#NephJC 10 Post Discussion
Tuesday, April 7th 2026, 9pm Eastern
NEJM Evid. 2026 Mar;5(3):EVIDoa2500086. doi: 10.1056/EVIDoa2500086. Epub 2026 Feb 24.
A Randomized Trial of Targeted Hyponatremia Correction in Hospitalized Patients
Julie Refardt, Laura Potasso, Anissa Pelouto, Moritz Trappe, Claudia Gregorian, Markus Koster, Ivana Dora Vodanovic, Dario Norello, Svenja Ravioli, Sadrija Cukoski, Maria Boesing, Basil Ryser, Lana Sambula, Nikola Rapsch, Sophie Monnerat, Julia Beck, Sven Lustenberger, Deborah R Vogt, Laura Werlen, Joyce Santos de Jesus, Martina Bontognali, Philipp Schuetz, Adrienne A M Zandbergen, Alessandro Peri, Darko Kastelan, Gregor Lindner, Joerg Leuppi, Stefan Bilz, Beat Mueller, Volker Burst, Ewout J Hoorn, Mirjam Christ-Crain
PMID: 41733398
DOI: 10.1056/EVIDoa2500086
Introduction
Don’t get salty, but you don’t know as much about sodium as you think you do.
In 2017, you knew that increased sodium intake leads to increased urine sodium output. Then, Dr. Roger Rodby opined about why everything we know about sodium might be wrong (Some Thoughts on Spooky Sodium| NephJC). Ninety-eight percent of total-body sodium is confined in the extracellular fluid (ECF) compartment in young, healthy humans. However, about 80% of exchangeable sodium is found in the interstitial and connective tissues, and only about 15% of exchangeable sodium is in plasma. When dietary salt intake increases, urinary sodium excretion increases, but it does not match intake immediately and thus generates a positive sodium balance until excretion again equals intake. Increasing NaCl intake also increases body water in most situations, although this does not result predominantly from more fluid consumption but rather from fluid retention. When dietary salt intake rises from low to moderate levels, body water increases. When salt intake rises further, sodium accumulation may occur without an increase in water (Rokova et al, J Clin Invest, 2017).
In 2021, you thought you knew that increased sodium intake led to increased thirst and elevated blood pressure. Doctors Ellison and Welling looked at controversies in salt handling and blood pressure. Increased salt doesn’t always lead to increased thirst but rather increased hunger and higher calorie burn. Over-restricting salt may lead to insulin resistance and higher blood pressure. The mechanistic basis for the link between ECF volume and vascular tone remains contentious. Drinking saline solutions to increase salt intake increases protein catabolism, glucocorticoid production, and urea generation, whereas salt loading through diet, with access to free water, does not. (Ellison et al, NEJM, 2021)
In 2022, you knew that lowering salt intake was better for heart failure and hypertensive patients. However, implemented dietary counseling (reducing sodium below 2 grams daily) did not show added benefit in the largest trial performed in this population (Ezekowitz et al, Lancet, 2022| SODIUM-HF NephJC). Don’t even get me started on fluid restriction in heart failure- hint: it isn’t helpful, stop torturing your patients (Hermann et al, Heart, 2026).
In 2023, sodium correction limits were emphasized to reduce the risk of osmotic demyelination syndrome. However, Seethapathy et al. showed that slower sodium correction rates were associated with higher mortality and longer hospital stays. Because the study evaluated observed correction rates rather than treatment intent, the association should not be interpreted as causal and may reflect greater illness severity in patients whose sodium corrected more slowly (Seethapathy et al, NEJM Evid, 2023| Standing Corrected? NephJC).
Nowadays, we know that hyponatremia is associated with osteoporosis, falls, fractures, gait disturbance, and subtle cognitive impairment (Verbalis et al, J Bone Miner Res, 2010| Brinkkoetter et al, Sci Rep, 2019). So, my homeostasis amigos, shouldn’t we fix it? Among patients with mild to moderate asymptomatic hyponatremia, it remains unclear if normalization of sodium levels is clinically beneficial.
Confused? Yeah, even we nephrologists still have much to learn about the ion that sets the tone for extracellular water. Homeostasis ain’t no joke!
The Study
Methods
This is a pragmatic, randomized, controlled, parallel-group, international, multi-center superiority trial with blinded outcome assessment.
The trial was conducted at 9 centers in Europe from August 2018 through April 2024.
