#NephJC Chat
Tuesday May 9th 9 pm EST
Wednesday May 10th pm BST, 12 noon Pacific
JAMA Pediatr. 2017 Apr 1;171(4):357-364. doi: 10.1001/jamapediatrics.2016.4538.
Peritoneal Dialysis vs Furosemide for Prevention of Fluid Overload in Infants After Cardiac Surgery: A Randomized Clinical Trial.
Kwiatkowski DM, Goldstein SL, Cooper DS, Nelson DP, Morales DL, Krawczeski CD
PMID: 28241247 (Paper free at JAMA Pediatrics)
Background
Up to 30-50% of children undergoing congenital heart surgery develop acute kidney injury (AKI), so pediatric nephrologists spend a lot of time in the cardiovascular ICU (CVICU). We have all of the intensivist’s numbers on speed dial, so we’re not surprised to get the consult for “POD 1 s/p complex congenital heart surgery repair with AKI, oliguria, and fluid overload” late on a Friday afternoon. (Confession: All congenital heart surgery to me is ‘complex’. I can’t visualize anything in 3D which is part of the reason I’m not a cardiologist, surgeon, or engineer.) There are a number of risk factors for AKI in the post-operative period:
Surgical factors: cardiopulmonary bypass duration, multiple cross clamps, intraoperative time
Cardiac factors: RACHS-1 category, functional single ventricle
Infant factors: young age, prematurity
Preoperative factors: prior AKI, mechanical ventilation, inotropic support
AKI is so common in this group of children that it is a test population for validating biomarkers of AKI that allow for early detection. AKI in these infants isn’t just bad because it increases the cost of hospitalization. AKI is associated with increased morbidity and mortality and prolonged mechanical ventilation, ICU stay, and hospital stay. As we’re becoming more and more aware, AKI is also a risk factor for chronic kidney disease (CKD) and these children are no exception. A recent Danish registry study showed the five-year cumulative incidence of CKD for children with AKI after cardiac surgery was 12%, significantly higher than the 3% for those without perioperative AKI.
But what should we do about it? A number of case series and retrospective studies have shown improved outcomes with early initiation of peritoneal dialysis for fluid management of AKI post congenital heart surgery (link, link). Because placing a peritoneal dialysis (PD) catheter in a fluid overloaded, critically ill infant after cardiac surgery is technically difficult, some centers have begun placing PD catheters in certain high risk infants preemptively: The recent JAMA Pediatrics article from Kwiatkowski et al. attempts to answer the question of whether early initiation of peritoneal dialysis in infants with AKI and oliguria after congenital cardiac surgery leads to improved outcomes compared to diuretic therapy alone.
