#NephJC chat
Tuesday Oct 18th 9 pm Eastern
Wednesday Oct 19th 8 pm BST, 12 noon Pacific
J Am Soc Nephrol. 2016 Sep 9. pii: ASN.2016040433. [Epub ahead of print]
A Multicenter Study of the Predictive Value of Crescents in IgA Nephropathy.
Haas M, Verhave JC, Liu ZH, Alpers CE, Barratt J, Becker JU, Cattran D, Cook HT, Coppo R, Feehally J, Pani A, Perkowska-Ptasinska A, Roberts IS, Soares MF, Trimarchi H, Wang S, Yuzawa Y, Zhang H, Troyanov S, Katafuchi R
PMID: 27612994
Full Text freely available here, thanks to JASN!
Background
IgA nephropathy (IgAN) is the most common glomerulonephritis worldwide. Its prevalence varies regionally, ranging from less than 10% of glomerulonephritis in the United States to 30-40% in Asia (1). Prognosis is variable according to different prognostic factors:
The likelihood of dialysis or death was recently estimated with the use of three risk factors that are documented at biopsy: urinary protein excretion of more than 1 g per day, hypertension (>140/90 mm Hg), and severe histologic lesions on the basis of glomerular, vascular, tubular, and interstitial features. The 20-year predicted survival without the need for dialysis was 96% among patients with no risk factors versus 36% among those with three factors. The 10-year renal sur- vival rate is about 90% among adults and children with normal renal function at diagnosis. (2)
IgAN can strike at any age with young people typically presenting with gross hematuria, and older patients with microscopic hematuria. Male to female ratio is 2:1 in North America, while it is 1:1 in Asia. (2). Its diagnosis was based on the histopathologic and immunohistochemical evaluation of renal biopsy (3)
The Oxford classification of IgA nephropathy was developed by the International IgA nephropathy Network Group working in collaboration with the Renal Pathology Society. The Oxford Classification is composed of four scores:
- M: mesangial hypercellularity
- E: endocapillary proliferation
- S: segmental glomerulosclerosis
- T: tubular atrophy
The Oxford Study established mesangial hypercellularity (M), segmental glomerulosclerosis (S), and moderate to severe interstitial fibrosis and tubular atrophy (T) as independent risk factors for poor renal outcome. Endocapillary hypercellularity (E), although not predictive of poor outcome in the Oxford cohort, was associated with response to steroids. (3) Subsequent work has validated this system in children (4) and additional cohorts of IgA patients (5).
However, the prognostic value of crescents was not addressed due to their low prevalence in the original Oxford Cohort.
The present study had three aims:
- Determine if the presence or absence of cellular/fibrocellular crescents is predictive of poor renal outcomes in IgA nephropathy
- Determine if there is a certain fraction of glomeruli with crescents that is associated with poor renal outcomes
- Determine if crescents and/or a certain fraction of glomeruli with crescents predicts response to immunosuppressive therapy
Methods
This study is a post hoc analysis of four retrospective cohorts (Oxford, VALIGA, and two Asian cohorts).
Inclusion criteria
- biopsy–proven IgA nephropathy with pathology slides available for review
- A minimum of eight glomeruli in the Oxford and the VALIGA Study
- A minimum of 10 glomeruli in the Chinese and Japanese cohorts
Exclusion criteria included patients with
- Henoch–Schönlein purpura
- Diabetes
- IgA nephropathy secondary to liver disease
- IgA nephropathy superimposed on ANCA–associated GN
The four cohorts had 3,140 individuals (adults and children), of which 3,096 met inclusion criteria. eGFR was estimated using the Modification of Diet in Renal Disease formula in adults and the Schwartz formula in children.
A crescent was defined as extracapillary proliferation of more than two cell layers of any size; this definition was uniform in all studies.
The outcomes were: the rate of renal function decline (slope of eGFR) and survival from a 50% reduction in eGFR or ESRD (eGFR<15 ml/min).
Results
Follow was for a median of 4.7 years, during which 37% received immunosuppression and 74% received renin-angiotensin system antagonists.
