STEPS to move beyond traditional endpoints and evaluate whether a coordinated system-level intervention can meaningfully change both the trajectory and the experience of patients navigating the transplant pathway.
Adapted from table 5, data collection outcomes from Boulware et al, Contemp Clin Trials, 2025
Statistical analysis follows an intent-to-treat framework, preserving randomization and minimizing bias from differential engagement with outreach and navigation. The primary comparison evaluates completion of transplant evaluation between groups using regression models adjusted for stratification variables (site, race, and baseline kidney function). Time-to-event analyses will assess progression through the pathway, accounting for variable follow-up. Secondary outcomes will be analyzed using generalized linear models appropriate for outcome type, with prespecified subgroup analyses by race, rural residence, and kidney function to examine effects on disparities. Missing patient-reported outcomes will be handled using multiple imputation, and sensitivity analyses will evaluate robustness to differential follow-up.
The study was funded by the Patient-Centered Outcomes Research Institute (PCORI) and the National Institute of Diabetes and Digestive and Kidney Disease (NIDDK), with total project costs of approximately 12.95 million $, split between NIH (66%) and PCORI (54%). Investigators reported limited competing interests: one author served as an editor for the publishing journal, and another disclosed a consulting relationship and financial interest in a health coaching company.
Why STEPS Matters in Context?
In the absence of outcome data from STEPS, prior interventions offer important insights into bridging LDKTs disparities. Across studies, a consistent pattern emerges: interventions that target a single barrier can improve individual steps in the transplant pathway but rarely turn into sustained, system-wide gains.
Integrated multicomponent STEPS intervention adapted from Boulware et al, Contemp Clin Trials, 2025
Educational strategies, particularly when structured and guided, have demonstrated improvements in knowledge, readiness, and evaluation completion. For example, standardized transplant education programs have been associated with reduced disparities in access, while tailored interventions have improved patient engagement and decision-making (Patzer et al, CJASN, 2012; Arriola et al, Prog Transplant, 2025). Similarly, the navigator-based and culturally tailored programs have demonstrated meaningful benefits in donor identification and evaluation completion, particularly in disparity reductions. However, these effects are highly context-dependent and often difficult to reproduce at scale (Sullivan et al, CJASN, 2018). This variability becomes more apparent in larger pragmatic studies. Multisite navigator trials and system-wide interventions, such as EnAKT (Garg et al, JAMA Internal Medicine, 2023| NephJC summary) have demonstrated that even well-designed multicomponent strategies may not improve overall progression toward transplantation when implemented across heterogeneous settings.
Importantly, several studies suggest that interventions may have a greater impact among populations experiencing baseline disparities, indicating that targeting structural barriers can both improve access and advance equity. However, these gains remain inconsistent, underscoring the need for approaches that are multilevel, integrated, and sustained across the entire care pathway (Sullivan et al, CJASN, 2018; Garg et al, JAMA Internal Medicine, 2023; Bailey et al, Wellcome Open Research, 2024).
STEPS represents a methodological shift. Rather than testing education, navigation, or referral improvement in isolation, the trial integrates automated identification, augmented usual care, structured outreach, and longitudinal navigation within a single coordinated framework. The design targets multiple failure points along the transplant pathway (candidate identification, referral, evaluation completion, and donor engagement), while embedding the intervention within routine care workflows across diverse health systems.
The transplant pathway is not defined by a single decision point, but by a sequence of interdependent steps shaped by clinical, social, and structural factors. STEPS represents a critical evolution in trial design. Rather than focusing on isolated components of care, it tests whether aligning early identification, education, referral, and navigation within a coordinated system can meaningfully change how patients move through the transplant pathway.
The question is no longer whether individual interventions can work, but whether the system itself can be redesigned to deliver them effectively, equitably, and at scale—ultimately addressing the persistent burden of CKD and the ongoing disparities in access to kidney transplantation.
Join us this week on the X-space to explore this trial design with Dr Boulware and the ISN-NephJC team, on April 10, 12 pm EST!
Summary prepared by
Milagros Flores, Cristina Popa
Reviewed by
Brian Rifkin, Swapnil Hiremath