#NephJC Chat
Tuesday, May 5th 2026, 9 pm Eastern on X and Bluesky
Lancet. 2026 Feb 7;407(10528):587-598., doi: 10.1016/S0140-6736(25)02255-X. Epub 2026 Jan 27.
TRPC6 inhibition for the treatment of focal segmental glomerulosclerosis: a randomised, placebo-controlled, phase 2 trial of BI 764198
Howard Trachtman, Matthias Kretzler, Loreto Gesualdo, Nicholas Cross, Biruh Workeneh, Jessica Kaufeld, Björn Meijers, Zhiming Ye, Qinkai Chen, Vimal K Derebail, Monica Suet Ying Ng, Bo Ji, Maximilian T Lobmeyer, Silke Retlich, Fabia T Licarião Rocha, Srinivasa Prasad, Nima Soleymanlou
PMID: 41616795
DOI: 10.1016/S0140-6736(25)02255-X
Introduction
Focal segmental glomerulosclerosis (FSGS) is a common histological pattern of glomerular scarring that can be due to diverse underlying pathophysiology, including unknown immune-related ‘podocyte-toxic’ factors (primary) or various secondary causes (see Fig 52). As genetic screening becomes more widespread, it has been suggested that known genetic causes of the hereditary FSGS account for upwards of 20% of cases (Satin S, et al. Clin J Am Soc Nephrol, 2011). The clinical presentation of FSGS is typically the nephrotic syndrome, with progressive renal failure, although the exact prognosis and presentation may vary by underlying etiology. It is generally recommended to treat the underlying cause of FSGS in secondary disease and consider use of immunosuppression, such as steroids and calcineurin inhibitors, in primary disease. To date, only the dual-acting endothelin type A and angiotensin II receptor blocker, sparsentan, has been FDA-approved for the treatment of FSGS in 2026.
Figure 51. KDIGO 2021 Clinical Practice Guidelines for the management of glomerular disease, KI 2021
Figure 52. KDIGO 2021 Clinical Practice Guidelines for the Management of Glomerular Diseases, KI 2021
