We had a lot of fun (sleepless hours) covering the simultaneous publications at Kidney Week. Since there are so many trials happening in nephrology now, even the ISN World Congress and the ERA have many trials and simultaneous publications. The jet lag (WCN being in Japan) also means that we are up at odd hours with not much to do (and without Bill Murray’s company). Hence we decided to cover the trials from Yokohama as well. Enjoy.
In this edition
The Assist trial (Efficacy and Safety of Atrasentan in Patients with IgA Nephropathy Receiving Sodium-Glucose Cotransporter 2 Inhibitors: Placebo-Controlled, Crossover Trial; Heerspink et al, JASN 2026)
24-month data from APPLAUSE-IGAN (Iptacopan in IgA Nephropathy — Final 24-Month Data; Barratt et al NEJM 2026)
Sexual Dimorphism and Age Effects in CKD and Type 2 Diabetes in the CONFIDENCE Trial (Agarwal et al, NDT, 2026)
Acute eGFR Changes and Their Mediation of Albuminuria Reduction with Empagliflozin and Finerenone (Agarwal et al, JASN, 2026)
The Assist Trial
J Am Soc Nephrol. 2026 Mar 29. doi: 10.1681/ASN.0000001076. Online ahead of print.
Efficacy and Safety of Atrasentan in Patients with IgA Nephropathy Receiving Sodium-Glucose Cotransporter 2 Inhibitors: Placebo-Controlled, Crossover Trial
Hiddo J L Heerspink, Irene L Noronha, Jose Luis Górriz, Soo Kun Lim, Sradha S Kotwal, Gianna M Kirsztajn, José Barros Neto, Jessica Ryan, Mei Sian Fu, Sung-Gyun Kim, Jonathan Barratt, Yasmin Brahmbhatt, Greggory J Housler, Rong Jiao, Marion Dahlke, Amit Lodha, Amy K Mottl; Atrasentan and Sodium Glucose Co-transporter-2 Inhibitor Efficacy and Safety Trial (Assist) Investigator Group
PMID: 41904616
VA created for JASN by Edgar Lerma
Why was this trial needed?
The natural history of IgA nephropathy (IgAN) is unforgiving: even after optimal RAS blockade, a proteinuria >0.5g/day predicts a 30-35% risk of end-stage kidney disease within two decades (KDIGO IgA group, Kidney Int, 2025 | NephJC summary). Recent “renoprotective” breakthroughs have turned flozins into the new backbone of supportive care (or “palliative” if you agree with Dr Barratt’s terminology) care. In the DAPA-CKD and EMPA-KIDNEY trials, patients with IgAN experienced a 26% reduction in albuminuria and a 71% decrease in the composite renal endpoint (Wheeler et al, Kidney Int, 2021| EMPA-KIDNEY Collaborative Group, Lancet Diabetes Endocrinol, 2024). Their mechanisms (tubulo-glomerular feedback-mediated reduction of intraglomerular pressure, natriuresis, anti-inflammatory, and podocyte-protective effects) soften the hemodynamic injury that fuels disease progression (Kim et al, NDT, 2026). Yet flozins alone leave a sizable residual risk. In both trials (which were not IgAN focused), median proteinuria after treatment remained >0.8 g/day, and the eGFR slope, although flatter, did not halt decline (Wheeler et al, Lancet Diabetes Endocrinol, 2021). Therefore, the sponsors and investigators believe IgAN calls for an additional, mechanistically distinct agent that targets the endothelin 1 (ET-1) pathway, a key driver of glomerular vasoconstriction, inflammation, and fibrosis. One could argue that targeting B Cells (or even complement - see the #NephMadness match-up) is more disease modifying than using Flozins and ETAs. Atrasentan, a selective endothelin-A receptor antagonist, achieves precisely this. In the phase-3 ALIGN study, atrasentan lowered the UACR by 38% versus a 3% rise with placebo (difference -36%, p<0.001) after 36 weeks, with a safety profile that lacked clinically relevant fluid overload (Heerspink et al, NEJM, 2025). Earlier SONAR data in DKD confirmed the proteinuria benefit and highlighted a manageable fluid retention signal when patients were carefully selected for low-risk phenotypes (Heerspink et al, Lancet, 2019 | NephJC summary).
The next logical question is whether adding atrasentan to a flozin amplifies the antiproteinuric effectS without compounding adverse effects. Real-world evidence with the dual endothelin-angiotensin blocker sparsentan (which shares the ETA antagonism of atrasentan) showed a 62% reductions in UPCR at 14 weeks when patients were already on maximal RAS blockers plus flozins (Schanz et al, CKJ, 2024). However, sparsentan confounds the interpretation because it simultaneously blocks the AT1receptor; a pure ETA antagonist, like atrasentan, is needed to isolate the endothelin contribution.
How was the trial done and what did it report?