Both university and community-based hospitals were included.
Inclusion and exclusion criteria are as follows. Patients with severe symptomatic hyponatremia requiring urgent correction, hypertonic hyponatremia, or end-of-life care were excluded.
Screening, Inclusion, and Randomization
Plasma sodium levels were reviewed by local trial physicians. Patients were invited to be included once hypotonic hyponatremia was confirmed (Na < 130 mmol/L, plasma osmolality ≤ 300 mOsm/kg). Hyponatremia could have occurred prior to, or during hospitalization. Patients could be enrolled anytime during their hospitalization. Initial assessment included reason for hospitalization, comorbidities, and additional lab values. A cognitive assessment was done with the Trail Making Test (essentially a connect-the-dots in numerical order).
The trial was open-label with respect to treatment assignment, but outcome adjudication and follow-up assessments were performed by investigators blinded to treatment allocation. Randomization was centralized and performed in a 1:1 ratio without stratification.
Targeted Correction of Hyponatremia
Patients were then randomly assigned 1:1 to either targeted plasma sodium correction (intervention) or standard treatment (control). Treating physicians in the control group were not restricted from correcting hyponatremia. Therefore, sodium correction also occurred in the standard-of-care arm, consistent with the pragmatic design.
The targeted treatment of hyponatremia was based upon the following protocol derived from specialty guidelines.
Figure S1. Diagnostic and treatment algorithm. Refardt et al, NEJM Evid, 2026.
The predominant cause of hyponatremia was determined by history, clinical presentation, assessment of fluid status, and lab testing. Hyponatremia determined to be due to “mixed etiologies” was classified by the predominant cause. Each classification of hyponatremia had its own stepwise treatment approach, determined by severity and response to therapy.
Treatment response and adherence were monitored daily. Treatment was intensified if sodium correction was < 2 mmol/L/d, was maintained if the correction was 2-12 mmol/L/d, and stopped if the correction was > 12 mmol/L/d (or > 18 mmol/L in 48 hours, considered an overcorrection). Overcorrection could be treated with hypotonic fluids with or without desmopressin. Apart from assuring adherence to the hyponatremia protocol, the trial team did not influence treatment or discharge decisions. After discharge, the treatment of hyponatremia was left to the treating physician.
Targeted plasma sodium was treated to a level of 135-145 mmol/L and was maintained for the duration of the hospitalization until discharge. Targeted correction was also stopped if the patient was hospitalized for > 30 days after inclusion, and they failed to attain normalization of sodium.
In the control group, patients received usual care, meaning hyponatremia was managed as in routine practice. Although described as “based on the European guidelines” (Spasovski et al, Eur J Endocrinol, 2014), no structured algorithm or treatment targets were mandated. Clinicians followed general principles, but therapy could range from active treatment to observation, and escalation to achieve normonatremia was not required. This contrasts with the intervention group, where care followed a predefined, stepwise protocol aimed at sodium normalization. Treatment decisions and sodium values were recorded retrospectively from the medical record after discharge.
Outcomes
Primary Outcome
-A combined risk of death or rehospitalization within 30 days of trial inclusion. The authors used no hierarchical weighting (death and rehospitalization were treated as equivalent events).
Secondary Outcomes
Mortality and rehospitalization within 30 days and 1 year of trial inclusion
Absolute change and rate of change of sodium levels
Maximum change in sodium levels from inclusion to discharge
Rate and time to normalization of sodium
Rate of recurrence of hyponatremia within 30 days
Complications due to overcorrection
Length of hospital stay
Falls and fractures within 30 days
Quality of life questionnaire
Trail Making test at admission and discharge
Corrected 30-day rehospitalization/mortality rates based upon etiology, age, sex, and treatment
Events during the trial period were recorded by the trial team. Additional assessments were done following the hospitalization by a team blinded to treatment assignment.
Statistical Analysis
The sample size was calculated assuming an event rate of 23% in the standard care and 18% in the treatment group. To achieve 80% power with an alpha error rate of 5%, 2050 patients were needed for evaluation.
The primary analysis followed a modified intention-to-treat approach, including all randomized patients who remained in the study for at least 24 hours. Predefined subgroups included severe hyponatremia (Na < 120), etiology, age (< or > 70 years), and sex. Secondary outcomes were considered exploratory without plans for multiple comparisons or adjustment of confidence intervals.
Missing outcome data were handled using available-case analysis without imputations.