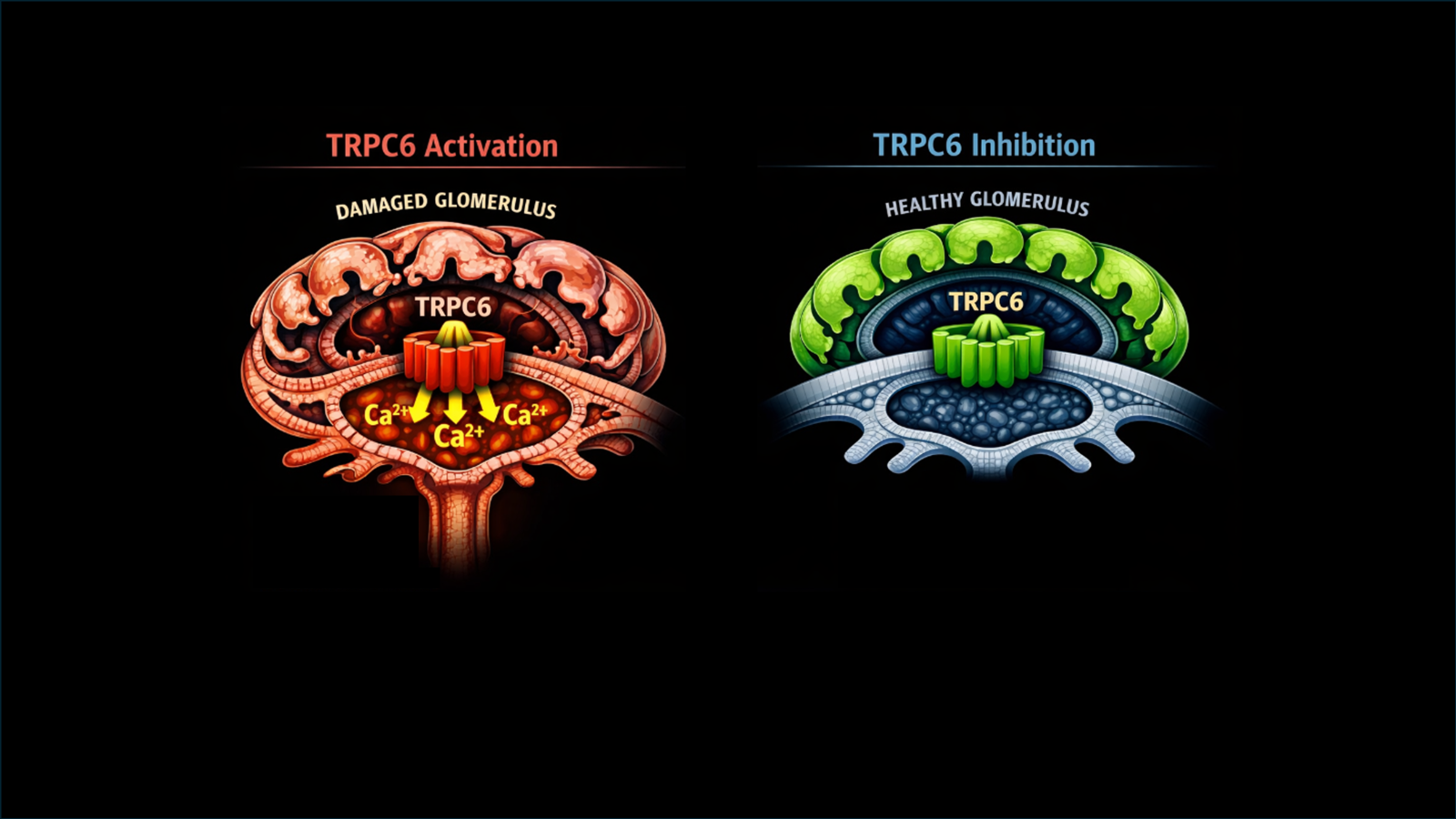
The transient receptor potential cation channel 6 (TRPC6) story is more recent and dates back to 2005, when a genetic variant was discovered in a large kindred with FSGS (Winn MP et al, Science, 2005). The variant was present on chromosome 11 and altered calcium signaling in podocytes and increased the activity of “calcium transients” (rapid, temporary increases in intracellular ionized calcium that are crucial signaling mechanisms). Higher calcium transients were suggested to cause disruption of the cytoskeleton, detachment, apoptosis, or decreased proliferation of podocytes. Patients with TRPC6 mutations typically present in the 3rd to 4th decade with severe proteinuria, and 50% of them progress to ESRD within two decades (Caridi et al, Pediatr Nephrol 2010). In TRPC6 variants, truncated mutations have no clinical significance compared with missense and gain-of-function mutations (Wooden B et al, CJASN; McAnnallen SM et al, NDT, 2025).
Figure 1. From Starushenko et al, KI 2019
Similar to medications being developed for APOL1-mediated kidney disease (AMKD), TRPC6 inhibitors, as part of precision therapy, were developed specifically for the inhibition of TRPC6 gain-of- function mutations (Zimmerman B et al, Nature, 2025).
In a mouse model of FSGS, the drug BI 749327 was observed to decrease kidney fibrosis and related fibrotic gene expression. Additionally, infiltration by inflammatory CD3⁺ T cells was reduced, suggesting that BI749327 may have utility in non-TRPC6 variant causes of FSGS-related decline in kidney function as well (Lin BL et al, Physiology PNAS, 2019). This current phase 2 study was designed to look at the efficacy and safety of this novel FSGS therapy.