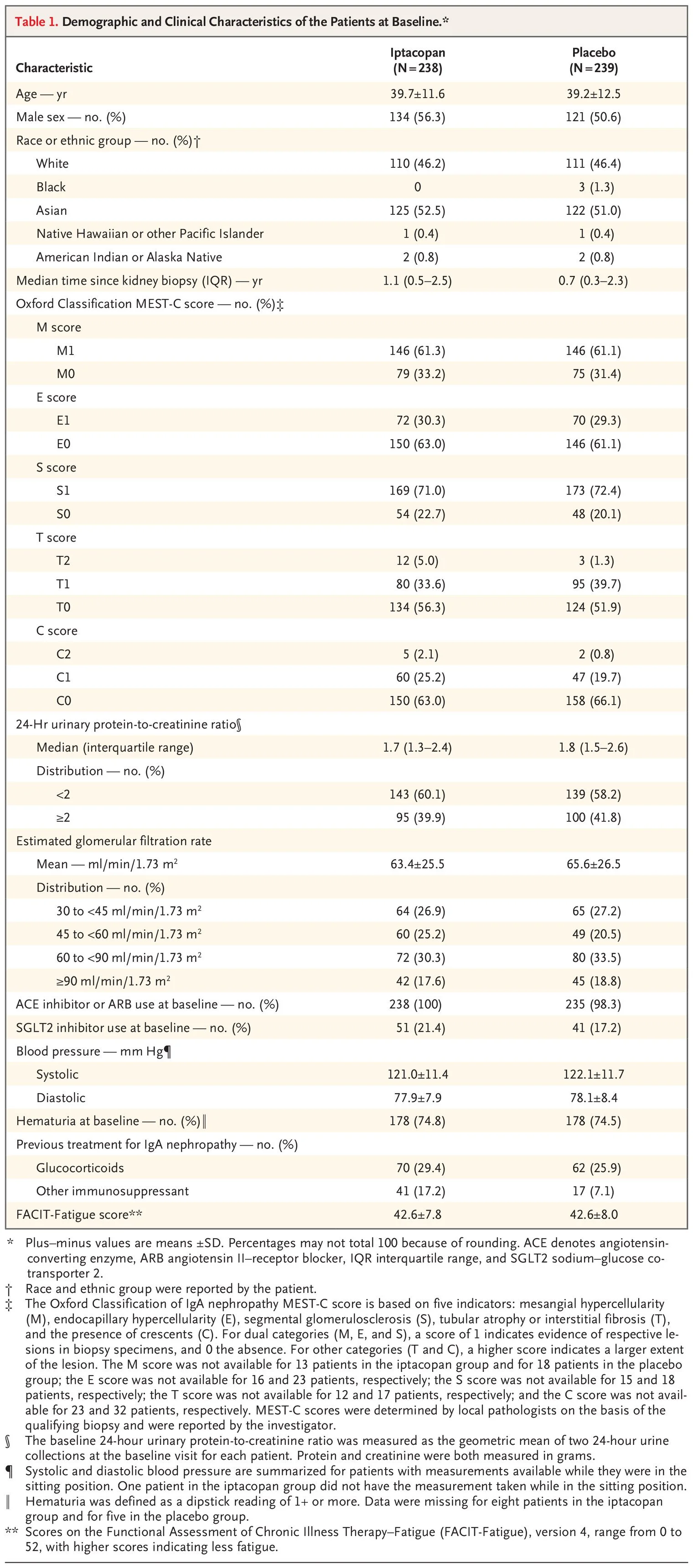
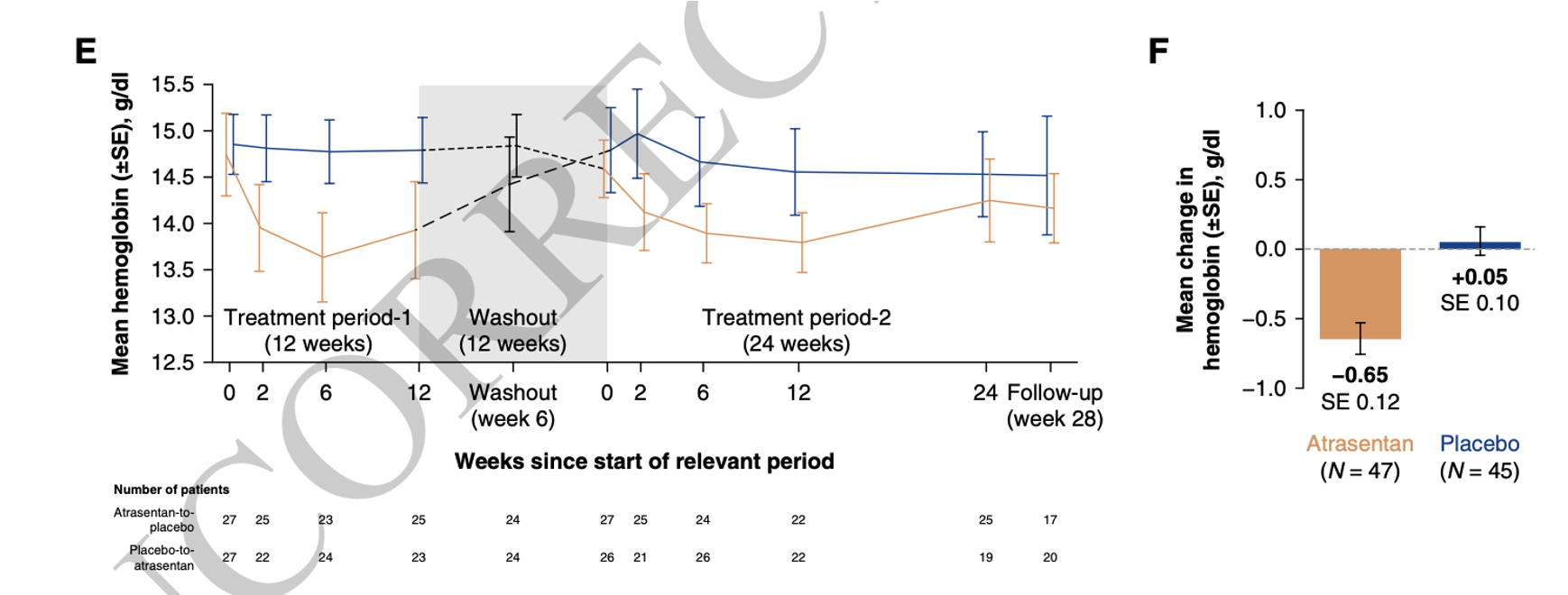
The ASSIST phase II trial was a double-blind, placebo-controlled crossover study in which each participant receives atrasentan (0.75 mg daily) for 12 weeks, a 12-week washout, then placebo (or reverse sequence) while remaining on stable flozin dosing (Heerspink et al, Kidney Int, 2021). The crossover format maximizes statistical power- each subject acts as its own control - while limiting the sample size needed to detect modest but clinically meaningful changes in proteinuria and eGFR. Moreover, the trial stratifies enrollment by SGLT2i status (stable vs run-in) to ensure that any safety signal, such as the fluid retention observed in SONAR, is captured in the exact context of contemporary IgAN care. The proteinuria required for enrolment was >0.5g/day for flozinated patients, and > 0.85 g/day for flozin-naive, who underwent a flozin run-in period to ensure their post flozinated albuminuria was still high enough. Notable exclusions were type 1 DM (flozin’s risk for causing DKA), prior history of heart failure or BNP > 200 pg/ml, or anemia (Hgb < 9 g/dL), as ETAs can cause heart failure and anemia. The primary outcome was proteinuria reduction at 12 weeks, see figure 1 for details. The trial was powered for a 25% reduction in UPCR, requiring 52 participants.