Methods
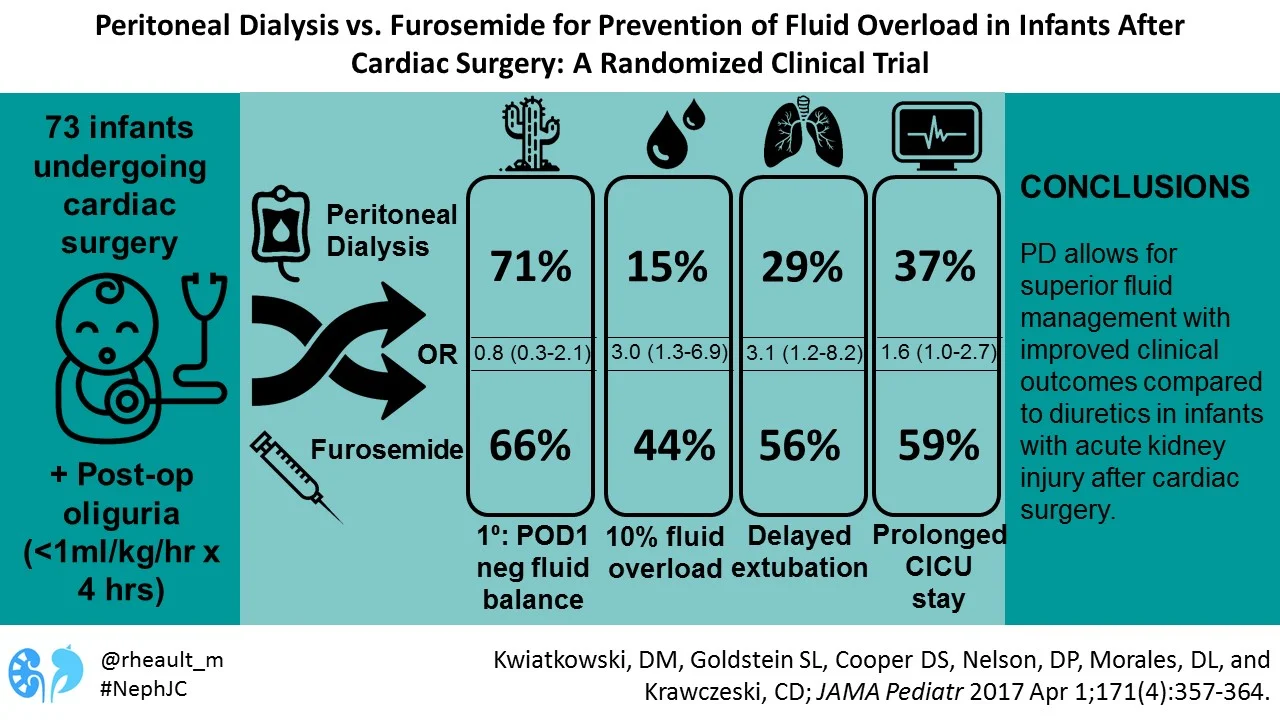
This was a prospective, single-center, randomized clinical trial.
Inclusion criteria included infants age <6 months undergoing congenital heart surgery with cardiopulmonary bypass with planned PD catheter placement per routine practice (eTable 1). Infants were excluded if they had pre-existing kidney disease (eGFR <60) or if they had severe complications within the first 24 hours including death, extracorporeal membrane oxygenation, or requirement for a second operation. Post-operative oliguria was defined as any 4 hours of urine output of less than 1 mL/kg per hour during the first 24 hours out of the OR. Infants who developed oliguria were randomized to:
Peritoneal dialysis: initially 10 ml/kg fill volume of 1.5% Dianeal, 1 hour continuous cycles
Furosemide: 1 mg/kg intravenously Q6 hours x 2, then per physician.
The primary outcome of interest was negative fluid balance on Post-Op Day one.
Results
73 infants completed the study. Although primary outcome and mortality were not different between the two groups (we can debate whether the primary outcome was the most clinically meaningful), there was significantly lower risk of 10% fluid overload in the PD treated group. Although median duration of mechanical ventilation and duration of CVICU stay were not different between groups, post-hoc analysis showed significantly more infants in the furosemide group with prolonged ventilation (> 3 days) or prolonged CVICU stay (>7 days). Not surprisingly, infants who received furosemide required more electrolyte replacement and had higher “electrolyte abnormality scores”. (I’m still sorting through the fine print to determine what an electrolyte abnormality score is.)
Nine infants had pleural-peritoneal communications precluding PD and there were two patients with PD related adverse events (peritoneal bleeding requiring a blood transfusion and hydrocele), lest you forget that sticking a plastic tube into the peritoneum and filling it with fluid may have complications.
Discussion
The authors should be commended on completing this study. There is a lot of religion that goes into fluid management in the post-operative period, so having randomized data in this common population is important to guide future therapy. Will it change your management? Will you recommend PD catheter placement in all at-risk infants? If your center already places a PD catheter, will you be quicker to use it? Are there risks that haven’t been discussed yet? What about the risk of the second anesthesia for removing the PD catheter? We look forward to a lively discussion for the next #NephJC.