The Study
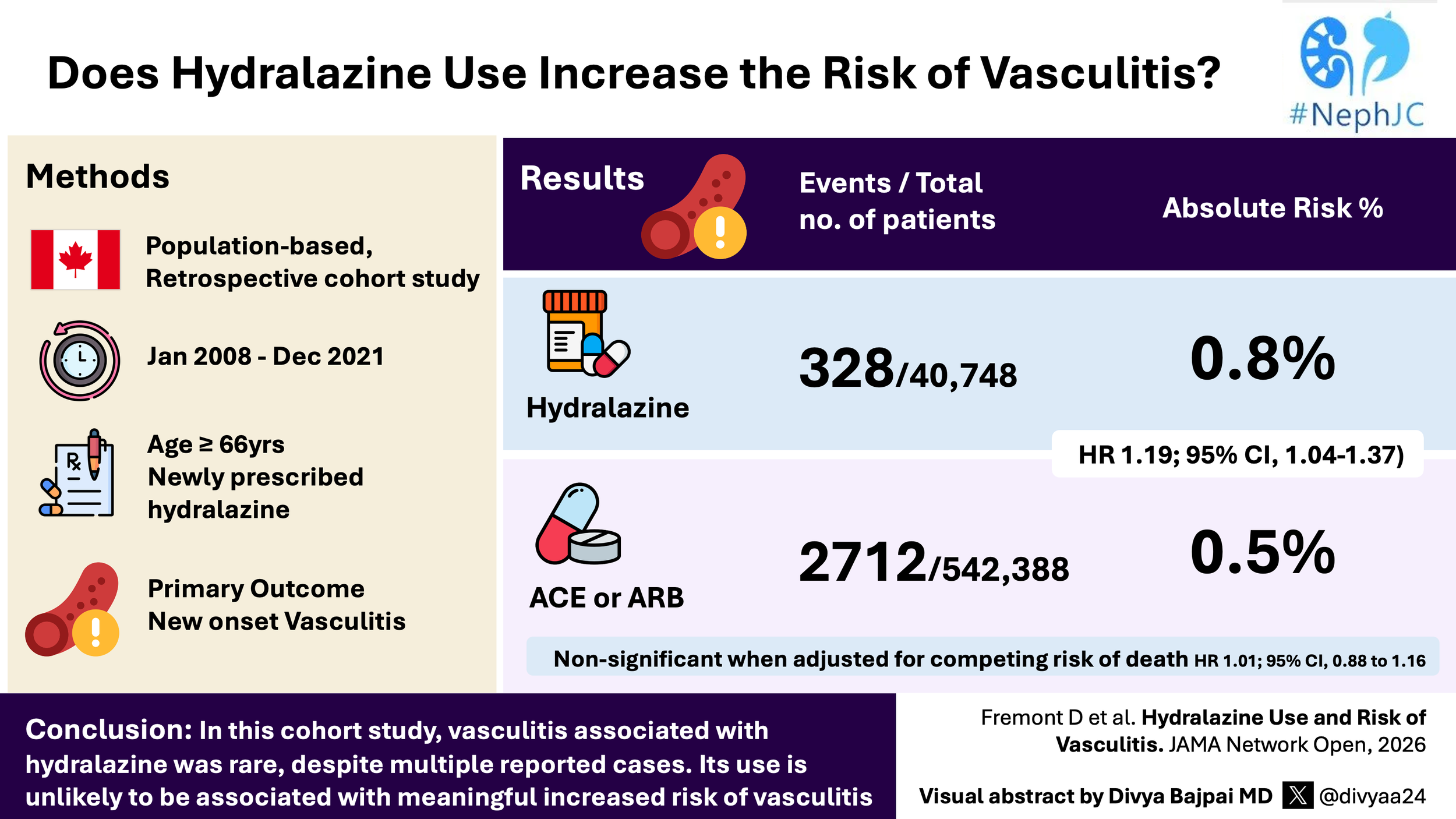
Methods
Study design
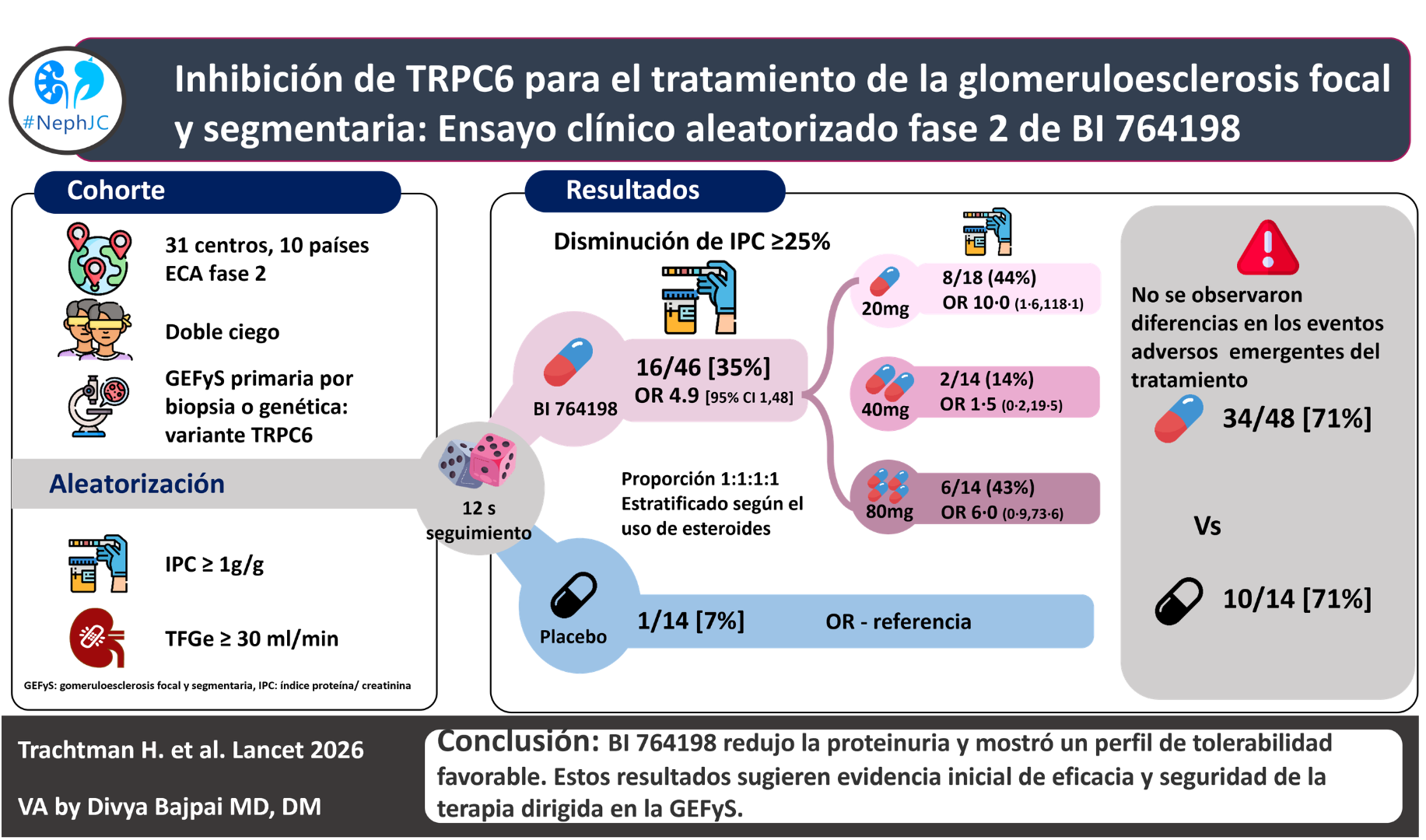
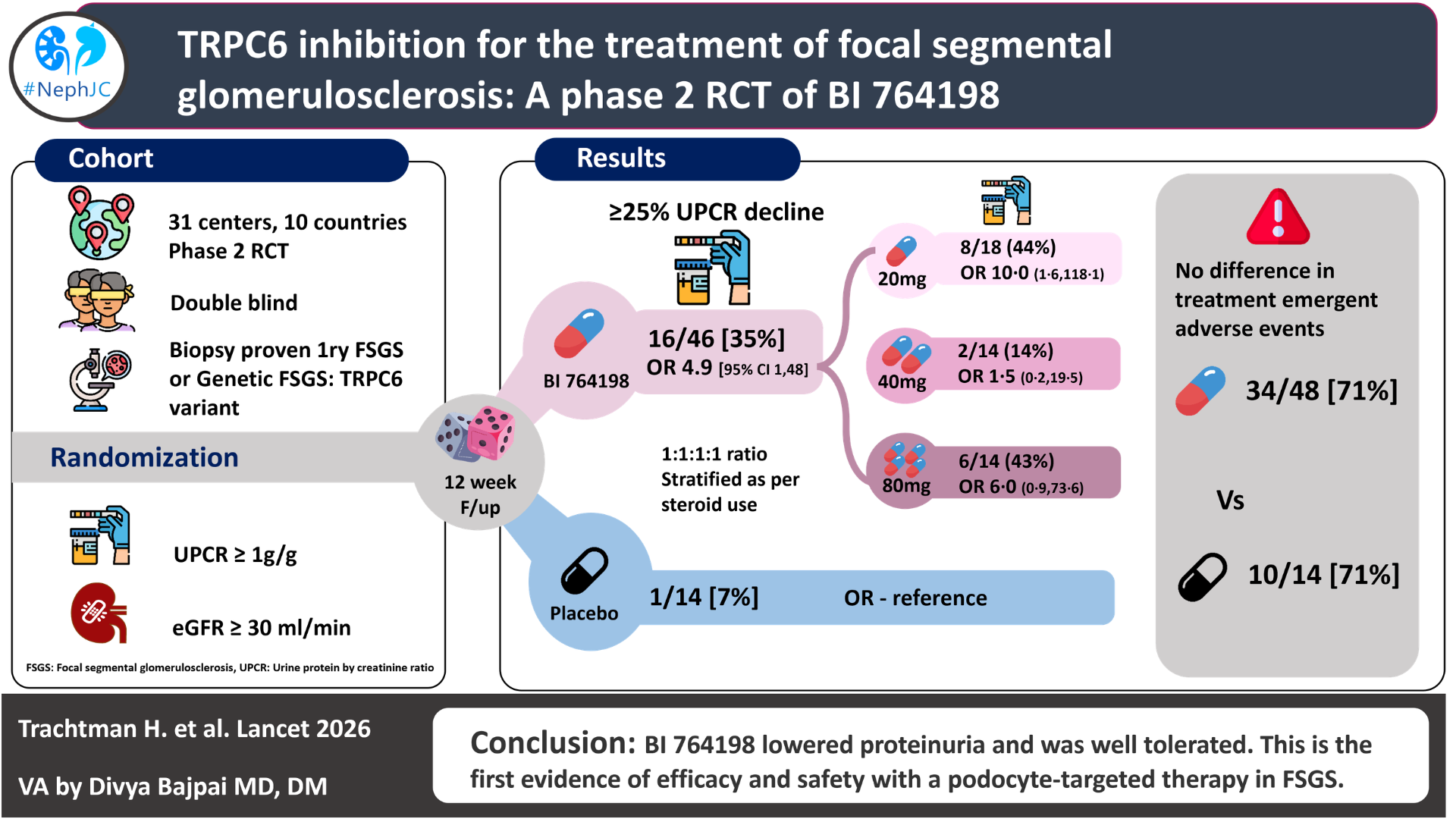
The study was a multicenter (31 centers), multinational (10 countries), parallel-group randomized controlled study. The data was collected from an online platform, BRAVE, through mobile research nurses under the supervision of an onsite investigator.
This placebo-controlled trial studied patients with primary FSGS or genetic FSGS resulting from a TRPC6 variant. The study started on Jan 27, 2022 and was completed on Jan 3, 2025.