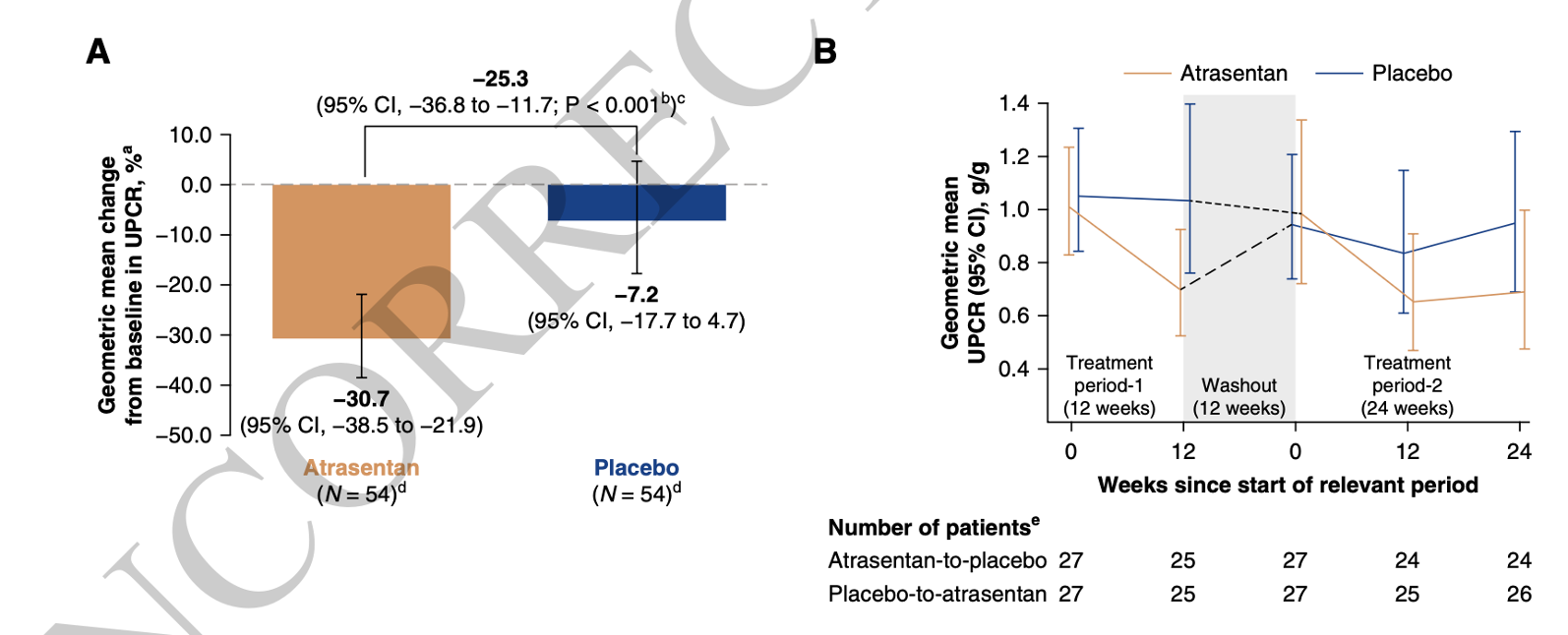
Figure 1 from Heerspink et al, JASN 2026
Overall, 54 patients were randomized, with a median GFR of 63 and UPCR of 1 g/g, and were mostly (65%) White. Only 13% were non-flozinated at baseline, requiring a flozin run-in phase. Atrasentan reduced proteinuria by 30% (versus 7% in placebo), thus hitting the powered for target and coming in at −25.3% (95% CI, −36.8 to −11.7; p < 0.001). Treatment sequence did not matter, and for those in sequence two who went on to receive atrasentan, there wasn’t much more of a proteinuria decrease at 24 weeks (treatment difference of -26.4% versus placebo).
Figure 3. 24-hour UPCR (primary outcome)
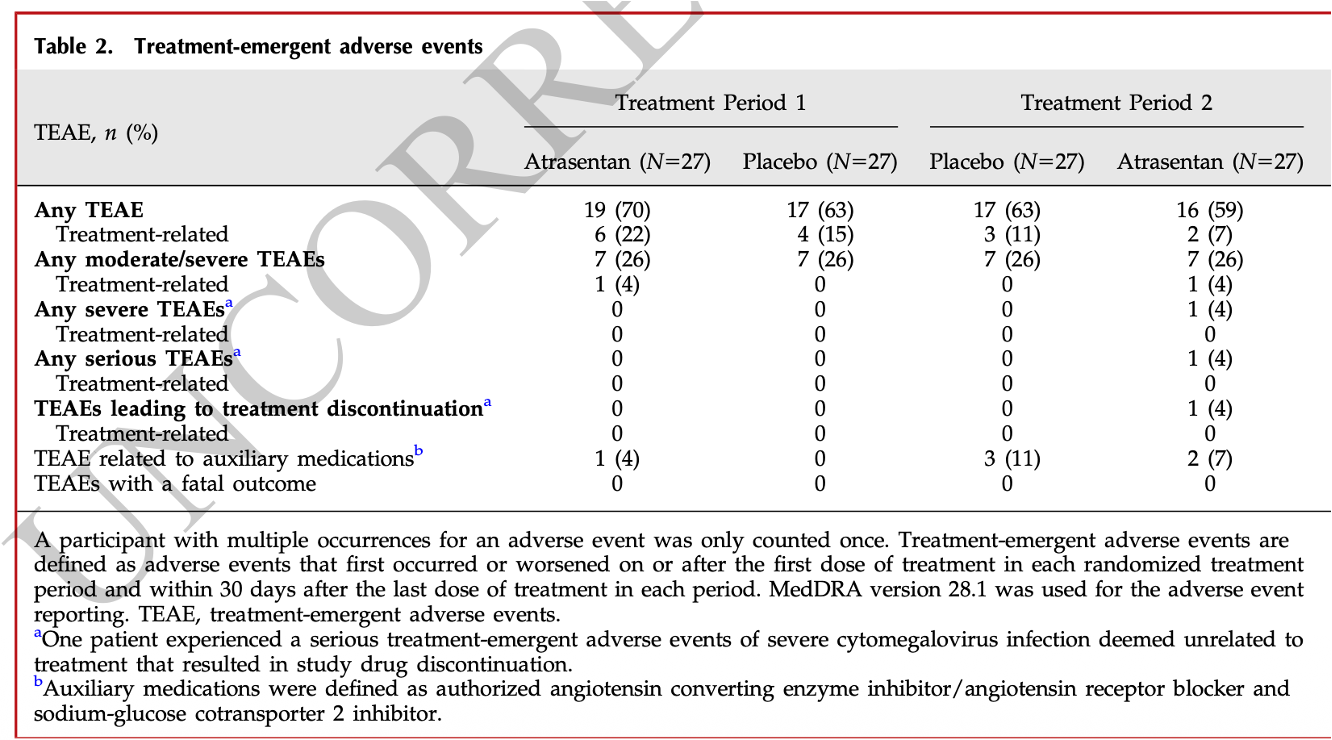
The GFR slope at 12 weeks was a meaningless: 1.2 versus 1.5 ml/min/1.73m2. Though overall safety events seem similar, one patient had AKI deemed to be related to atrasentan, fluid retention was twice as common with atrasentan, BNP increased in sequence 1 but not sequence 2, and the hemoglobin dropped a bit with atrasentan.
What does this trial change?
The trial proved atrasentan reduces proteinuria at 12 weeks in flozinated participants with UPCR > 0.5 g/day by about 25%. Does this matter, and is it really as safe as the authors claim? We would posit the answer to both is, “No”.
