In type 1 diabetes is finerenone helpful to reduce proteinuria?
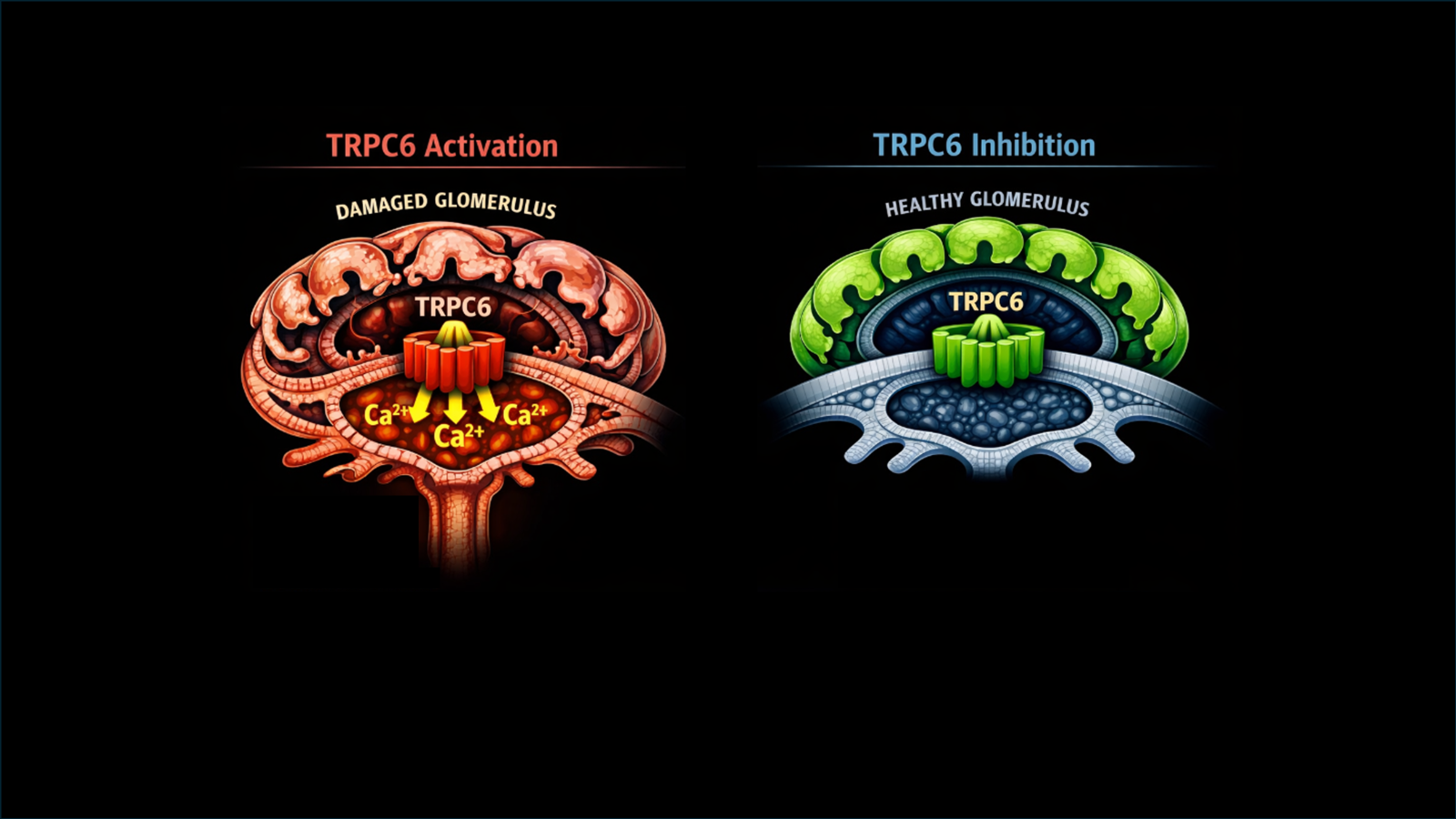
Is TRPC6 inhibition for FSGS a trick or a treat?