Inclusion and Exclusion Criteria
Concomitant CNI use was not permitted; participants on ACEI/ARB/finerenone/SGLT2I treatment were required to be on a stable dose for > 4 weeks prior to screening. In the initial phase, researchers included only those participants who had completed a steroid course, but with unresolved proteinuria >1.5 g/g. This was amended in Oct 2022 to include all participants with a stable dose of steroids >4 weeks prior to screening.
Randomization and masking
It was a double-blinded RCT. Participants were randomly allocated in a 1:1:1:1 ratio to receive one of three oral doses of BI 764198 (20 mg, 40 mg, or 80 mg) given once daily or a matched placebo with automated block randomization and further stratified according to the usage of steroids with interactive response technology. The drugs were shipped directly to the patient once allocation was verified; treatment was given for 12 weeks with follow-up on days 7 and 30 after treatment.
Procedure
In the initial 2 visits, 24-hr urine protein was collected and averaged for a baseline. In later visits, urine PCR was quantified on day 4 (Visit 2) and weeks 4, 8, 12, and 13, and serum creatinine (eGFR) was measured on day 4 (Visit 2) and weeks 2, 4, 6, 8, 10, 12, and 13. Throughout the dosing interval, all doses maintained sufficient drug concentrations, even at trough levels, and target exposure was also adequate.
Outcomes
Primary endpoint: Proportion of participants with a ≥25% reduction in 24-hour UPCR from baseline to week 12, with sensitivity analysis done with the per-protocol analysis set (PPS).
Secondary endpoints:
Change in 24h total urine protein excretion (TPE) from baseline to week 12
Change in 24h UPCR from week 3 to week 12
Steady-state trough concentration of BI 764198 at week 4 and 12
Change in eGFR from baseline to week 12
Statistical analysis
All statistical analyses were performed using SAS software, version 9.4, including PROC GLM and PROC LOGISTIC for efficacy analyses. The final analysis sets were as follows:
1) The Per Protocol Analysis Set (PPS): Contains all patients who were randomized and completed treatment with measurements of the primary endpoint at both baseline and end of treatment. It is used in sensitivity analysis.
2) The Electrocardiogram Pharmacokinetic Concentration Analysis Set (ECGPCS):
This set includes all subjects from the treated set, providing at least one pair of valid drug plasma concentrations and a corresponding ECG endpoint to be used in the exposure response analysis (e.g., drug concentration and QT plots).
3) Initial UPCR change from baseline was compared through ANOVA in different groups (placebo, 20 mg, 40 mg, and 80 mg). After including the corticosteroid as a covariate, researchers used the ANCOVA variant to ensure a formal distribution of covariates among all groups, removing the confounding effect of steroids.
Sample size & assumptions: 60 participants (15 per group) were chosen without a formal power calculation. Expected responder rates were 20%, 30%, and 40% for BI 764198 doses (20 mg, 40 mg, 80 mg) vs. 9% for placebo, giving a 73.4% chance of detecting a ≥25% difference. Missing data were not imputed.
Interim analysis: An exploratory, unmasked interim analysis was done on the first 24 participants by the sponsor’s internal team, but no protocol or analysis plan changes were made. Trial staff and participants remained blinded.
Primary endpoint analysis:
Binary endpoint: ≥25% reduction in UPCR at week 12. Missing data classified as non-responders (hypothetical estimand).
Continuous endpoint: Change in log-transformed UPCR at week 12 (treatment policy estimand).
Statistical methods:
Logistic regression for binary endpoint (fixed effects: treatment, baseline UPCR, corticosteroid use).
ANCOVA for continuous endpoint (same covariates).
No multiplicity adjustments (exploratory phase 2 study).
Side effects estimated via targeted maximum likelihood; 95% CIs calculated accordingly.
Secondary endpoints & safety: Reported descriptively. Safety was explored with Wilson score CIs.
Funding
Funding was provided by the pharmaceutical company Boehringer Ingelheim. BI was involved in the design and conduct of the study, data collection, data analysis, and data interpretation, and funded medical writing support for the article.