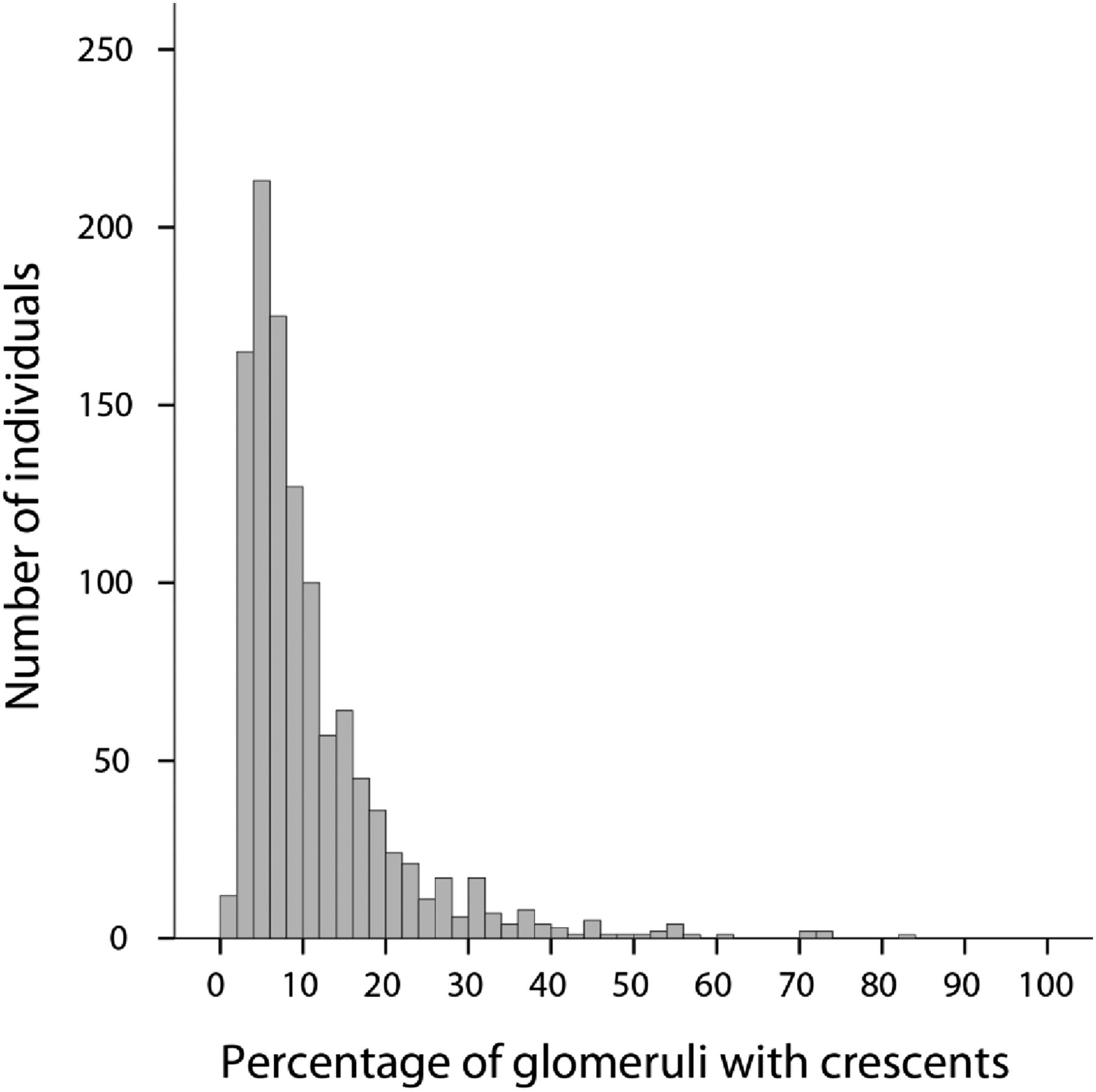
Initial proteinuria was 1.2 g/d. Mean arterial pressure was well controlled, and the follow-up proteinuria was 0.8 (0.4–1.4) g/d. Cellular and/or fibrocellular crescents were present in 36% of individuals. The rate of renal function decline was just under 2 ml/min per 1.73 m2 per year; 13% experienced a combined event, and 11% experienced ESRD.
Figure 1 from Haas et al, JASN 2016
Of those with any cellular/fibrocellular crescents:
- 61% in < 10% of glomeruli with crescents
- 20% had > 18% of glomeruli with crescents
- 9% had > 25% of glomeruli with crescents
The presence of crescents correlated with each component of the MEST score, with the strongest association found with the Endocapillary score (OR of having E concurrent with any crescents, 5.98 [95% CI 4.94 to 7.24; P<0.001]).