This week, we will discuss a phase 2 trial of the TRPC6 inhibitor BI 764198 in FSGS—an early signal for a podocyte-targeted therapy showing proteinuria reduction but set against small numbers, heterogeneity, and methodological trade-offs that frame this as direction-finding rather than definitive evidence.
Inhibidor de TRPC6: El Resumen Visual
Por primera vez, una terapia dirigida al podocito muestra eficacia clínica en GEFyS. Esta semana #NephJC revisa el ECA fase 2 de BI 764198, inhibidor oral de TRPC6, que redujo proteinuria, estabilizó el FGe y fue bien tolerado. ¿El inicio de una nueva era en GEFyS? Revisa el resumen visual por la Dra Divya Bajpai
TRPC6 inhibition: The Visual Abstract
For the first time, a podocyte-targeted therapy shows clinical efficacy in FSGS. This week #NephJC covers the phase 2 RCT of BI 764198, an oral TRPC6 inhibitor that lowered proteinuria, preserved eGFR, and was well tolerated. A new era in FSGS? Check out the visual abstract by Divya Bajpai
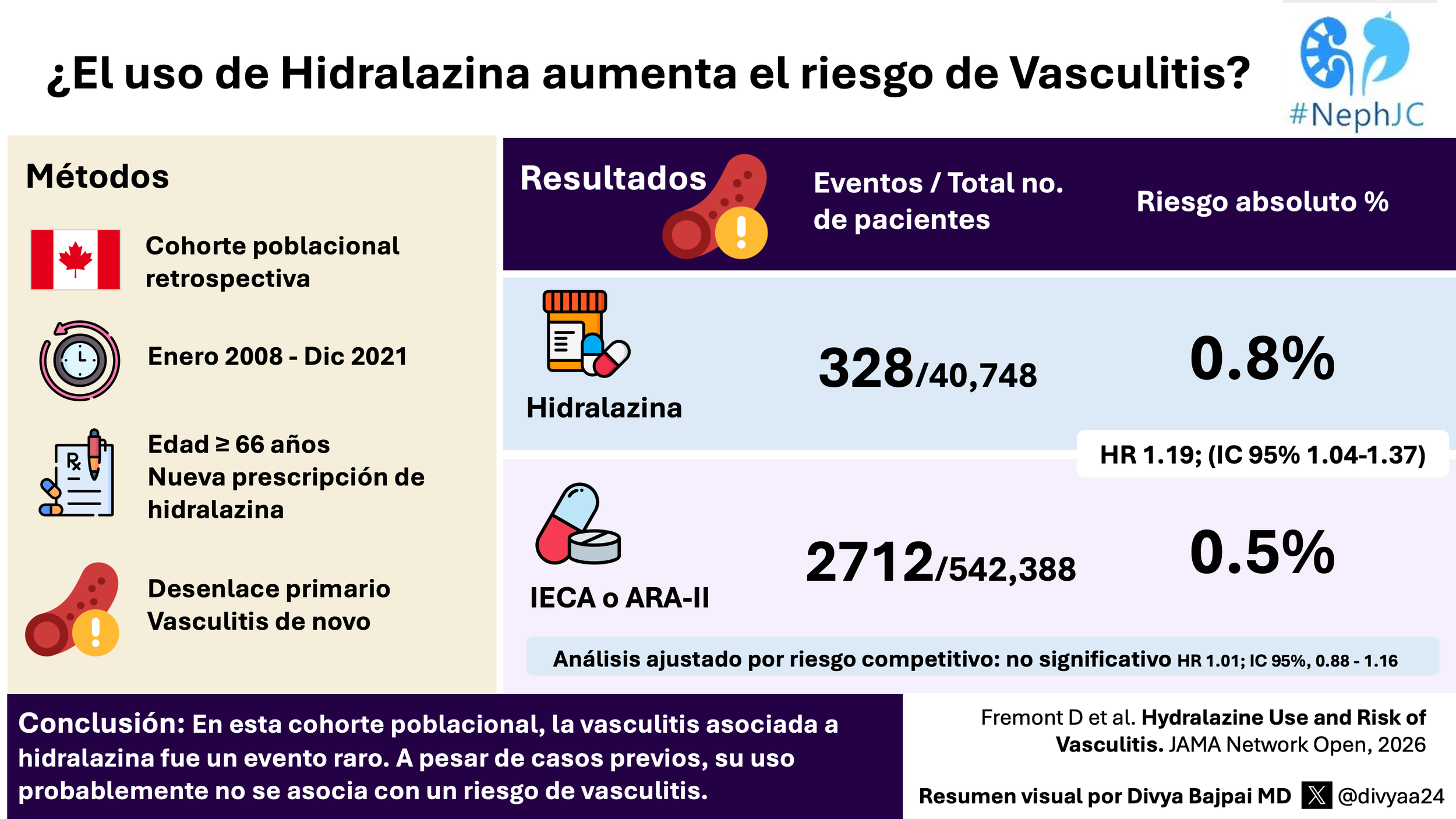
Why-dralazine?! Population risk of drug induced vasculitis
This week, we will discuss why a large registry cohort was needed to move past decades of scattered case reports and clarify the true risk of hydralazine‑associated vasculitis. When rare events hide in noise, only scale can reveal the signal. Can population‑level data finally bring this paradox into focus?