Funding
The study was supported by a grant from the Swiss National Science Foundation. The funders had no role in the design and conduct of the trial; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and the decision to submit the manuscript for publication.
Results
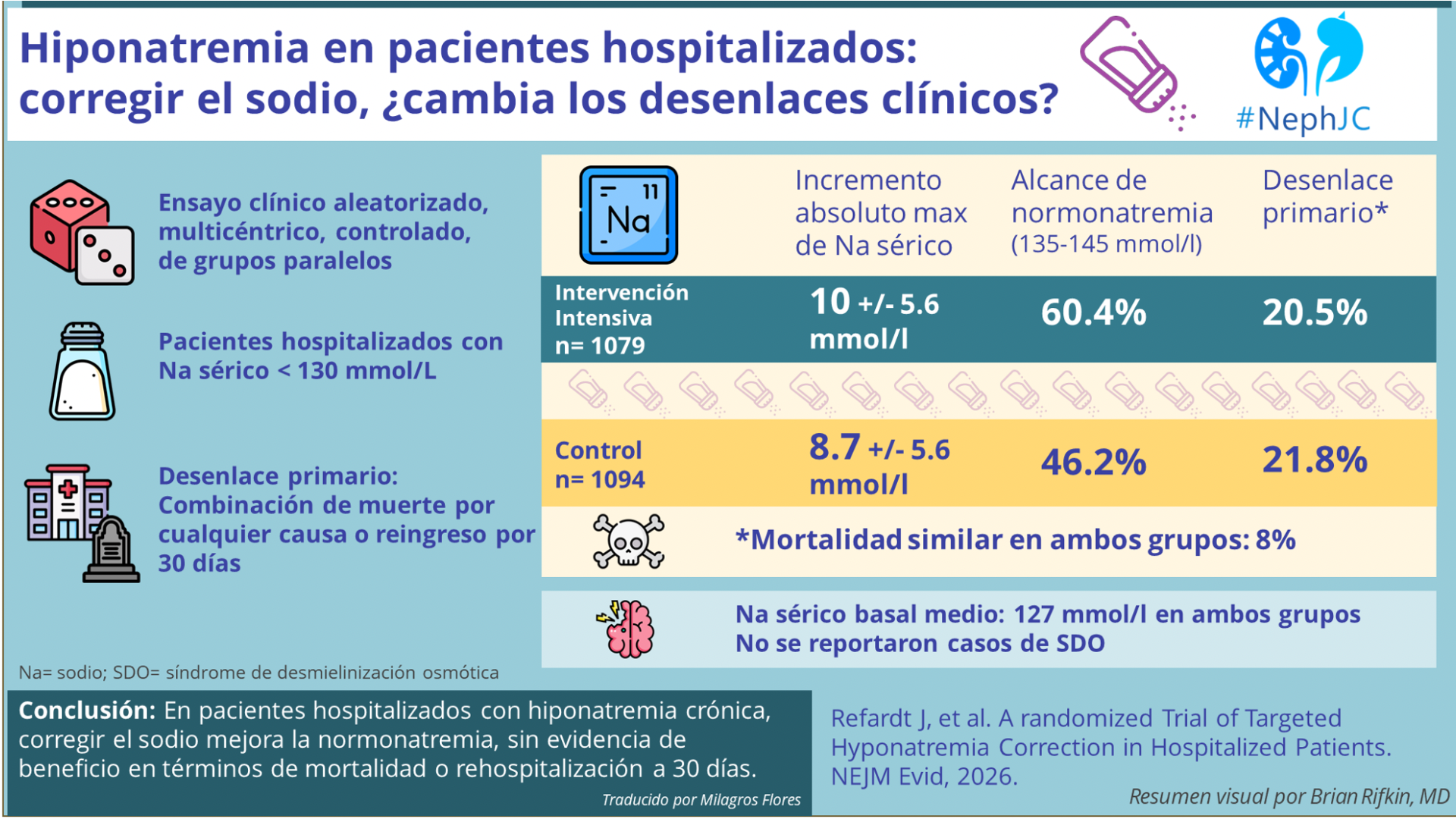
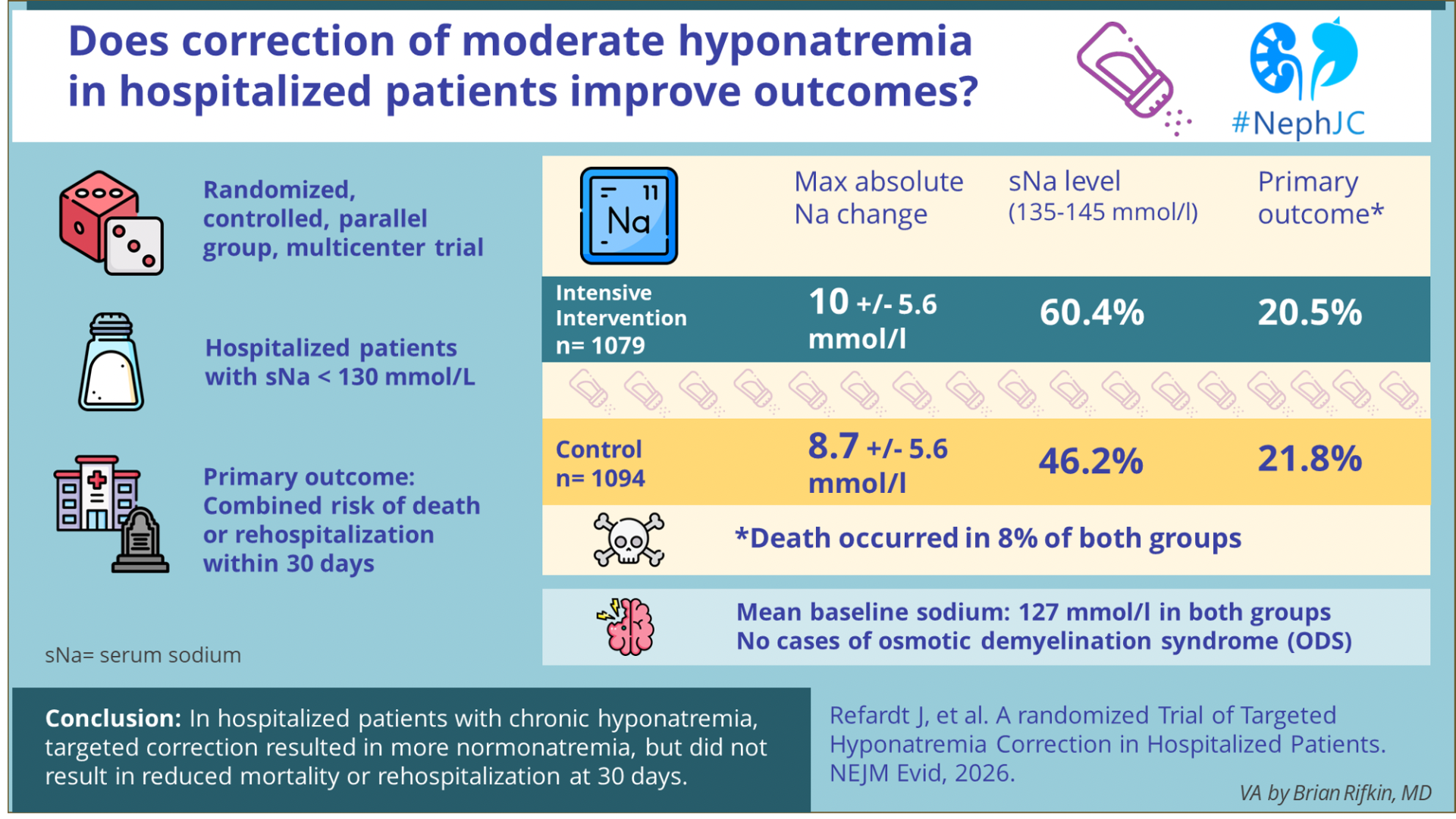
The per-protocol analysis included 2049 patients. In the full analysis set, 1079 were assigned to the intervention and 1094 to the control group.
Figure 1. Patient inclusion flow diagram. Refardt et al, NEJM Evid, 2026.
The median age was 73, and 48% of the included patients were male. At inclusion the median plasma sodium level was 127 mmol/L in both groups. Roughly 6% of the entire cohort had severe hyponatremia (< 120 mmol/L).
Table 1. Patient characteristics. Refardt et al, NEJM Evid, 2026.