Firstly, IgAN therapeutics are a crowded space with many efficacious disease modifying drugs (steroids, targeted-release budesonide, BAFF/APRIL, and complement inhibitors). Are ETAs disease-modifying or supportive/palliative therapies like RASi and flozins? Theoretical effects of ETAs aside, it is difficult to see them being disease modifying. Their effects are likely hemodynamic (the mechanistic lack of a huge effect on BP in an underpowered RCT should not be overinterpreted). Time will tell how good they are in establishing a position for themselves in this space.
Unfortunately, safety remains a concern which cannot be allayed in a 54 patient RCT of 12 to 24 weeks of exposure. Even in this carefully selected (preserved GFR, no heart failure, low BNP) population, there were some concerning BNP signals (albeit in one sequence) and an associated hemoglobin drop.
This trial is useful in showing the pharmacologically and volumetrically rationale combination of a flozin and ETA (note the ongoing trial with zibotentan and empagliflozin in DKD summarized here) is a promising option that attenuates the sodium retention abilities of ETAs while synergizing the anti-proteinuric effects. Will this supplant B-cell therapies? Again, the answer is, “No”. Is it good to have more options for the ‘most common primary glomerulonephritis’? To this we give a halfhearted, “Yes”.
The APPLAUSE-IgAN Trial 24 month Data
N Engl J Med, 2026 Mar 29. doi: 10.1056/NEJMoa2600743. Online ahead of print.
Iptacopan in IgA Nephropathy — Final 24-Month Data
Jonathan Barratt, Necmi Eren, Naoki Kashihara, Bart Maes, Dana V Rizk, Brad Rovin, Hernán Trimarchi, Hong Zhang, Weiming Wang, Ismail Kocyigit, Chuanming Hao, Vladimir Tesař, Kenan Turgutalp, Li Yang, Guangqun Xing, Valter Duro Garcia, Seung Hyeok Han , Wanhong Lu, Antonio Pisani, Julia Weinmann-Menke, Annabel Magirr, Ronny Renfurm, Thomas Hach, Vlado Perkovi; APPLAUSE-IgAN Study Group
PMID: 41910396
Why was this trial needed?
Yet another IgAN trial? (in fact, ASSIST and APPLAUSE were part of a dedicated IGAN trial session, and sandwiched a fascinating phase 2/3 trial of targeted release budesonide - aka nefecon alternative - that we look forward to reading when published). But our cynical snark aside, the foundation for the APPLAUSE-IgAN study rests on an established unmet need, a strong mechanistic rationale, and the regulatory requirement for confirmatory evidence.
Unmet need: despite optimized supportive care with RAS inhibitors, a significant proportion of patients with proteinuria > 1 g/day progress to end-stage renal disease (Reich et al, JASN 2007| Liu et al, Exp Ther Med, 2016). The use of systemic corticosteroids is limited by substantial toxicity (Kim et al, CJASN, 2025). This created the need for targeted, disease-modifying therapies. Keep in mind this RCT began enrolling in 2020, when all we had in IgAN was the initial TESTING data (Lv et al JAMA 2017 | NephJC summary).