Thirty-three percent of patients without any crescents received immunosuppression compared with 43% of patients with crescents (P<0.001).
Figure 2 from Haas et al, JASN 2016
Patients with crescents who were not treated had greater eGFR, lower proteinuria, fewer crescents, less E, and less severe T compared with those who received immunosuppression (Table 2).
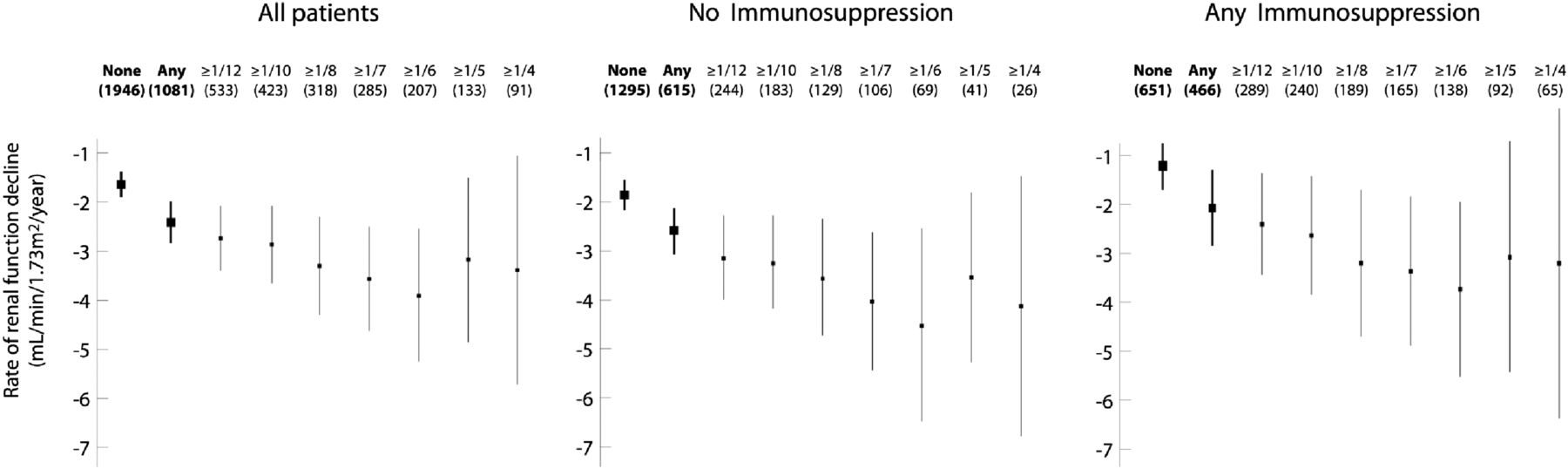
Figure 3 provides data for univariate analyses, examining the rate of renal function decline and renal survival based on crescents and fraction of glomeruli with crescents.
Figure 3 from Haas et al, JASN 2016
The presence of crescents was associated with a faster rate of renal function decline compared with its absence (−2.4±7.1 versus −1.6±5.8 ml/min; P=0.004) and a lower renal survival (HR, 1.32 [95% CI 1.06 to 1.65; P=0.01]).
