#NephJC Chat
Tuesday Oct 24th 9 pm Eastern
Wednesday Oct 25th 8 pm BST, 12 noon Pacific
Pediatrics. 2017 Sep;140(3). pii: e20171904. doi: 10.1542/peds.2017-1904. Epub 2017 Aug 21.
Clinical Practice Guideline for Screening and Management of High Blood Pressure in Children and Adolescents.
Flynn JT, Kaelber DC, Baker-Smith CM, Blowey D, Carroll AE, Daniels SR, de Ferranti SD, Dionne JM, Falkner B, Flinn SK, Gidding SS, Goodwin C, Leu MG, Powers ME, Rea C, Samuels J, Simasek M, Thaker VV, Urbina EM; Subcommittee on screening and management of high blood pressure in children.
PMID: 28827377 Full text free at Pediatrics
Introduction
First off, this is a beast of a paper but required reading if you work with kids or adolescents. You’ll most likely find you’ll need to print or save it for future reference including the new blood pressure (BP) normative values, recommended investigations and treatment algorithms. Ordinarily, we would spend more time in journal club discussing the methods and approaches to the problem but this paper is so vast that I think it’s best to cover this briefly and then skip straight to the recommendations.
In brief, the Clinical Practice Guideline (CPG) subcommittee was co-chaired by a paediatric nephrologist and a general paediatrician and was made up of 17 members including a parent representative. This is the first CPG I’ve seen of this quality that has patient experience front and centre along with implementation science and health economics making it an incredible resource for systemic change. A detailed description of the methodology will be published as a technical report but the authors explain in good detail their selection process. The guidelines are based on a comprehensive review of nearly 15,000 published articles from Jan 2004 – Aug 2015 using the PICOT format to address the diagnosis, workup and management of hypertension (HTN).
A quick primer for our adult colleagues
Firstly, HTN in children is based on normative values, rather than any evidence of a hard outcomes in future life. This of course means that a percentage of the healthy, normal paediatric population will always be classed as hypertensive. One may argue that perhaps this so-called 'normal' are not normal if you consider longitudinal studies such as Young Finn and Bogalusa.
Secondly, BP in children should be measured by auscultation in the right arm using an appropriate sized cuff. Screening can be done using an oscillometric machine but diagnosis and management are based on auscultatory measurements.
What are the recommendations?
The guts of this paper are in a few tables and I’m going to tackle these first. Table 1 summarises the Key Action Statements for the screening and management of high BP – there are 30 statements and table is the cheat sheet for the entire paper.
Table 1 from Flynn et al, Pediatrics, 2017
In addition to this Table 2 lists 27 consensus opinion recommendations which are also required reading. The rest of the paper works through Tables 1 and 2, reviewing the literature, expanding on the recommendations and highlighting potential concerns.
Table 2 from Flynn et al, Pediatrics, 2017
What’s changed in these recommendations?
The normal BP data in this report come from a reanalysis of the National High BP Education Program database that excludes obese children and so should be more representative of a healthy population. The prevalence of HTN in children is generally quoted as 3.5% and a higher BP in childhood correlates with HTN in young adulthood. Tables 4 and 5, give these new normative values and should be printed and given to all practicing paediatric nephrologists. The simplified format of table 6 makes an excellent quick reference tool for the office wall.
The diagnosis of HTN requires confirmed BP readings >95th percentile on 3 different visits. Following this guideline is going to mean multiple visits for children, even before treatment is initiated, which is acknowledged as a potential barrier to implementation by the authors. The flow chart in figure 3 gives a great guide as to how to measure BP but it is immediately obvious that this will also take time, particularly in a stressed or upset child.
Figure 3 from Flynn et al, Pediatrics, 2017
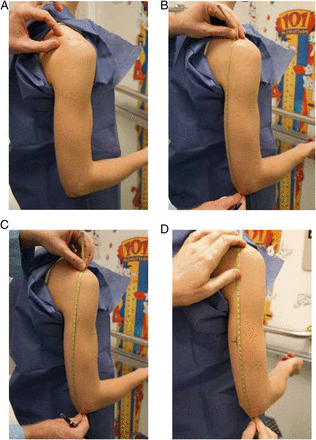
An appropriate sized cuff for BP measurement means that the bladder should encircle the arm and the authors go into some detail about measurement to determine the appropriate cuff size. Fig 2. The most important thing of course is to have easy access to a range of cuff sizes and since I’ve struggled with this in several sites across different countries, I imagine it’s a problem common to other paediatric nephrologists. Purchasing and maintaining a stock of cuffs to facilitate measurement from the preterm neonate through to the obese adolescent is a boring but essential part of practice management, this article backs up the importance of this equipment to all paediatricians. (As an aside if anyone has an efficient and attractive way to store all those cuffs please send me pictures).
Figure 2 from Flynn et al, Pediatrics 2017
Determination of proper BP cuff size.
A, Marking spine extending from acromion process.
