#NephJC Chat
Tuesday Jun 26 9 pm EST
Wednesday Jun 27 8 pm BST, 12 noon Pacific
This week's #NephJC is a big one. We are discussing 3 articles on renal denervation for hypertension. These articles look to rewrite the history written by Symplicity-3.
Before you read ahead - make sure you read all the actual trials - or the summaries of the three trials - linked above. SPYRAL HTN OFF, SPYRAL HTN ON and RADIANCE HTN SOLO are the three trials we will discuss this week. Check out two editorials in the Lancet: this one by Kjeldsen and Esler; and this one from Blankenstijn and Bots.
Did these trials need to be done at all?
We know that the sympathetic system is closely linked to hypertension even before all this brouhaha about renal denervation. A recent meta-analysis reported a close relationship between sympathetic activity (as measured by muscle sympathetic nerve activity - MSNA) and essential hypertension, to clinic and ambulatory blood pressures, and to left ventricular mass index. Our historical overview discusses the dramatic responses with surgical resection of the sympathetic nervous system. But since we have excellent blood pressure lowering medications, is there a need for device therapy? More importantly, SYMPLICITY HTN-3 (S-3) was gloriously (or infamously) negative, and many of us think we were fooled by a combination of regression to the mean, placebo effect and measurement bias prior to SYMPLICITY HTN-3. 'Ding-dong, denervation is dead' ran the refrain after S-3. Either renal denervation doesn't effectively ablate the nerves - or that pathway is not really that important, except for a tiny minority perhaps. Perhaps.
So there was a lot of soul searching that happened post SYMPLICITY HTN-3 (briefly covered in our historical overview). One analysis related the discrepant results from S-3 to inadequately performed denervation, in inexperienced operators hands.
A bunch of papers from the Imperial College group - principally James Howard and his irascible supervisor Darrel Francis - went over some possibilities from an epidemiological and statistical purview. As an aside, these are a must-read for anyone trying to understand the resistant hypertension literature in particular. Their initial analysis, at the peak of renal denervation hysteria, pegged the 'real' effect of denervation at perhaps 10 mm Hg (this was when the numbers being bandied about were 20 - 30 mm Hg drop).
Fig 2 from Howard et al, Heart, 2013. Pre-SYMPLICITY - 3, even the skeptics predicted an eventual result of about -10 mm, not the null result actually observed.
To reiterate, even the skeptics predicted an eventual result of about -10 mm, not the null result actually observed. Were these cynics also perhaps optimistic, or was there something else at play? The three biases that confused the investigators (and us) can be summarised as
- big-day bias regression to mean: choosing patients with really high values will result in a subsequent low value, irrespective of the efficacy of the intervention
- check-once-more bias knowing the patient has had an intervention influences measurement of blood pressure) and
- I-will-take-it-now bias a patient who underwent/believes they underwent a procedure will now be more adherent to their medications
These reasons lead to the high variance seen - which increases with the number of medications taken, as is the case in resistant hypertension. A Catch-22 situation - the patient population in whom there is equipoise for invasive interventions will confound your careful clinical trial. So what is the way out? Something counterintuitive:
- a trial in a population not taking blood pressure medications, or withholding medication for the trial period
- confirm adherence the best way you can during the trial
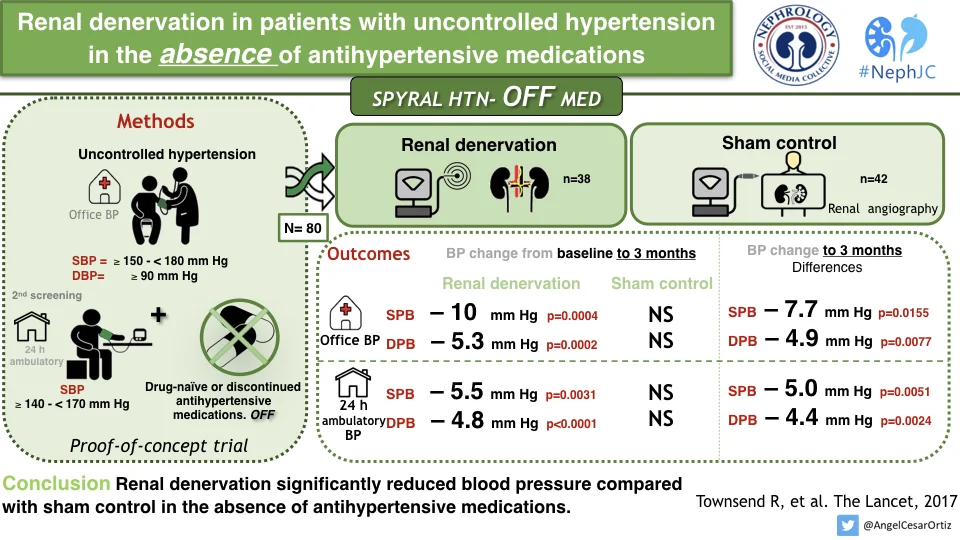
These principles - apart from newer technology - were applied in the three new trials: SPYRAL ON and OFF, and RADIANCE SOLO. Also, like S-3, they were all:
- randomized controlled trials
- control patients underwent sham procedure
- outcomes were based on Ambulatory Blood Pressure Monitoring (ABPM)
Why did these results differ from SYMPLICITY HTN - 3?