Can hydralazine trigger ANCA vasculitis?
A large registry cohort was essential to move beyond scattered case reports and finally quantify the true risk of hydralazine‑associated vasculitis. When rare events blur into background noise, only scale can clarify the signal. Can population‑level data sharpen the picture?
Check out Divya Bajpai VA
¿Puede la hidralazina desencadenar vasculitis ANCA? El resumen visual
Un gran registro poblacional era esencial para superar décadas de reportes aislados y cuantificar por fin el riesgo real de la vasculitis asociada a hidralazina. Cuando los eventos raros se confunden con el ruido de fondo, solo la escala permite distinguir la señal. ¿Puede la evidencia a nivel poblacional aclarar por fin este panorama?
Revisa el resumen visual por Divya Bajpai , traducir por Milagros Flores
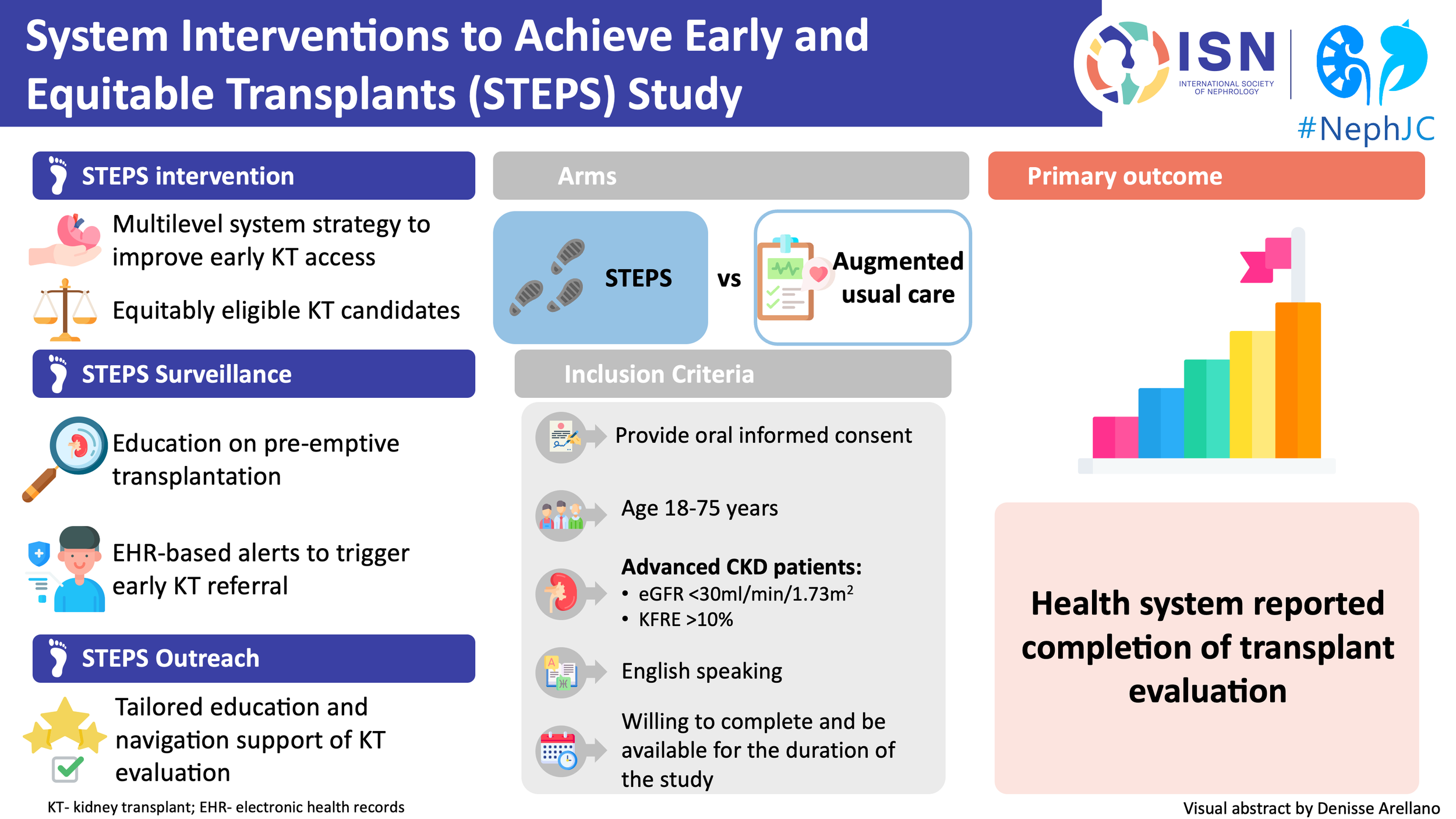
From missed opportunities to equitable access: Can STEPS redesign the kidney transplant pathway?
Regarding the Situation with…Social Media in General
Regarding the Situation with…Twitter, Bluesky, Social Media
We at #NephJC pride ourselves on bringing you the best in nephrology publications and innovations without commercial bias. As we’ve stated during many a donation drive, we are beholden to none but our followers and readers.
Having said that, we’ve noticed a trend since the death of Twitter (yes, it will always be Twitter to the real FOAMed illuminati). The diaspora of NephTwitter were scattered throughout the nephro-universe of social media, never quite finding a new home. Perhaps the downfall of the Nephrologians can be a plot line for the next Mandalorian season?? Many left the social media space altogether because of the division, politicization and plain BS of some of the algorithms (I for one don’t need to see EVERYTHING Elon posts). However, we still believe there is a need for our unique style of nephrology education. Jon Stewart- yes, the comedian- recently likened social media to smoking: an addictive, attention seeking habit that is generally recognized as not healthy for most people and that is likely to be regulated for teen and self-regulate by adults. He believed the antidote was more long-form, nuanced edu-tainment. So, it light of the changing landscape, we are going to focus more on our blogs, VAs, and podcasts and less on live discussions on Bluesky.
NephJC is nothing if not nimble, and we have never been afraid of change.
So, given the low attendance at live chats (no judgement) we’re changing to a summary thread (a new 10 posts) with provocative questions to answer, at your leisure. We hope this keeps the discussions going without the need to be on social media for prolonged periods or at a specific hour and day. These threads will be posted at the familiar 9pm EST every other Tuesday. This shorter form will also allow us to post on multiple sites, and hopefully we will be easily accessible to you where you chat with other nephrophiles. Feel free to reply, chat, tag people in conversations whenever and wherever you see a #NephJC post. The team will be around, but we won’t be actively ‘moderating’ a one hour chat.