Mechanism: the alternative complement pathway has been reported to be a driver of glomerular inflammation and injury in IgAN (Medjeral-Thomas et al, Semin Immunopathol, 2021). As Jon Barratt points out in his NephMadness commentary, it has been universally noted that for every bit of IgA deposited within the glomerulus, there is accompanying C3. The deposition of galactose-deficient IgA1 (Gd-IgA1) immune complexes in the mesangium activates the alternative pathway, leading to the formation of C3 and C5 convertases and the terminal membrane attack complex (MAC), which mediate tissue damage (Teh et al, AJKD, 2026). Iptacopan is a first-in-class oral inhibitor of Factor B, an important component of the alternative pathway C3 convertase. By selectively blocking the alternative pathway, it targets a central step in the disease’s inflammatory cascade without affecting the classical or lectin pathways, theoretically offering a more precise therapeutic strategy (Schubart et al, Proc Natl Acad Sci USA, 2019| Risitano et al, Lancet Hematol, 2021). Since we all need reminders of (or have nightmares of trying to remember) the complement pathway, here it is, along with the various therapeutic targets developed or in development.
Figure 2. The complement system as a therapeutic target in IgAN, from Teh et al, AJKD, 2026
Regulatory imperative: while the initial data (Perkovic et al, NEJM, 2025) showed that iptacopan reduced proteinuria at 9 months in a dose-dependent manner, full regulatory approval requires the 24 month GFR slope data from a large-scale phase 3 trial. Based on the 9 month data, iptacopan has already received accelerated approval from the FDA in August 2024.
NephMadness VA of 9 month proteinuria data, by Edgar Lerma
How was this trial done, and what did it report?
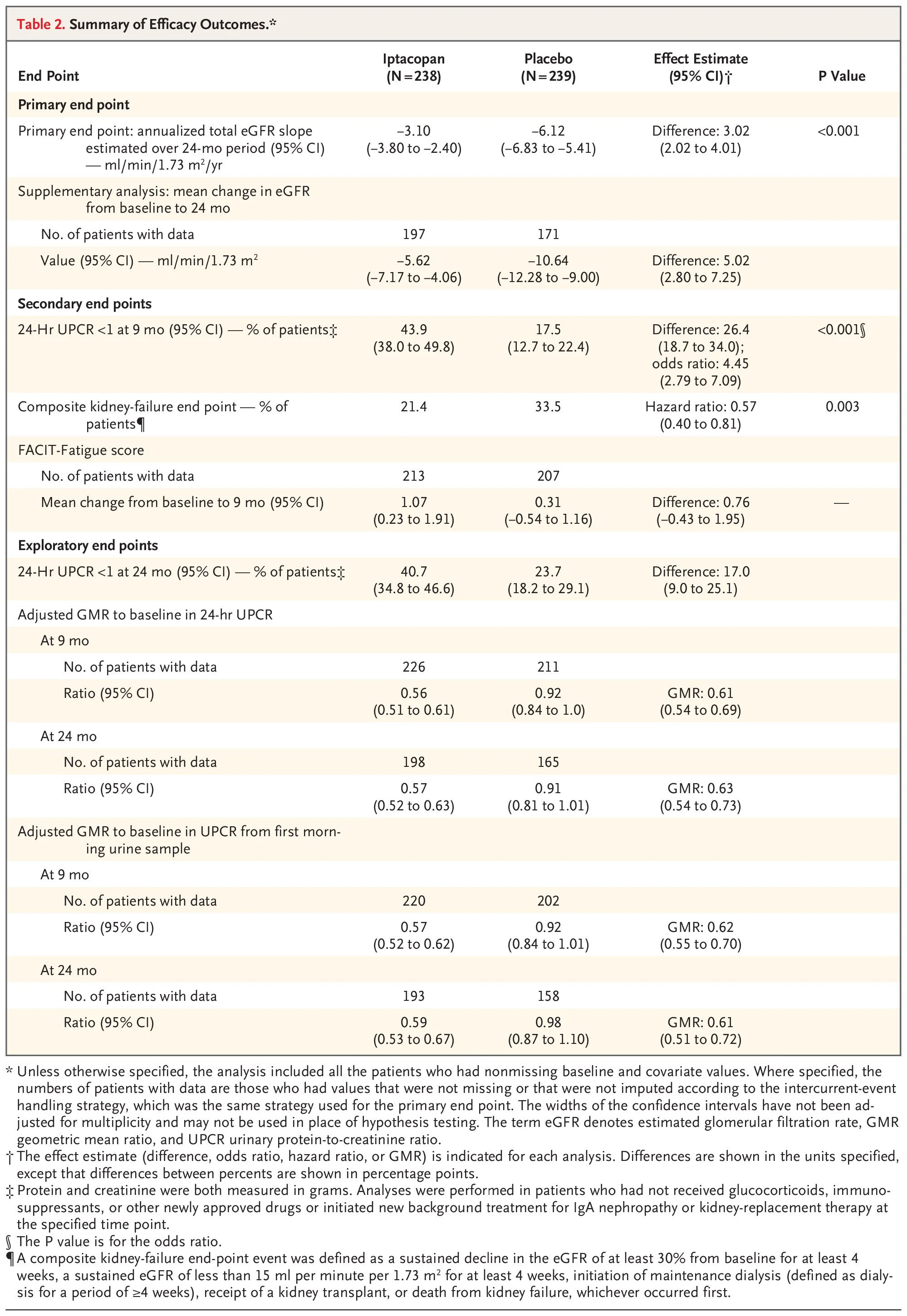
This was a large multicenter prospective placebo-controlled trial, sponsored by Novartis (the maker of iptacopan) and overseen by the academic team. It was the typical IgAN trial design that you have read already on NephJC: patients with IgAN with more than 1g/g UPCR and GFR > 30, receiving iptacopan or placebo, on a background of RASi with optional (not mandated) flozin use. Kidney biopsy was required within 2 years for those with GFR 30-45 and within 5 years for those with GFR > 45, and with less than 50% IFTA (though this was the reported and not a central pathology re-reading of biopsies). Pneumococcal and meningococcal vaccination was required, and Hib vaccination was recommended, given planned complement inhibition. The primary outcome was the annualized GFR loss over the 24-month follow-up period with several secondary outcomes including a composite kidney-failure endpoint (sustained decline in the eGFR of ≥30% from baseline, a sustained eGFR of <15, initiation of dialysis, kidney transplant, or death from kidney failure, whichever occurred first) and the change from baseline to 9 months in the score on the Functional Assessment of Chronic Illness Therapy (FACIT) – Fatigue, version 4 (on a scale from 0 to 52, with higher scores indicating less fatigue).
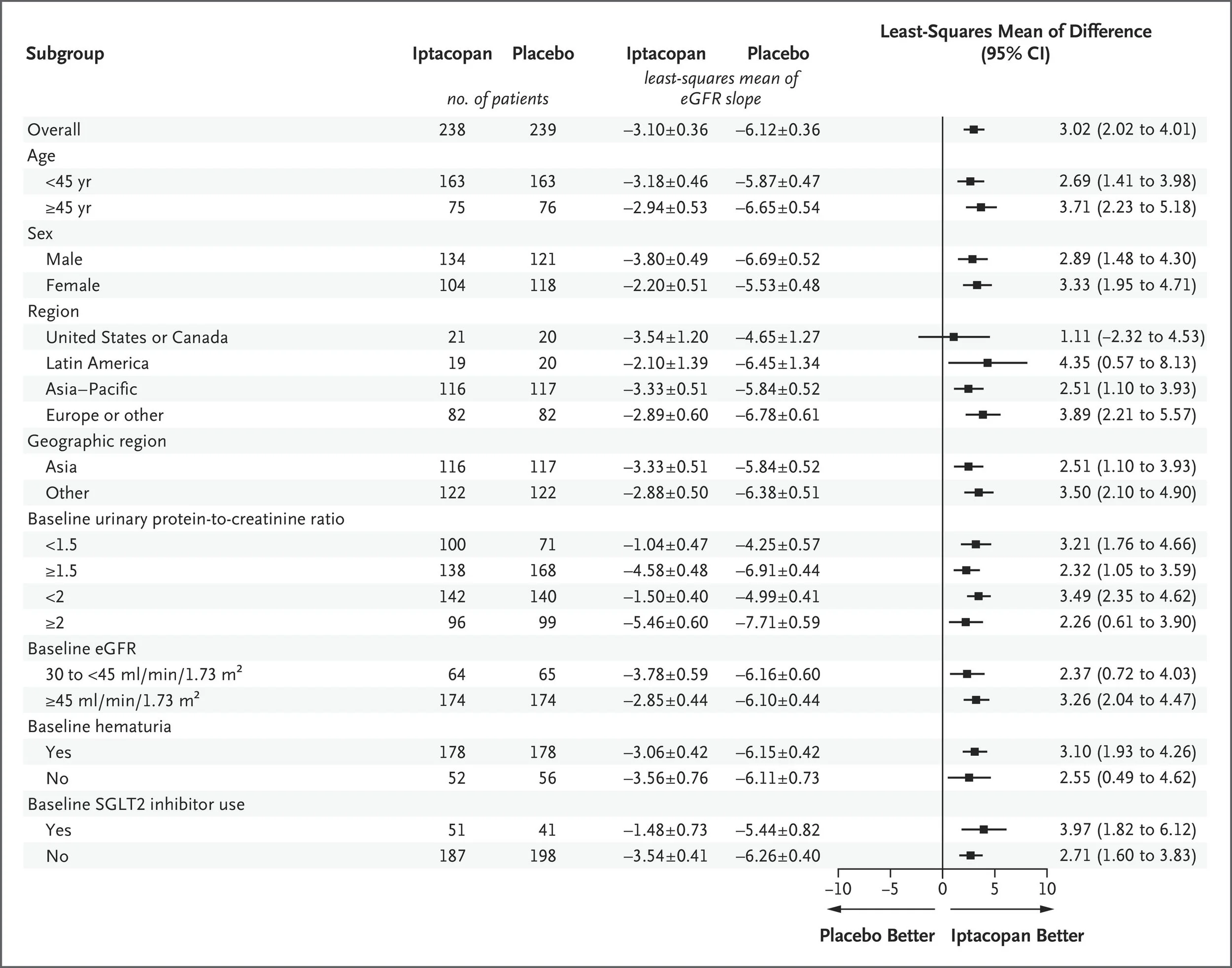
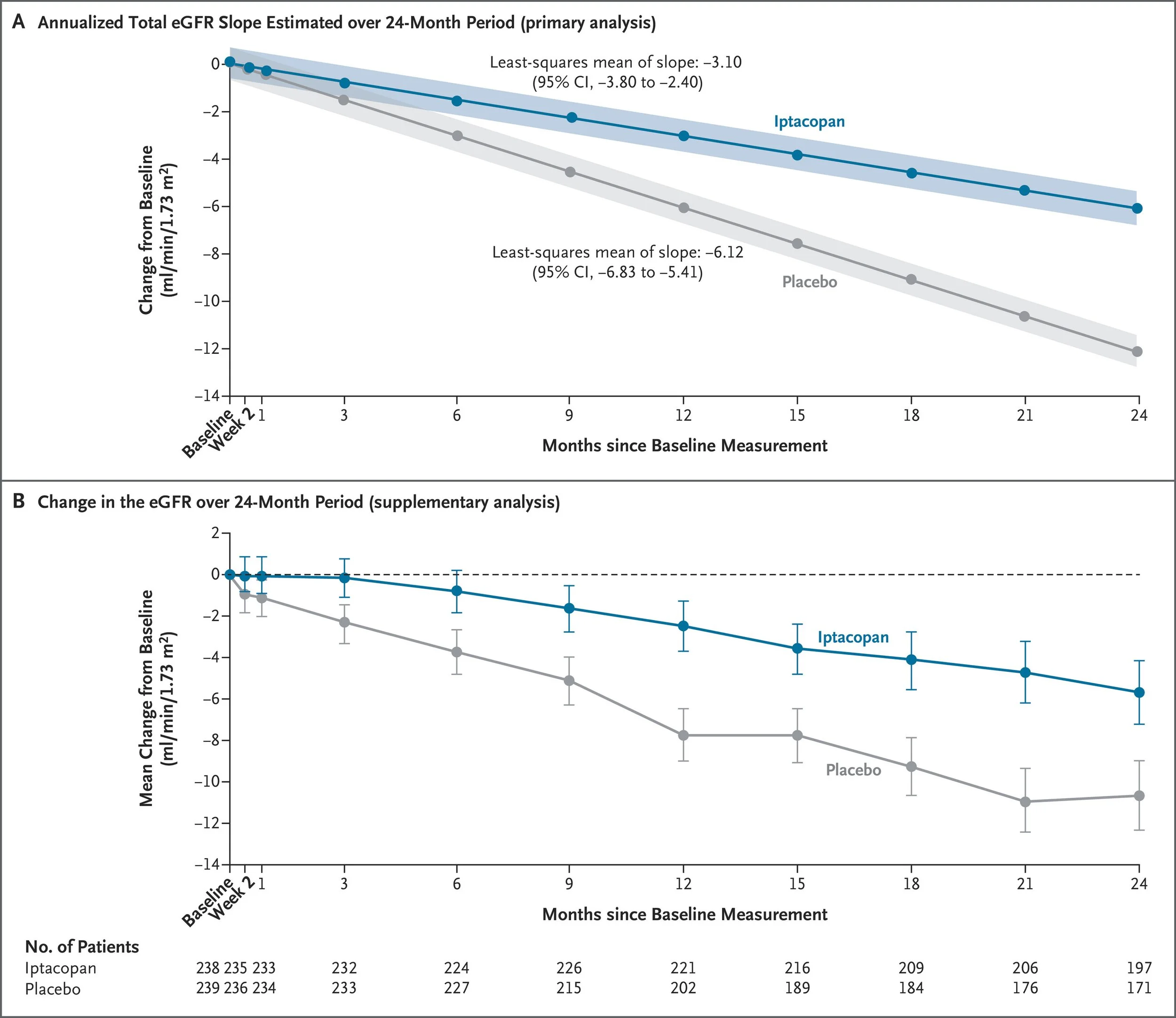