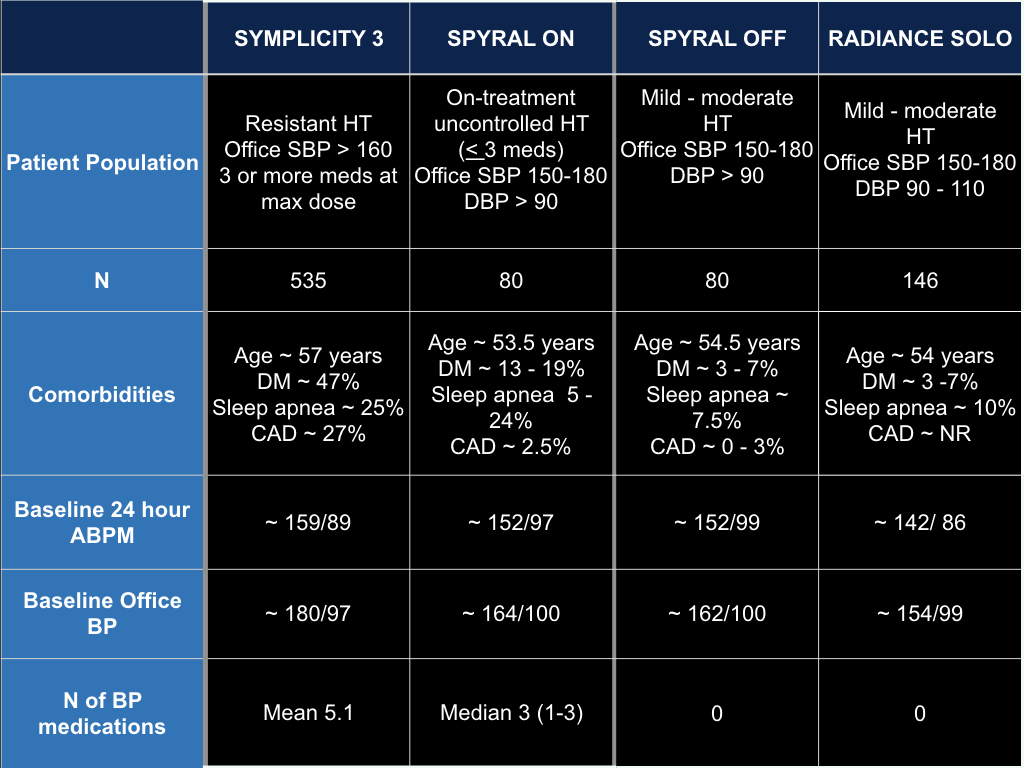
Despite the similarities mentioned above - many things were different between S-3 and the three new trials. Let's briefly look at the patient population:
So indeed, the S-3 population was a bit sicker - a bit older, with possibly a worse phenotype of hypertension and more comorbid conditions.
A few salient observations:
- note the different methods of denervation and effect size (bottom row)
- is the effect size indeed larger with longer follow up (among the three newer trials: greatest effect at the 6 month vs the 2 month follow up)?
- Does ultrasound denervation (main renal artery) have similar efficacy as the catheter ablation method (main + branch/accessory > 3 mm)?
- note the effect in control arm: greatest decrease in S-3 (? 'I-will-take-it-now bias') and also a small decrease in RADIANCE, which was less rigorous on adherence measures than SPYRAL
Do you believe these results, ie does renal denervation really reduce blood pressure compared to sham?
These trials are as meticulous as could be. Yes, they were all funded by the device manufacturers - but in the aftermath of S-3, no one else would still have been foolhardy enough to perform further trials. Are there other threats to internal validity?
What do we know about long term efficacy? From animal data, we have seen that re-innervation does occur, along with an increase in detectable sympathetic activity.
Are we convinced about the safety of Renal Denervation?
The three trials did not report any safety signals - and neither did S-3. However, it behooves us to think about some possible adverse events:
- Renal artery stenosis: cases have been reported. Will the more intensive ablation with the newer catheter and strategy of targeting branch/accessory arteries result in a higher likelihood of possible damage?
- Renal denervation is permanent. Blood pressure medications can be stopped. Why does this matter? Maybe if a patient subsequently develops sepsis or hypovolemia? The sympathetic system exists for a reason....See this case report. In this study of sheep with CKD, bleeding caused a much more severe drop in blood pressure.
- Lastly, blood pressure declines in the last few years of age - as can be seen from this interesting study. For patients post denervation, will this mean their blood pressure will trend even lower? Maybe lower blood pressure is not always a good thing.
Figure 1 from Delgado et al, JAMA Int Med 2017
Of course, one could say: many patients have had denervation so far. And we haven’t seen many adverse events from the global Symplicity Registry (see below, from a recent presentation at PCR, via Andrew Sharp). Yet.
So what should happen now? More trials?
A few larger trials are already planned:
- SPYRAL PIVOTAL: with a sample size of 433, geared towards FDA approval, in a SPYRAL OFF like setting (i.e. off medications)
- REQUIRE (resistant HT, in Japan, Korea) and RADIANCE TRIO (on 3 BP meds - fixed dose combo)
- Update (July 4 2018) Also RADIANCE-II (which seems similar to RADIANCE-HTN)
Questions to ponder:
- If all these trials show a significant effect on blood pressure, will that be sufficient to start recommending this therapy? Do you need to see long-term outcome data (for safety, as well as efficacy - remember reinnervation)? Update: Prof Francis shows in this tweetorial that a hard outcome trial will include 150,000 patients and cost $3 billion dollars.
- How about some comparative data: this small trial suggests spironolactone might be more efficacious than denervation.
- Which patients will be good candidates for renal denervation?
- Resistant hypertension (assuming adherence checked and confirmed)
- Non-adherent patients (considering the extreme pill dysutility - bordering on pill hatred - in some patients)
- Renal denervation potentially provides a smoother BP lowering, unlike the vagaries of troughs and peaks, heightened by the occasional missed dose, with pharmacotherapy - does this justify using renal denervation more widely?
To discuss these questions, and more - join us for the #NephJC chat.
Swapnil Hiremath, Nephrologist, Ottawa