Sincerely,
NephJC Leadership
PS: As always we love to hear from our audience. If you have suggestions, or a better way to reach more people, we are all ears. Heck, if we hear enough complaints we might even bring back the chats!😉
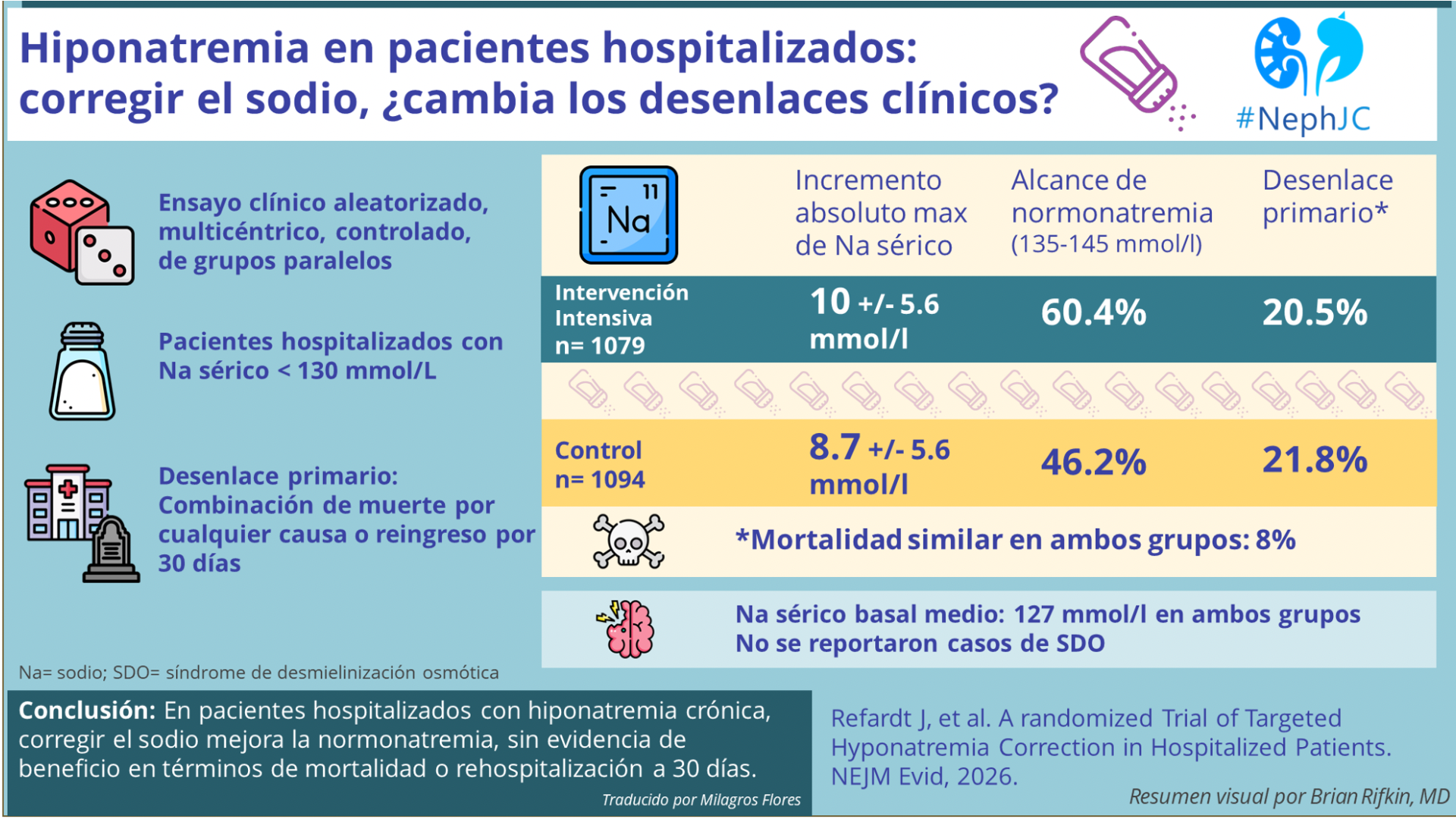
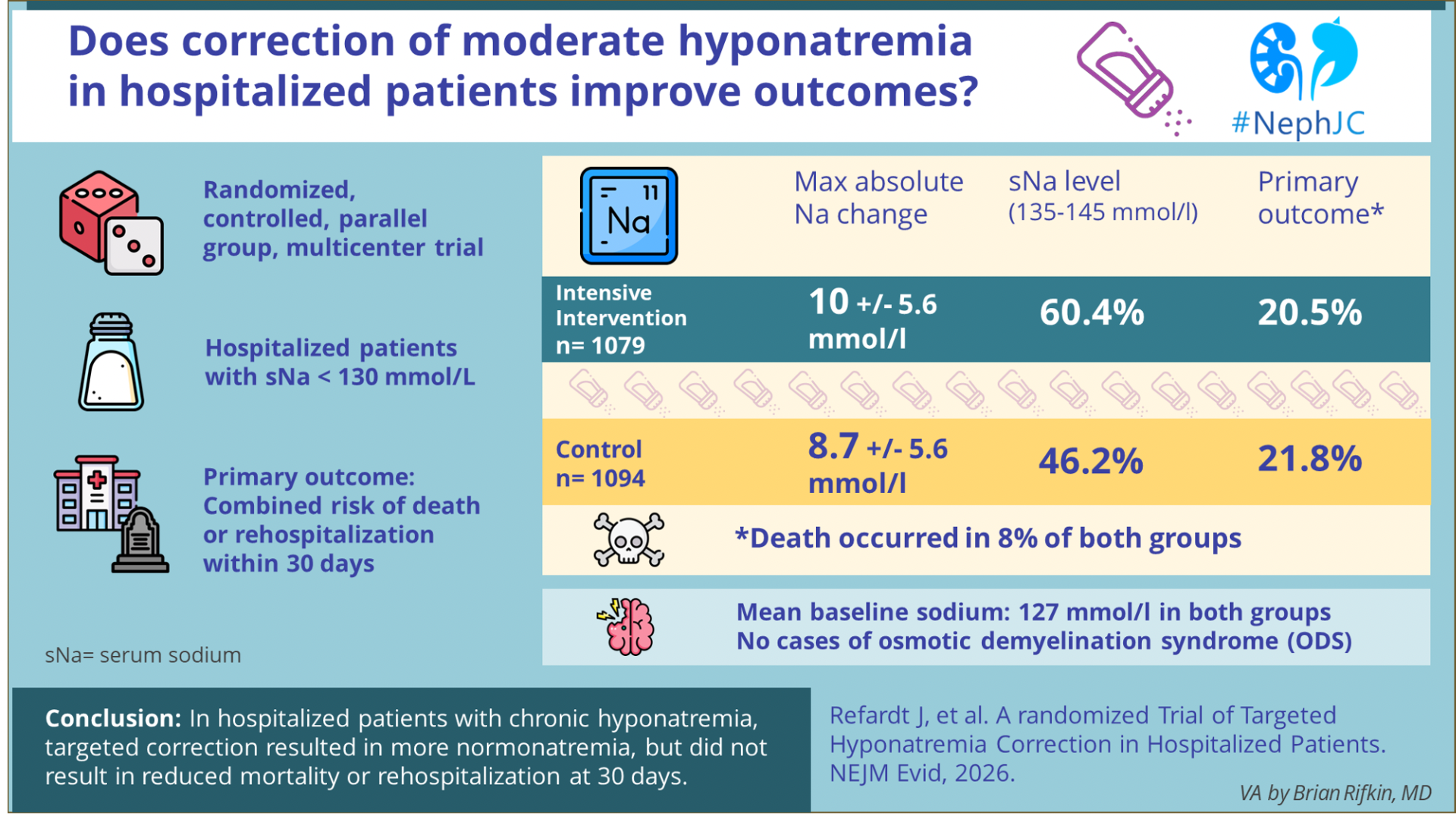
Searchin’ For My Lost Shaker of Salt- Targeted Treatment of Hyponatremia
Live Blogging Trials from Conferences
Hiponatremia: El Resumen Visual
La hiponatremia es uno de los trastornos electrolíticos más frecuentes en pacientes hospitalizados y se asocia de manera consistente con peores desenlaces. Su corrección mejora los valores, pero ¿realmente mejora los desenlaces clínicos ?
Revisa el resumen visual por Brian Rifkin y el nuevo formato de #NephJC
Hyponatremia Correction: The Visual Abstract
Hyponatremia is one of the most common electrolyte disorders in hospitalized patients and is consistently linked to worse outcomes. Correcting sodium improves the numbers—but does it improve short-term clinical outcomes?