478 patients were enrolled, and the discontinuation rates were higher with placebo (37%) than with iptacopan (19%). The mean age was about 40 years, just over half were men, just over half were Asian, with the rest mostly White, a UPCR of 1.7 g/g and a GFR just over 60 ml/min at baseline. Almost all were on RASi, about 20% flozinated, with just over a quarter having previously received steroids and 75% with hematuria at baseline. Iptacopan slowed GFR progression by half, from about 6 to 3 ml/min/year (between-group difference of 3.02 ml per minute per 1.73 m2 per year, 95% CI, 2.02 to 4.0; adjusted p<0.001). The composite kidney outcome was also positive (HR 0.57, 95% CI, 0.40 to 0.81; adjusted p = 0.003) but the FACIT score was not, and thus the p values for subsequent hierarchical outcomes were not reported. From the subgroups (enrollment stratified a priori for GFR 30-45 and > 45, Asian or not, UPCR >/< 2 g/g), the effects were quite consistent. Though adverse events were overall similar, ~ 60% of them were infections (consider enrolment started 2020) and serious infections were more common with iptacopan (6.7% versus 2.1% with placebo).
What does this trial change?
Clearly, trial results deserved the APPLASE it received at the presentation. Iptacopan does slow kidney disease progression at 24 months in IgAN, and seems safe in this large phase 3 trial. Notable aspects of the trial results are the substantial 3 ml/min/year GFR decline that still occurred in the iptacopan group (compared with presented telitacicept data of almost no decline, while atacicept data is awaited), the rapid proteinuria reduction with iptacopan, and the higher infection rates despite prior vaccination and close care provided in the trial setting.
It is useful to revisit the therapeutic arena in IgAN as covered by NephMadness (and the superb GN in 10 podcast episode) to frame this discussion. We have B-cell therapies (APRIL/BAFF inhibitors) to act at the proximal hits in the IgAN pathophysiology. Complement inhibitors are clearly distal to that mechanism - so would that make them redundant in someone already on B-cell therapies? Quite possibly - or at least lower down in therapeutic importance? In an ideal world with unlimited money (or seamless access) one could even consider using both these agents? Actual disease progression is not as clean as it is portrayed in these mechanistic figures, and combination therapy might well be the best approach. We are still in the early phase of drug development in this area, so we won’t have empirical data to make these decisions and are relying on mechanisms and pathophysiology to read the tea leaves. Hopefully we will eventually have this data to make better decisions - and a drug that costs less than half a million dollars annually. Until then, we can applaud the development of another therapeutic agent, which also helps us understand how the complement pathway is intricately involved in IgAN disease progression.
CONFIDENCE: Age and Sex interactions
Nephrol Dial Transplant. 2026 Mar 27:gfag075. doi: 10.1093/ndt/gfag075. Online ahead of print.
Sexual Dimorphism and Age Effects in CKD and Type 2 Diabetes in the CONFIDENCE Trial
Rajiv Agarwal, Jennifer B Green, Hiddo J L Heerspink , Johannes F E Mann, Janet B McGill, Amy K Mottl, Masaomi Nangaku, Julio Rosenstock, Muthiah Vaduganathan, Charlie Scott, Li Li, Na Li, Carolina Aldworth, Meike Brinker, Peter Rossing
PMID: 41894225
Visual Abstract from NDT
Why was this analysis needed?