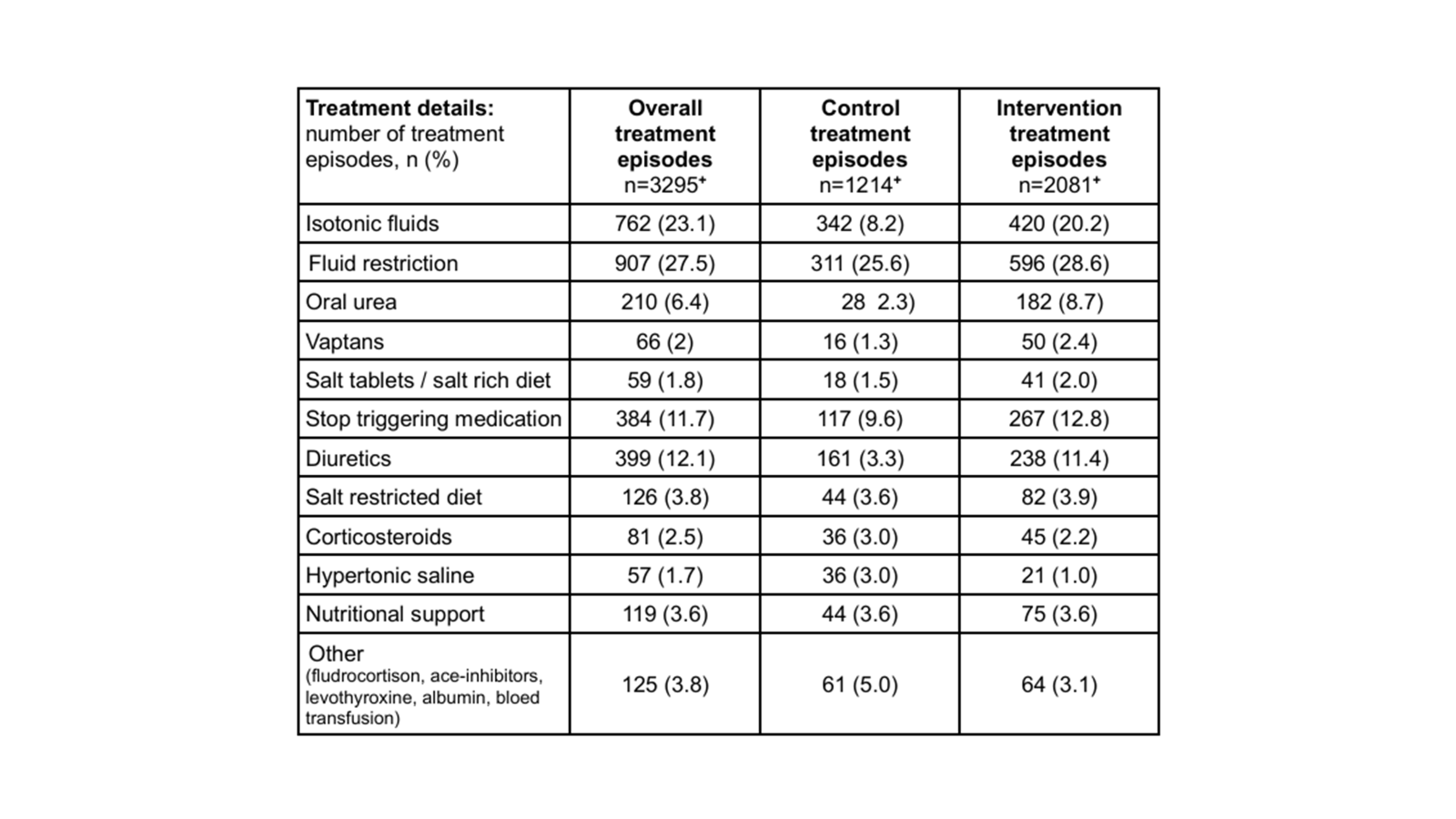
Hyponatremia treatment strategies
Table S3. Hyponatremia treatments during the observation period. Refardt et al, NEJM Evid, 2026.
Etiology-directed treatment was used more frequently in the intervention group, including fluid restriction for SIADH, isotonic saline for hypovolemia, and diuretic adjustments for hypovolemia. Hypertonic saline and desmopressin were used infrequently in both groups. Patients in the intervention group experienced a maximum absolute change of 10 +/- 5.6 mmol/L, while the control group had an 8.7 +/- 5.6 mmol/L change.