Check out Brian Rifkin VA and the new #NephJC format
NephJC Short: The Push Trial
NephJC Covers LBCTs at the World Congress
Announcing the NephJC Editorial Internship
Are you a curious, motivated nephrology fellow or early‑career faculty member with a passion for science communication? NephJC is inviting a small group of talented individuals to join our Editorial Internship Program and become part of the team that shapes how nephrology is discussed, illustrated, and shared online.
As an intern, you’ll help craft the bimonthly NephJC Journal Club, one of the most vibrant hubs for post‑publication peer review in nephrology, while developing your skills as an editor, writer, illustrator, or podcaster. We’re also planning to expand our Instagram presence this year, so creative voices with an eye for visual storytelling will have new opportunities to shine.
What this internship offers
Collaboration with an experienced, dynamic production team
Engagement with cutting‑edge nephrology research
Increased professional visibility within the global nephrology community
Take your passion for kidneys, curiosity, and community to the next level- join the NephJC Editorial Internship Program and help us keep the nephrology spirit alive across platforms, including a more active IG presence.
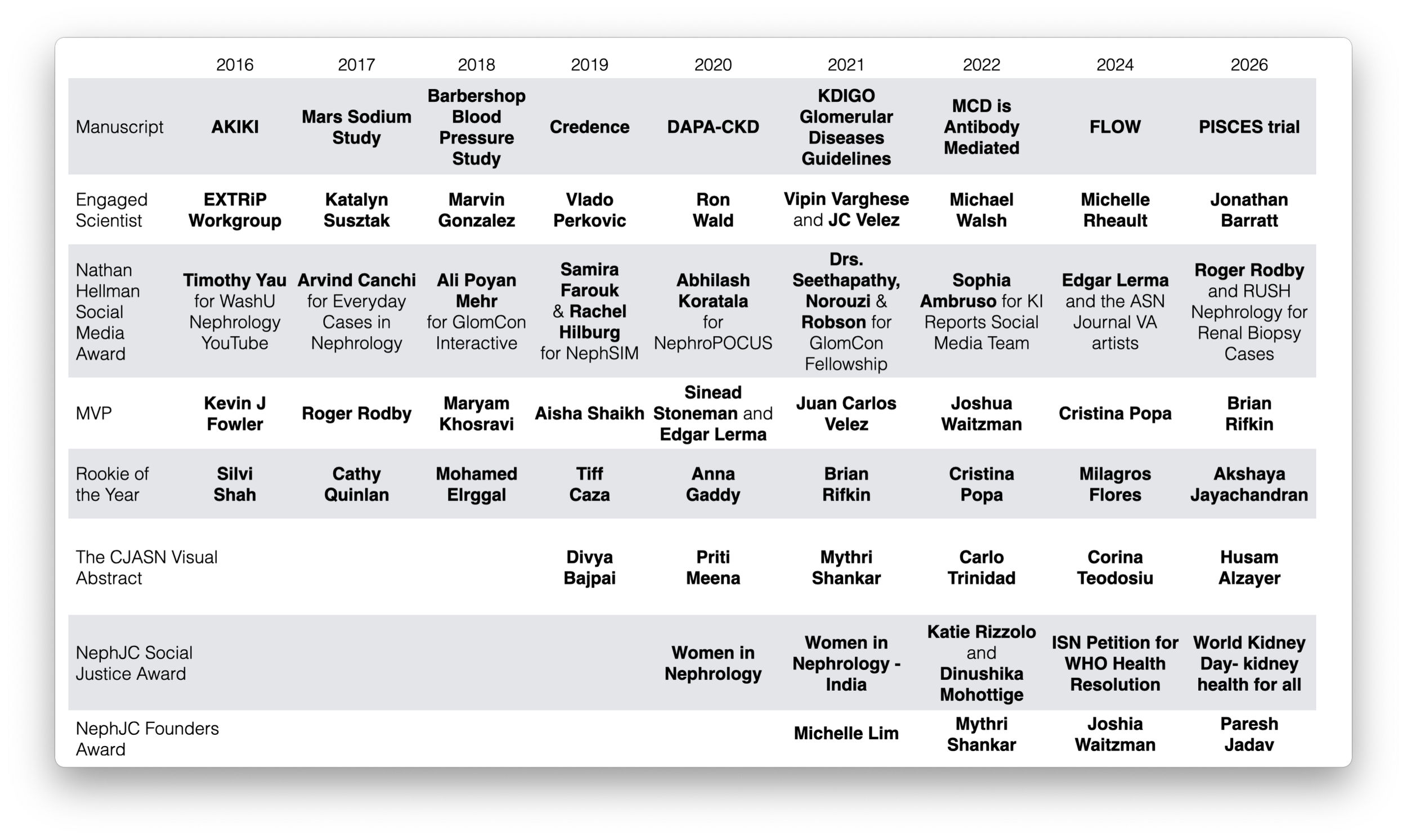
The 2026 NephJC Kidneys Award Winners
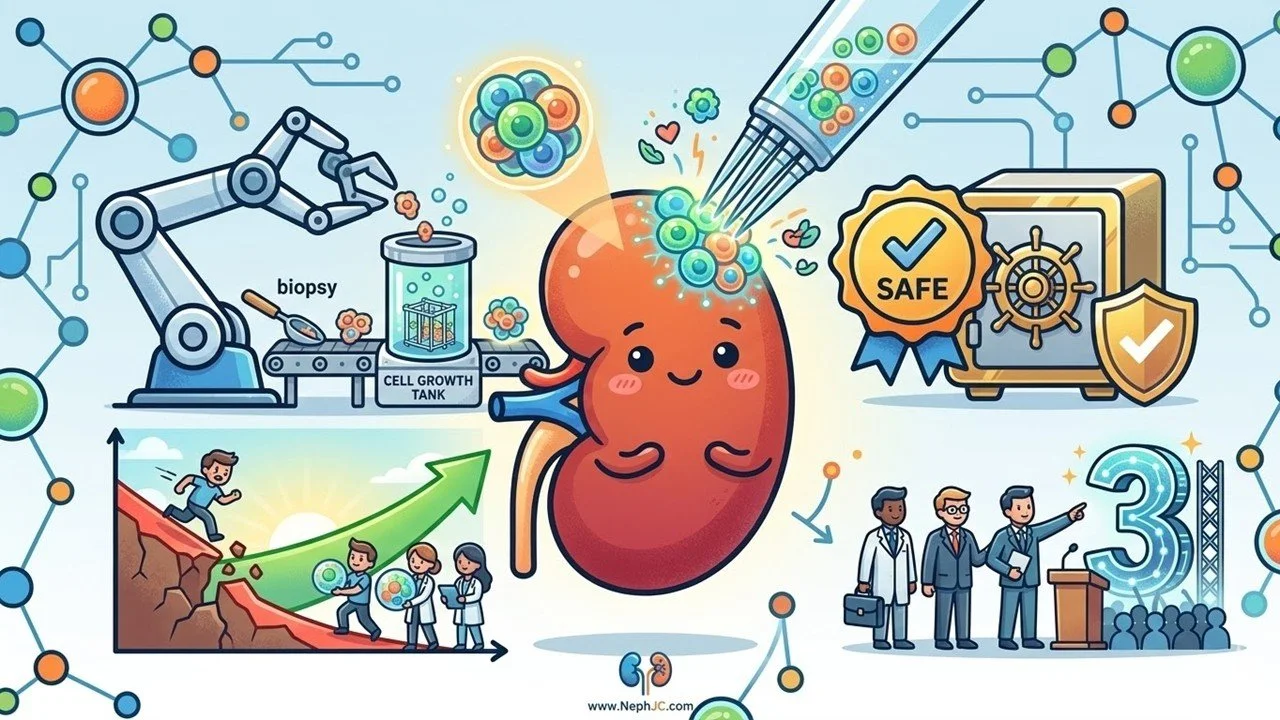
TenPosts Rilparencel phase 2
Can we move beyond slowing CKD to actually repairing the kidney? Here is the summary of the last #NephJC chat discussing Rilparencel, a renal autologous cell therapy (REACT) infusion, and its effect on eGFR slope.
Thread by Akshaya Jayachandran