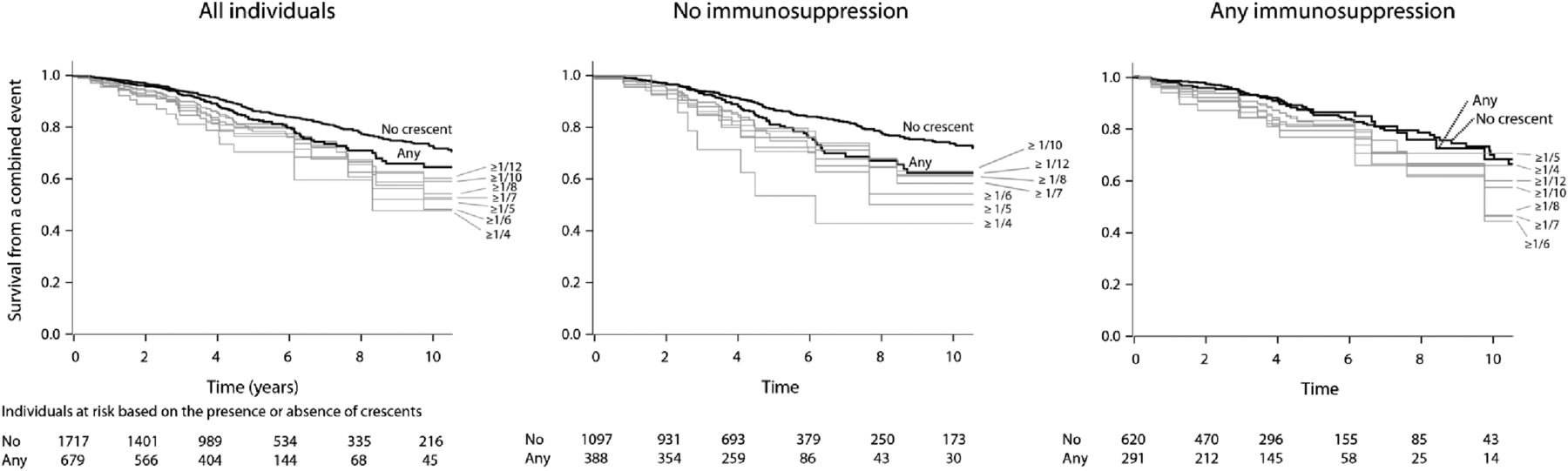
Figure 4 Survival from a combined event progressively declined up to greater than or equal to one fourth of glomeruli with crescents in the total cohort and those not receiving immunosuppression.
Figure 4 from Haas et al, JASN 2016
The proportion-dependent association between crescents and survival from the combined event was lost in patients receiving immunosuppression. Overall, treated patients did better than untreated patients.
Table 3 shows that the presence of crescents was predictive of renal survival with a HR of 1.37 (95% CI, 1.07 to 1.75; P=0.01)
However, using models with two cutoffs of crescents, untreated individuals with a higher fraction of crescents tended to progress more rapidly than those without crescents. Compared with those with no crescents, untreated patients with crescents had adjusted increases in the rate of eGFR decline.
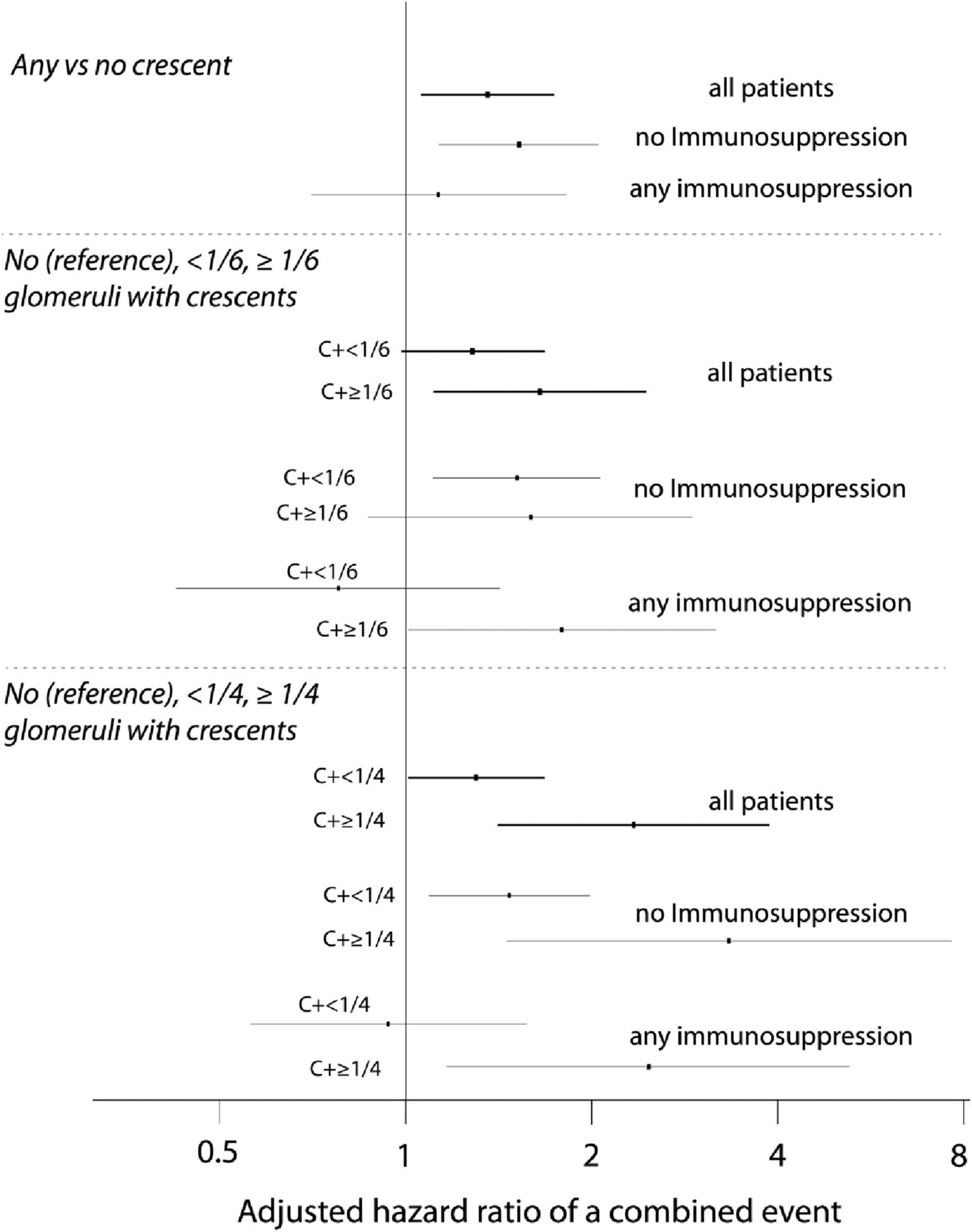
Figure 5 Adjusted HRs of a combined event using different fractions of glomeruli with crescents in all patients, patients not receiving immunosuppression, and those receiving immunosuppression. HRs are in reference to no crescents and adjusted for the MEST scores, initial eGFR, and time-averaged MAP and proteinuria. C+, with crescents.
Discussion
Since untreated individuals with a higher fraction of crescents tended to progress more rapidly than those without crescents the authors propose adding crescent scores to the Oxford Classification: C0 (no crescents); C1 (crescents in less than one fourth of glomeruli), identifying patients at increased risk of poor outcome without immunosuppression; and C2 (crescents in over one fourth of glomeruli), identifying patients at even greater risk of progression, even with immunosuppression. This criterion only applies to well-defined cellular or fibrocellular crescents and not fibrous crescents.