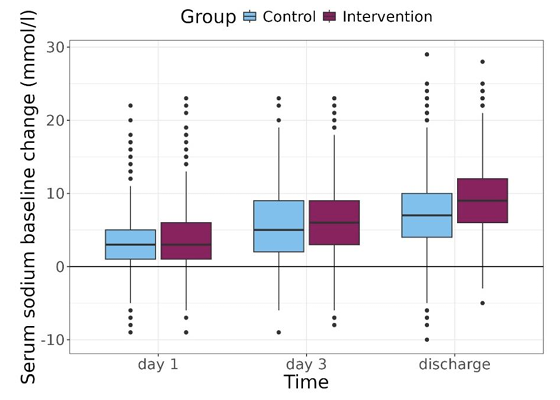
Figure S2. Course of plasma sodium levels. Refardt et al, NEJM Evid, 2026.
A normal sodium level was obtained by 60% of the intervention patients versus 46% of the control group. At discharge, 56% of intervention and 37% of control patients had normal sodium levels. Hyponatremia recurrence or persistence at 30-days was approximately 41% in both groups. The mean rate of sodium correction during hospitalization was modestly higher in the intervention group, but the overall sodium trajectories overlapped between groups. The median time to sodium normalization was shorter in the intervention arm.
Figure 2. Time- to-event analyses. Refardt et al, NEJM Evid, 2026.
Primary Outcome
Death or rehospitalization within 30 days of study inclusion occurred in 218 intervention patients versus 234 control patients (estimated absolute difference -1.3%; 95 CI: -4.9 to 2.2, p = 0.45). The observed event rate and sample size were consistent with the prespecified power assumptions. The finding was consistent when correcting for potential site effects, as well as best and worst-case scenario analyses, in which missing 30-day outcomes were assigned extreme assumptions favoring either the intervention or control group.
Table 3. Mortality and rehospitalization rates. Refardt et al, NEJM Evid, 2026.
Secondary outcomes
Death occurred in 8% of both groups. In the same time frame, 13% of intervention patients and 14% of control patients were rehospitalized within 30 days. The intervention was not associated with time to the primary outcome, time to death, or time to rehospitalization. The median length of stay (7 days) was similar in both groups. There were no differences in cognitive assessment, quality of life, or rates of falls or fractures at 30-day follow-up. One-year mortality and rehospitalization rates likewise did not differ, indicating no sustained benefit of target sodium correction.
Subgroup analyses
Prespecified subgroups of etiology, severity, and sex did not suggest an association with the primary outcome. Tests for interaction were not statistically significant across prespecified subgroups, including baseline sodium severity, etiology, age, and sex. It is noteworthy that in patients > 70 years old, the intervention was associated with a lower risk of the primary outcome (OR 0.76, 95% CI, 0.57-1.00). This apparent age interaction was borderline and not adjusted for multiple comparisons. There was no evidence of correlation for reaching normal sodium levels and the primary outcome. However, reaching a normal sodium level (in either group) was associated with decreased odds of death or rehospitalization within 30 days (OR 0.74, 95% CI, 0.60-0.91). The association between normonatremia and improved outcomes appeared observational and was not modified by treatment assignment.
Figure 3. Subgroup analysis. Refardt et al, NEJM Evid, 2026.
Safety analysis
Overcorrection occurred in 2.3% of intervention and 1.4% of control patients. Most overcorrection events occurred within the first 48 hours of treatment and were managed with desmopressin and hypotonic fluids. Adverse events (e.g., prolonged hospitalization) occurred in 1.2% of intervention and 0.8% of control patients. No cases of osmotic demyelination syndrome were observed. Symptomatic worsening of hyponatremia occurred in 0.5% of intervention and 0.1% of control patients. All patients responded to therapy intensification. Rates of ICU transfer, neurologic complications, and treatment-related adverse events were low and similar between groups.
Weaknesses
unblinded (design, with potential bias from increased awareness and monitoring of hyponatremia in the intervention arm)
predominantly mild to moderate hyponatremia, limiting applicability to severe disease
Intervention limited largely to the hospitalization period, with no standardized post-discharge management
Modest separation between groups, with only small differences in achieved sodium level and substantial overlap in treatments
Strengths
prospective, pragmatic, randomized design
large sample size
diverse (academic/community) hospital settings
blinded outcome adjudication despite open-label treatment
prespecified power calculation met with the expected event rate
algorithm-based intervention grounded in physiology
hard clinical primary endpoint (death or rehospitalization) rather than biochemical outcome
Discussion
“To treat or not to treat?” is a profound question in asymptomatic hospitalized patients.