The data arrives clothed in certainty: a larger fall in albuminuria, a tolerable drip in filtration, a modest lowering of the pressure. Yet, at the bedside, this order dissolves- and biology refuses abstraction. The CONFIDENCE trial (Agarwal et al., NEJM, 2025 | NephJC summary) enrolled a broad DKD population and reported an average ≈ 30% extra UACR fall, a modest, reversible dip in eGFR, and a small systolic blood pressure (SBP) decline (Vaduganathan, NDT, 2025). Those aggregate numbers are compelling, yet they mask two biologically potent variables- sex and age- that shape the natural history of diabetic kidney disease. If the combination behaves differently in a 45-year-old man versus a 78-year-old woman, clinicians must know it before committing patients to a new regimen.
Sex is not a neutral descriptor. Men tend to experience earlier glomerular hyperfiltration and progression to end-stage renal disease, while women, despite higher CKD prevalence, progress more slowly, partly because estrogen confers vascular protection (Farahmand et al., BMC Endocr Disord, 2021). A pooled analysis of the FIDELITY data set (Bansal et al, BMJ Open, 2024) highlighted that finerenone’s reduction in heart-failure hospitalizations was more pronounced in men, whereas preliminary observations from CONFIDENCE hinted that women were more likely to achieve a >30% UACR drop (Mottl, et al, JASN, 2025). These divergent hints suggest that the finerenone-empagliflozin duet may deliver different magnitudes of benefit depending on the patient’s sex. Ignoring this possibility would be equivalent to prescribing a one-size-fits-all approach to patients with CKD.
On the other hand, age governs both risk and tolerance. Elderly patients carry a higher baseline cardiovascular risk and a reduced renal reserve, making them especially vulnerable to the acute eGFR dip and hyperkalemia that follows mineralocorticoid-receptor antagonism. Younger patients, by contrast, have the potential to harvest a lifetime of protection if aggressive albuminuria lowering is instituted early. The primary CONFIDENCE manuscript assures us that safety was “consistent across KDIGO risk categories”, yet “consistent” can conceal clinically meaningful gradients- especially in the oldest quartile, where potassium handling and blood pressure autoregulation can differ markedly (Mottl, et al, JASN, 2025). An age-stratified analysis would therefore quantify whether an 80-year-old safely tolerates the same initial dose titration as a 55-year-old and whether the magnitude of UACR reduction, and its downstream impact on hard outcomes, remains comparable.
Mechanistic certainty already exists for SGLT2i and ns-MRAs to decrease albuminuria. A mediation analysis demonstrated that early UACR reduction accounts for roughly 84% of finerenone’s kidney-protective effect and about 37% of its cardiovascular benefit, establishing albuminuria as a validated surrogate endpoint (Agarwal et al, Ann Intern Med, 2023). However, it examined finerenone alone, not the synergistic combination now under scrutiny. The CONFIDENCE interplay analysis supplies the raw efficacy signal of the duet (larger UACR decline, modest eGFR and SBP changes) but does not translate those averages into probabilities of achieving a clinically meaningful albuminuria response for a specific demographic group. Only by overlaying the mediation framework with age- and sex-specific outcomes can we predict how a 30% UACR fall will affect an individual’s risk of end-stage renal disease or heart-failure hospitalization.
How was it done, and what did it report?
To move beyond the aggregate data, the investigators conducted a prespecified exploratory analysis of the 798 patients in the CONFIDENCE trial. They employed a linear mixed-effects model to dissect the longitudinal trajectories in UACR, allowing evaluation of how age and sex influence response over the 180-day treatment period. Age was modeled both as a continuous variable and categorically by quartiles, while sex was analyzed as a binary variable. The models tested for interaction between these demographic factors and the treatment arms to determine if the benefits were uniform. The analysis showed:
A linear age-efficacy gradient: advancing age was a robust factor predicting a greater albuminuria-lowering response. The analysis quantified a significant 10.2% incremental reduction in UACR for every 10-year increase in age (95 CI: -15.5 to -4.6; p=0.001). This demonstrates that therapeutic efficacy does not wane, but scales positively with age.
Figure 2. Mean changes in UACR from baseline by treatment group as a function of age quartile, from Agarwal et al, NDT, 2026
Pronounced sexual dimorphism: the data revealed a sex-based difference in treatment response. Women achieved an 18.6% greater reduction in UACR compared to men by day 180 (95% CI 6.3 to 28.2, p=0.008).
Figure 1. Mean change in UACR from baseline by treatment group as a function of age and sex, from Agarwal et al, NDT, 2026
Treatment-independent biological effect: the age and sex effects were independent of specific therapy. Whether a patient received finerenone, empagliflozin, or a combination, the magnitude of their response was similarly modulated by their age and sex. This points toward a fundamental biological mechanism rather than a drug-specific interaction. Importantly, the enhanced efficacy in older patients and women was not accompanied by a compromised safety profile; the incidence of adverse events, including hyperkalemia and acute kidney injury, remained consistent across all age and sex cohorts (see table 4).
What does this analysis change?
The analysis reframes the interpretation of the CONFIDENCE trial, shifting it from a straightforward report on additive efficacy to a more nuanced text on precision nephrology. However, this enthusiasm must be tempered by the analyst's inherent limitations.
First, it provides a compelling, if preliminary, counter-narrative to the “prescribing paradox” in geriatric medicine. The pervasive clinical inertia that leads to under-prescription in older patients is directly challenged by data suggesting that albuminuria reduction is enhanced with age. This provides an evidence-based rationale to treat the elderly more aggressively (or at least, just as aggressively). A crucial note of caution, however, is that this conclusion is based on a 180-day reduction in a surrogate marker. We are assuming that this ultimately translates into cardiorenal benefits.
Second, it generates a powerful hypothesis regarding the “female paradox” in DKD. The superior response in women makes the authors say that the historical disparities in outcomes may stem from systemic undertreatment, rather than biological resistance. However, while statistically robust, these exploratory findings are from a trial not powered to definitively assess sex-specific outcomes. Though prespecified, these are from smaller subgroups looking at surrogate outcomes. Never-the-less these signals may inform future studies.
Finally, while this analysis sharpens our clinical focus, it is important to interpret its findings with scientific precision. The lack of a significant treatment interaction means that age and sex are prognostic factors for the response to these classes of drugs, not that the synergy of the combination is uniquely amplified in these subgroups. An older woman responds better to empagliflozin, finerenone, and their combination. Furthermore, the reliance on albuminuria as a surrogate endpoint, though supported by mediation analyses, is not suitable for hard outcome data. History is replete with trials where promising changes in surrogate markers failed to translate into clinical benefit.
