#NephJC chat
Tuesday Dec 6th 9 pm Eastern
Wednesday Dec 7th 8 pm GMT, 12 noon Pacific
The CKD Classification System in the Precision Medicine Era
Hall YN, Himmelfarb J.
Clin J Am Soc Nephrol ▪, 2016. doi: 10.2215/CJN.09310916
PMID: 27903598
Precision Medicine & CKD: A Call to Arms or a Howl at the Moon?
At the ASN in San Diego in 2015 the society’s then President, Jonathan Himmelfarb, delivered an impassioned yet nuanced address about the use and abuse of the CKD and eGFR concepts. I loved this talk, and was therefore pleased to see it converted into the CJASN commentary that we will be discussing in #NephJC this week.
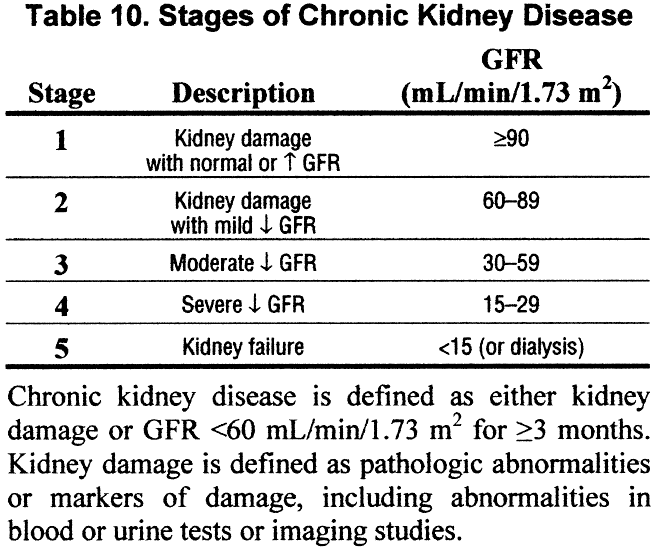
First, Himmelfarb and his co-author Yoshio Hall, outlined the progress following the introduction of the eGFR led concept of CKD in 2002: a massive increase in awareness of kidney disease, ever more rigorous epidemiological data on CKD, and a framework to predict and proactively treat the complications of advancing renal impairment.
But then the authors go on the attack.
- Why do we need so many different ways to estimate GFR?
- Why do none of these estimators take any account of patient age?
- What are CKD 1 and 2?
- Can we, as nephrologists, frequently held up as the most thoughtful medical specialty, be proud of a classification system that lumps together diverse disease processes on the basis of the single derived measurement of eGFR? Especially as this derived measurement makes no measure or mention of the activities of certain nephron segments, proteinuria, or other unmeasurables such as uraemic solutes.
With a final nod to Claude Bernard’s wonderful quote about the blindness of knowledge,
“It is what we think we know already that often prevents us from learning”
Hall & Himmelfarb exit the stage without mentioning how precision medicine might drag nephrology away from one-dimensional eGFR watching.
Well, what is precision medicine? To use an analogy first used by Isaac Kohane, precision medicine resembles the connection Amazon and Netflix have with their users: they know a lot about the population as a whole and a lot about each individual. They can use an individual’s purchase or browsing history to match it to the population with a similar history and make recommendations back to individuals based on the behaviour of the population. It is expected that the precision medicine “browsing history” will be based on big data both of a cellular (genomic, transcriptomic, proteomic measurements) and phenotypic (EHR information, information derived from portable technology) nature.
Clearly, nephrology lends itself well to this approach: frequent measurements of creatinine, proteinuria, blood pressure and disease specific parameters such as antibody titers create granular datasets for each patient. Many kidney diseases are immune mediated and therefore could leverage the whole-genome datasets already available in this area. Emerging knowledge about the contribution of genetic variants to common phenotypes such as hypertension adds another layer of potential interest.
However, this being said, it is not clear where on the famous/notorious technological hype cycle precision medicine currently sits. As John Ioannidis has recently mentioned, there is scant evidence that any of the themes of precision medicine have had any “measurable effect on population mortality, morbidity, or life expectancy”. Concerns remain as to whether EHRs are able to capture the information required to enable precision medicine, and if current consent paradigms can ensure that the necessary data sharing can be accomplished in a way that does not disadvantage patients.
I think we can expect a #NephJC chat of two parts this week. Looking firstly at what we as a community have won and lost by adopting the eGFR-led CKD paradigm, and secondly looking forward to how the future of kidney disease care might include precision medicine concepts.
Background by NephJC Work Group Member Tom Oates.





One last patient perspective on the Himmelfarb Editorial.