Therapeutic apathy and inertia can drive inactivity. However, there is precedent that intensifying treatments in the controlled hospital setting can lead to worse clinical outcomes and complications (e.g., hypoglycemia and hypotension) and often includes medication confusion following discharge. In the case of mild to moderate hyponatremia, the question arises- is euboxia and normonatremia the ultimate goal? It is interesting to note that in this well-run, large trial of hyponatremia, achieving a normal serum sodium (in either group) was associated with improved outcomes of death and rehospitalization at 30 days. However, this association does not establish causation and likely reflects improvement in the underlying disease rather than sodium correction itself. Hyponatremia represents disturbed water homeostasis and often serves as a marker of illness severity. This interpretation is consistent with pharmacological trials, such as vasopressin antagonists, which failed to show improved clinical outcomes despite normonatremia (Schrier et al, NEJM, 2006). Association versus causation appears to be in full effect for the hyponatremia correction conundrum.
This study found no significant difference in its primary outcome. However, the difference between treatment strategies was small (only 1.3 mmol/L higher in the intervention group), and therapies were not necessarily continued after discharge from the hospital setting. Despite greater normalization of sodium in the intensive group, there was a significant portion of patients who had recurrence of hyponatremia at 30 days (approximately 42% in both groups). Also, the use of therapies further down the algorithm was limited: only 8% were prescribed urea, and 3% vaptans. One could argue that urea is cheap, safe, and effective and could be used more (beyond what the guidelines call for). On the other hand, the vaptans are costly and do have significant side-effects (including liver toxicity and increased risk of GI bleeding) that might have limited their use, even in severe cases. One is left wondering if perhaps the intensive arm wasn’t intensive enough to make a significant difference. Especially since 40% of patients were discharged with uncorrected sodium levels. If we want to test if fixing the sodium makes a difference, we should use an algorithm that does a better job of fixing the sodium.
The elderly (> 70 years old) appear to benefit the most from intensive interventions. This may be because this group is at higher risk for complications (cognitive, falls, fractures). Other than age, no other factors, including etiology and severity of hyponatremia, seem to have a significant association with the primary outcome. Because hyponatremia is common and ubiquitous, further large studies of subgroups might identify patients with a greater opportunity to benefit from intensification of sodium correction.
Hyponatremia is not a single disease but a manifestation of disturbed water homeostasis arising from multiple underlying conditions, including SIAD, diuretic use, hypovolemia, heart failure, cirrhosis, and endocrine disorders. Because these etiologies differ in pathophysiology, prognosis, and response to therapy, clinical studies of hyponatremia inherently pool biologically distinct populations. In this context, Hoorn and Zietse note that “the patient with hyponatremia does not exist”; because outcomes are largely characterized by the underlying disease complicated by hyponatremia rather than the sodium concentration itself (Hoorn, Zietse, JASN, 2017). Despite this heterogeneity, the present trial did not demonstrate differences in outcomes according to etiology. This might reflect both limited power for subgroup analysis and imprecision in diagnostic classification. Differentiating hypovolemic from euvolemic hyponatremia based on clinical assessment is unreliable, with reported sensitivities of 50-80% and specificities. 30-50%, and mixed etiologies are common. Patients frequently have multiple contributing mechanisms, further blurring etiologic distinctions. These diagnostic limitations dilute treatment effects in pragmatic trials and reduce the impact algorithm-based correction strategies, particularly when treatment is guided by imperfect volume classification.