Conclusion: This analysis doesn’t provide a definitive new set of rules but rather a map. It changes the questions we must ask at the bedside: not “Does this patient need combination therapy?" but, "How might this patient’s age and sex profile predict the magnitude of their response?” It forces a reevaluation of clinical biases, while simultaneously highlighting the urgent need for long-term outcome data to validate these provocative, but as yet incomplete findings. Bottom line, treat older people and women just as aggressively as you treat younger men. No need for sexism or ageism while erecting your four pillars of DKD.
CONFIDENCE: Mediation analysis
J Am Soc Nephrol. 2026 Mar 29. doi: 10.1681/ASN.0000001071. Online ahead of print.
Acute eGFR Changes and Their Mediation of Albuminuria Reduction with Empagliflozin and Finerenone
Rajiv Agarwal, Ricardo Correa-Rotter, Sankar D Navaneethan, Kei Fukami, Hiddo J L Heerspink, Johannes F E Mann, Janet B McGill, Amy K Mottl, Masaomi Nangaku, Julio Rosenstock, Peter Rossing, Muthiah Vaduganathan, Charlie Scott, Li Li, Carolina Aldworth, Jennifer B Green, Matthew R Weir
PMID: 41905767
Visual abstract for JASN by Edgar Lerma
Why was this analysis needed?
The progression of CKD is propelled by a destructive interplay among declining eGFR, rising systolic blood pressure (SBP), and increasing UACR. The pathophysiological triad creates a vicious cycle where systemic and intraglomerular hypertension damage the glomerulus, leading to albuminuria; this protein leakage then incites tubular inflammation and fibrosis, accelerating nephron loss and a further decline in eGFR (Martinez Leon et al., Nat Rev Endocrinol, 2026). Therefore, effective therapy must address the entire disease axis.
Figure 1. Pathogenesis and progression of DK, from van Raalte, et al. Nat Rev Nephrol, 2024
While foundational RAS inhibition tempers this cycle, substantial residual risk remains. The past decade has armed us with two mechanistically distinct therapies: the non-steroidal finerenone, which targets aldosterone - and possibly also inflammation and fibrosis as shown in FIDELIO-DKD (NephJC Summary) and FIGARO-DKD trials, and the flozins, which correct glomerular hemodynamics, with profound benefits seen in trials such as CREDENCE (NephJC Summary), DAPA-CKD (NephJC Summary) and EMPA-KIDNEY (NephJC Summary). Post-hoc analyses hinted at a powerful synergy, setting the stage for the CONFIDENCE trial to prospectively test the simultaneous initiation of both agents (Rossing, et al, Diabetes Care, 2022).
This context makes a detailed interplay analysis of the CONFIDENCE trial essential (Agarwal, et al, NEJM, 2025 | NephJC). The trial’s primary findings - superior albuminuria reduction with combination therapy, without a safety signal with simultaneous start - is the first step to a more complete picture. A holistic analysis of all these markers is required to confirm the therapeutic synergy, clarifying whether the UACR reduction is achieved without an excessive initial eGFR dip or concerning blood pressure effect. By mapping the trajectory of eGFR, SBP, and UACR over time, we can determine if the combination produces a more favorable cardiac/renal state than either drug alone.
Furthermore, dissecting this interplay may provide mechanistic and safety insight. It allows us to untangle the hemodynamic effect, driven by empagliflozin, from the anti-inflammatory benefits of finerenone. This moves beyond simple adverse-event reporting to reveal the dynamic physiological trade-offs of combination therapy, such as whether the natriuretic effect of empagliflozin mitigates the hyperkalemia risk posed by finerenone.
How was it done, and what did it report?
This analysis is somewhat similar to the one published in Annals for FIDELITY (Agarwal et al, Annals of IM 2023), but now applied to CONFIDENCE. In this exploratory analysis of the CONFIDENCE trial, the purpose was to ascertain if the acute eGFR decline (defined as > 30% from baseline to day 14) mediated the treatment effect of the interventions on the primary outcome (UACR decrease at 180 days). A linear mixed model using fixed effects was used for acute eGFR decline estimations. For more on mediation analysis, check out this helpful tutorial (Lange et al Epid Health 2017).
Supplemental Figure 1. eGFR decline as a mediator of UACR lowering. From Agarwal et al, JASN 2026
Determinants of acute GFR decline
The acute decline in GFR (> 30 at 14 days) was, as expected, highest in the combination, followed by empa and then finerenone - but the nadir came earlier (at day 14) for the combo and empa, and much later (~ day 90) for finerenone.
This acute GFR decline was associated in those with higher baseline GFR, and those on concomitant diuretics. Higher baseline UACR was associated with the chronic, not acute decline (noting that chronic is still only over 180 days). Notably there was not much of a GFR decline noted in those with lower levels of kidney function, GFR ~ 30.
Figure 2 from Agarwal et al JASN 2026
Determinants of any GFR decline
Examining > 30% decline at any time point over 180 days, combo therapy (versus either agent alone), higher baseline UACR, eGFR, and diuretic use were associated with this (Table 2, below).
Mediation analysis
In the mediation analysis (table 3, below), comparing the effect on UACR at 180 days that was mediated by the acute eGFR decline, this contribution was seen much more in combination versus finerenone (28%) than combination versus empagliflozin (5.2%), suggesting little of the additional benefit provided by finerenone is mediated by the acute eGFR decline.
Adverse effects by eGFR decline
These are shown in table 5, and it is notable that hyperkalemia was slightly more common in those who had an acute eGFR decline than in those who did not.
What does this analysis change?
This study provides rich granular data exploring the determinants of 30% GFR declines as well as mediation analysis of whether the acute GFR decline is important in albuminuria reduction. The decline only seen in higher GFR might reflect regression to mean or hemodynamic unloading of hyperfiltrating glomeruli. The authors posit that since only 5% of the UACR effect of finerenone mediated by eGFR decline reflects the antiinflammatory and antifibrotic (dare we say ‘pleiotropic’) effects rather than hemodynamics. This would support synergistic and independent effects of finerenone compared to flozins or RASi in DKD.
These results should not make you more or less inclined to use these drugs together, but they do help in how we think about the creatinine bumps and changes happening upon initiation. The lack of GFR decline in those with low GFR is reassuring - one should perhaps be bold at initiating flozin + finerenone together even if GFR is 30 ml/min. The hyperkalemia seen with the combination group was somewhat disappointing in CONFIDENCE, especially for those of us who were hoping empa would attenuate the hyperkalemia seen with finerenone. As suspected, (listen to Joel on the pod) this was mostly mediated by the higher rates of acute GFR decline seen with the combination. What should one make of the mediation analysis? ¯\_(ツ)_/¯. Does it matter how a drug works, if we know it works? No, not when making decisions on whether to use it - but possibly on future research pathways more than anything else.
By Swap Hiremath & Cristina Popa
Reviewed by Brian Rifkin