Results
From March 10, 2022, to Sept 3, 2024, 139 participants were screened, and 67 were randomly assigned to receive placebo or BI 764198 at doses of 20 mg, 40 mg, or 80 mg.
Sixty-two participants received treatment; among them, 90% completed the study and treatment period.
Fig 1. Trial profile from Trachtman H et al, Lancet, 2026.
The study had a male predominance of 60%, a mean age of 40.7 years, and a predominant white race of 63%. At random allocation, the baseline drugs included 81% of patients on ACEI/ARBs, 42% on SGLT2 inhibitors, and 23% on corticosteroids. Ten participants with documented TRPC6 variants (genetic variants) were enrolled, and seven had complete proteinuria data.
Table 1. Demographic and disease characteristics from Trachtman H et al, Lancet, 2026.
In the 60 patients in the full analysis set, the primary endpoint, a 25% or greater reduction in UPCR from baseline at week 12, was seen in one (7%) of 14 participants receiving placebo and in 16 (35%) of 46 participants across all BI 764198 dose groups (odds ratio [OR] 4.9 [95% CI 1.0–48.8]).
Table 2. Primary, secondary and clinical endpoints of interest from Trachtman H et al, Lancet, 2026.
In the ANCOVA analysis, the 20 mg group saw a -40% decline in UPCR (95% CI: -56% to -17%). At the end of 12 weeks, 9 (23%) of BI 764198 patients attained less than 1 g/g of proteinuria. In the subgroup analysis of the TRPC6 variant set, which included 7 patients receiving the drug, there was a 100% response rate, indicated by a reduction in proteinuria of more than 25% from baseline. Though there was a rise in serum creatinine due to reversible inhibition of transport channels MATE1, MATE2-K, and OCT2, with no change in serum cystatin C and eGFR with cystatin C at 12 weeks of treatment.
Treatment-related adverse events were similar in both the drug and placebo groups. Serious adverse events that were seen in the 20 mg group included: 1) edema and 2) osteonecrosis, but neither of them was attributed to the drug. Among 3 patients, the drug was stopped, but all were non-serious events and occurred in patients receiving 40 mg. A mild increase in liver enzymes was seen in one person in the placebo group, and non-severe QT prolongation without any events was seen in one person receiving the 80 mg dose. Lenticular opacity was seen in one person receiving the 20 mg dose.
Table 3. Adverse events from Trachtman H et al, Lancet, 2026.
Discussion
Discussion
This phase 2 study demonstrated that the TRPC6 inhibitor BI 764198, a once-daily oral treatment for primary and TRPC6 variant FSGS, achieved significant reductions in proteinuria compared to a placebo. The study drug was well-tolerated, with a similar adverse event profile as the placebo. Stability of blood pressure and eGFR was presented as circumstantial evidence for a ‘direct’ beneficial effect of the study drug on podocytes, rather than as an effect secondary to hemodynamic alterations. The authors do note that a 20 mg dose was sufficient to achieve 90% inhibition of TRPC6 in vitro, and higher doses did not appear to have additional benefits or risks.
It was clear that the trial faced difficulties in recruitment, despite being rolled out in 11 countries and multiple sites. This is acknowledged, while key amendments in version 3.0 dated Oct 2022 were stated. To ease the burden on participants, the frequency of visits was reduced, and single readings of baseline UPCR were made acceptable. Likewise, to ease the burden on trialists, inclusion criteria were expanded to include patients on stable steroid doses (for 4 weeks minimum), in contrast to the prior criteria of being off steroids > 4 weeks. Even the proteinuria criteria were lowered to ≥ 1gm/gm compared to the previous of ≥ 1.5gm/gm, making this study less aligned with findings from PARASOL.