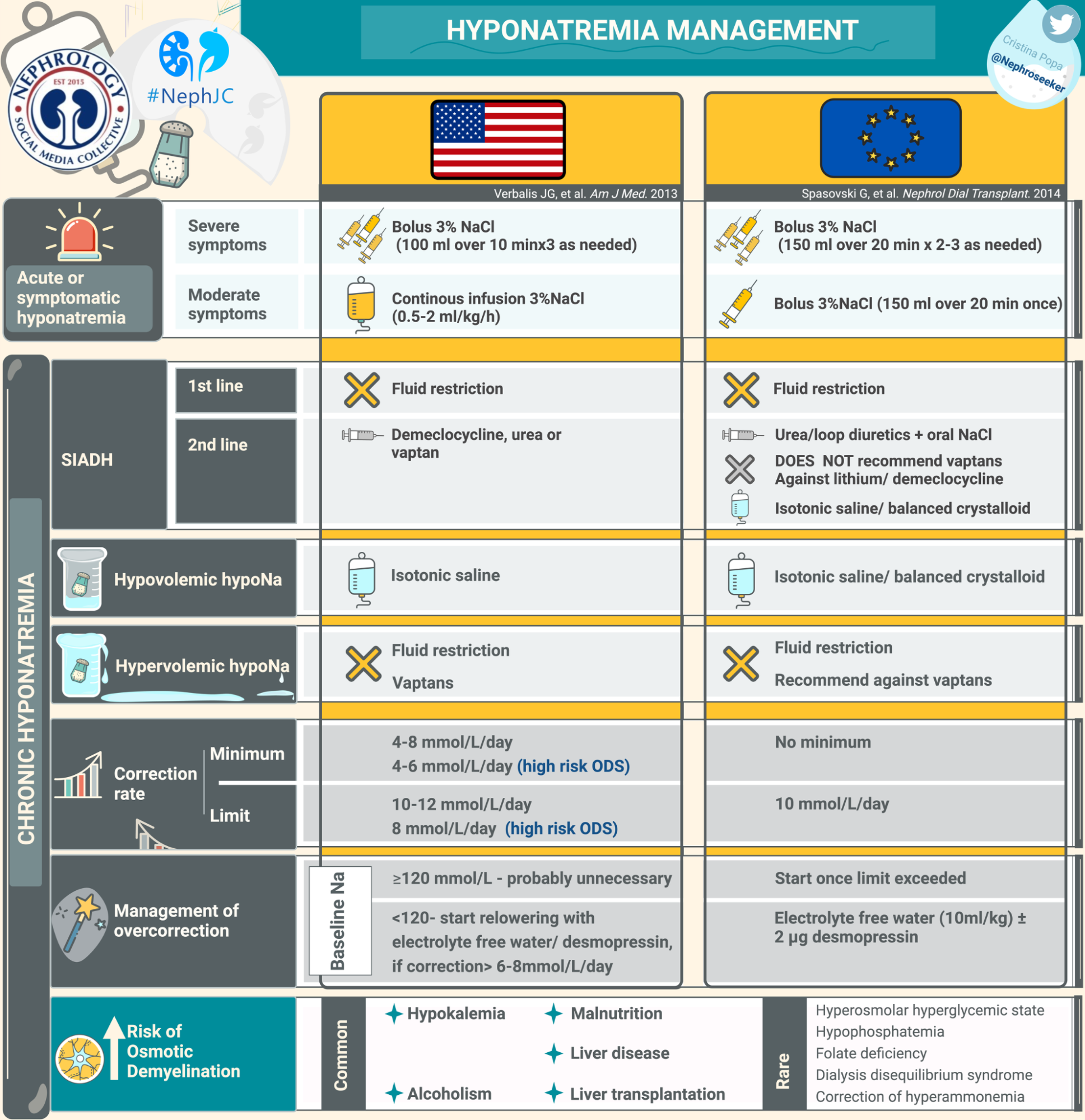
The HIT trial fits within this framework and provides randomized evidence addressing a central assumption underlying current guidelines. Both US (Verbalis JG, et al, Am J Med 2013) and European (Spasovski et al, Eur J Endocrinol, 2014) recommendations emphasize etiology-based therapy and cautious correction but differ in how strongly they prioritize biochemical normalization. The present trial shows that guideline-based intensification improves sodium normalization without improving short-term clinical outcomes. These findings suggest that, particularly in heterogenous populations with mild hyponatremia, normalization of sodium alone is unlikely to modify outcomes driven primarily by the underlying disease
Appropriately, the authors conclude that the trial does not suggest “do nothing” for patients with mild to moderate hyponatremia, but rather that intensive treatment does not confer clear or significant benefits over standard therapy. Increasing the number of therapeutics and intensifying treatment of moderate hyponatremia does not appear to be beneficial, at least in the short term. Standard treatment, which included doing nothing in 35% of patients, did not have any significant downside or safety issues.
Conclusion
In hospitalized patients with mild to moderate chronic hyponatremia, a more intensive targeted correction strategy modestly improved sodium levels but did not reduce 30-day mortality or rehospitalization, suggesting that in this population, where hyponatremia was generally mild, biochemical correction alone may be insufficient to influence short-term clinical outcomes.
Summary by
Brian Rifkin MD, FASN, FASDIN
Hattiesburg Clinic