B, Correct tape placement for upper arm length.
C, Incorrect tape placement for upper arm length.
D, Marking upper arm length midpoint
The nomenclature around BP definitions has changed for consistency with forthcoming new adult guidelines and to aid the management of adolescent. The term “elevated BP” has replaced “prehypertension”. The cut-offs are outlined in table 3.
How is this going to change paediatric nephrology practice?
As in past pediatric guidelines, all children >3 are recommended to have their BP checked, but now most children only need to be screened at their annual well-child checks (the 2004 NHBPEP recommended BP be checked every visit - which may not have happened in real life). If followed, this could result in potentially more referrals to clinic, more inpatient consults and more use of ABPM monitoring, given that the thresholds are now lower, compared to 2004. Specifically, the recommendation is that healthy children should have an annual BP check and that those at increased risk (ie those with obesity, renal disease, diabetes, aortic coarctation or those on medications which can cause HTN) should have a BP check at every health encounter. Compared to Table 8 lists common agents associated with elevated BP which could be useful for trainees and table 9 gives a list of conditions under which children younger than 3yo should have BP measured (my spidey senses tell me that we’ll be getting a lot more referrals from neonates and the sleep unit).
Routine ABPM measurements are recommended for a wider population than previously. This population includes all at risk children (table 12) including all those with CKD. More emphasis than previous is placed on abnormal circadian patterns and masked HTN (which I’ve always thought was due to great rapport with your nephrologist though everyone else stressed you out, but apparently it’s and actual diagnosis). The authors also comment that there is a higher prevalence of masked HTN in obesity which is particularly concerning for anyone working in a centre without a thigh cuff for their ABPM machine.
So what do I do next?
The workup of the child with HTN has relaxed somewhat and the guidelines now suggest that children >6yo do not require an extensive evaluation for secondary causes of HTN if they have a positive family history of HTN, are overweight and/or have an unremarkable history and exam. Whilst this is reassuring I suspect many nephrologists will continue with their previous habits – we’ve all had that one patient – and table 10 outlines the potential investigations to consider. Tables 14 and 15 give a great list of suspicious exam findings and endocrine causes of HTN which will be treasured by exam candidates.
ECGs remain out and echoes are now in, though at a more pragmatic frequency compared to 2004. An echo is recommended at the time of starting meds, if normal then it should be repeated annually in those with incompletely treated HTN. If abnormal then it should be repeated 6-12 monthly. Thankfully there’s no recommendation to perform routine vascular structural or functional scanning.
Doppler ultrasonography for RAS has a sensitivity of 64-90% and a specificity of 68-70%. The author’s advice here is eminently practical. They acknowledge that the best results are obtained in co-operative, non-obese children >8yo, who are examined in a facility with extensive experience., and that doppler ultrasonography should not be obtained in patients who do not meet these criteria. Further imaging, if RAS is suspected, should be with CTA or MRA – weighing the risk of radiation against that of a general anaesthetic.
How do I treat the child with HTN?
The use of non-pharmacological management is detailed in depth with an excellent discussion about diet and exercise. This should be trialled in the first instance in the asymptomatic child. Table 17, which details drugs for the initial treatment of HTN, will be a boon to paediatric nephrology fellows everywhere. It lists drugs by class and gives starting dose, intervals and preparations for different age brackets. Another table to print and keep handy. Table 19, which details the oral and IV treatment of severe acute HTN, has already made its way onto my bedside table for quick reference in an emergency.
First line agent should be ACE, ARB, calcium channel blocker or a thiazide – which is pretty broad and allows us all to maintain our prescribing quirks. Though an ACE should be considered the first line in CKD, proteinuria and diabetes.
There are clear recommendations about frequency of follow up and monitoring. Essentially every 3-6 months if non-pharmacological and every 4-6 weeks while titrating up on meds until BP is controlled and every 3-4 months thereafter.
What do I do for the child with HTN post-transplant?
This is a common problem for a paediatric nephrologist with generally around 50% of our patients requiring BP management in the long term. Achieving normotension in this group is linked with better graft outcomes and they are flagged as having both masked HTN and nocturnal HTN and so ABPM measurement is recommended – though just how frequently isn’t clear, I’m guessing annually but would appreciate opinions. The recommendation is for ACE or ARB as a first line agent.
Was there anything else I liked about this paper?
Three sections I particularly liked about his guideline were implementation science, economic impact and patient engagement. Practical, grounding and useful. That the challenges are common to us all, is comforting and having them in print is useful when making the case with management for extra equipment and appointments. As is the suggestion that ABPM is cost effective because it has the potential to save USD 2155 in unnecessary investigations (although the study quoted does not seem to have included patient costs such as days of work/school and transport). Reminding all of us of our role as healers and to be mindful of patient and parental perceptions is important, particularly with a symptom that is impacted by stress. Seeing this sort of section in this paper gives me hope for the future of research and the impact that patient engagement will have on our research priorities in the future.