For being a phase 2a trial with a small sample size, the trial is heavy on statistics. Sample size calculations was not based on a formal power calculation but rather on expected response rates (20-40% vs 9% placebo), with unclear provenance (phase 1 signal vs external data not explicitly stated). The reported ~73% probability falls short of conventional power thresholds (80-90%), implying a non-trivial risk of false negatives. With ~15 patients per arm and heterogeneous inclusion criteria, signal detection becomes fragile. Dropouts were not accounted for, and missing data were not imputed, which is unusual and may have contributed to bias in the results. Wide-ranging baseline UPCRs are known to cause large variances in confidence intervals. To reduce the skewness, log-transformed urine protein measures were not used, and instead, 24-hour UPCR and total proteinuria were used to report the findings. Homoscedasticity (homogeneity of variance) violation, fifteen-yard penalty? This alone could invalidate statistical significance, which assumes that modeling errors all have the same variance. Biased standard errors lead to biased inferences, so the results of the hypothesis test are possibly wrong. This is a common problem with ANOVA statistical methods.
The authors state, “no missing data was imputed” several times. However, in an attempt to account for the intercurrent events causing missing data (including those leading to discontinuation, after which patient data was not collected), a hypothetical estimand was used. In practice, this assumes outcomes as if patients had remained on treatment- functionally similar to imputation, but without actual observed data. Conversely, the treatment policy estimand requires outcomes data irrespective of intercurrent events, but such data were not collected, creating a mismatch between the estimand definition and available data. For a phase 2 trial, this flexibility may be intentional- aimed at identifying a directional signal rather than definitive effect estimation. ORs are too wide for the primary endpoint (reflecting heterogeneity and small sample size again), giving us merely a ‘sense of direction of effect’ rather than an ‘effect size’, which may be good enough a reckoning for a phase 2 trial. And yet, the study findings support the adequacy of a 20mg dose of BI 764198.
Safety data was reported as per participant and not as the number of events, raising a question about the suitability of this approach in a phase 2a trial. A definitive statement regarding any participants having >1 AEs/SAEs would have helped; however, this is not to be found in the paper. If indeed a proportion of participants had multiple AEs/SAEs, it is supremely important that safety information is plainly stated in the paper.
Certain pharmacodynamic endpoints mentioned in the initial protocol are not mentioned in the current study. These endpoints are urinary biomarkers reflecting podocyte health (podocin [mRNA]: creatinine ratio [UPodCR], nephrin [mRNA]: creatinine ratio [UnephCR], and podocin [mRNA]: nephrin [mRNA] ratio [UPNR]) and drug target modulation (TRPC6 mRNA, nuclear factor of activated T-cells [NFAT] mRNA, and downstream markers of the calcineurin-NFAT pathway). This data could further support the idea that the effects are related to the improvement of podocyte function and longevity. Similarly, although pre-treatment biopsies were required, no post-treatment biopsies were performed.
Finally, the primary endpoint was a reduction in proteinuria, a reasonable surrogate for progressive CKD in many instances. FSGS has had difficulty identifying directed therapies because it is not a single disease process but rather a microscopic description of multiple nephropathologies. Is it useful to include all primary FSGS in a trial, or is there too much “noise” to determine a drug's effectiveness? There were animal models that showed that this medication might have down-steam effects on a common fibrosis pathway (Wu YL et al, KI 2017; Zheng Z et al, Int J Mol Sci 2022) and hence be useful in all varieties of FSGS and even diabetic nephropathy (Ran Sun et al, Mol Med Rep 2024). Although the numbers were small, the effects were certainly more impressive for those with a known TRPC6 mutation. We await more evidence for this, particularly from the NEPTUNE cohort, where biomarker-matched therapy is offered to TRPC6 variant-harboring participants (currently underway). Finally, is it proper to project expectations of eGFR from a 12-week trial in a disease like FSGS with significant variation in progression? Although it might be mildly reassuring that there was no change in cystatin C eGFR, this will need to be followed for a much longer period to show changes in chronic slope versus placebo.
Summary by
Reviewed by
Brian Rifkin, Swapnil Hiremath, Cristina Popa, Akshaya Jayachandran
Header image designed by AI prompts from Dr Saivani Yellampalli