The selection of one fourth of glomeruli with crescents as the cutoff for C2 was on the basis of the findings shown in Figure 5, the predictive value of greater than or equal to one fourth crescents remained significant in both untreated and treated individuals, reaching HRs for the combined event of 3.26 (95% CI, 1.47 to 7.34; P=0.004) and 2.43 (95% CI, 1.17 to 5.06) for untreated and treated patients, respectively.
Limitations include
Study excluded patients with eGFR <30 ml/min per 1.73 m2 or rapid eGFR decline and did not report histologic findings
As a retrospective study, patients were from multiple centers, criteria and specific treatment regimens for treatment with immunosuppressive agents was not uniform.
Criteria for use of RASB, length of follow-up, and data collection during follow up varied.
Selection of one fourth of glomeruli with crescents as the cutoff for C2 is based on the multivariate analysis including clinical parameters, Oxford MEST scores, and the effect of immunosuppressive therapy.
Julian BA, Waldo FB, Rifai A, Mestecky J. IgA nephropathy, the most common glomerulonephritis worldwide. A neglected disease in the United States? The American journal of medicine . 1988;84(1):129.
- Wyatt RJ, Julian BA. IgA nephropathy. N Engl J Med. 2013;368(25):2402-14.
- Cattran DC, Coppo R, Cook HT, et al. The Oxford classification of IgA nephropathy: rationale, clinicopathological correlations, and classification. Kidney Int. 2009;76(5):534-45.
- Coppo R, Troyanov S, Camilla R, et al. The Oxford IgA nephropathy clinicopathological classification is valid for children as well as adults. Kidney Int. 2010;77(10):921-7.
- Roberts IS. Oxford classification of immunoglobulin A nephropathy: an update. Curr Opin Nephrol Hypertens. 2013;22(3):281-6.
Summary prepared by Scherly Leon, Nephrologist, New York (and #NSMC alumnus)