2025 was rich in successful stories. Here are the top 10 that impressed our audience.
Top Stories in Nephrology 2024
Should we call 2024 the Renaissance of nephrology? It was probably the richest year in RCTs in the nephrology world, reflected in the higher number of Late-Breaking Clinical Trials sessions at every big nephrology congress. Probably 1st place won’t surprise anyone; it was the anticipated FLOW of the year, but this Top 10 Nephrology Stories definitely includes some unexpected titles
Top Stories in Nephrology 2023
2023 brought us has dogma shredding data on hyponatremia and the importance of different diuretics. It has new therapies for old diseases like IgA and hypertension. New drugs for new diseases like inaxaplin for AMKD. And it has new data on old debates like what IVF is best and do thiazides really prevent kidney stones. It is a great list. Dig in!
The Top Nephrology Stories of 2022
SPYRAL ON: Renal Denervation in Patients ON Medications
SPYRAL OFF: Can Renal Denervation Reduce Blood Pressure in Hypertension?
Renal Denervation: the Story so far...
The Sympathetic System and Blood Pressure
We know that the sympathetic nervous system plays a role in blood pressure regulation. A long long time ago, when we did not have many - or any - blood pressure lowering medications - especially safe ones, options considered were colloidal sulphur and sodium thiocyanate.
In the early years of hypertension, some thought that elevated blood pressure was a homeostatic response to decreased tissue perfusion. In renal failure, high blood pressure was supposed to be required to excrete larger amounts of urea despite fewer nephrons. See Ludwig Traube in 1856.
One of the few options to lower really high blood pressure was to perform a dorsolumbar sympathetectomy, first hypothesized by Brüning in 1923. After a few unsuccessful attempts, and some on serendipitious observations of the effects of spinal anesthesia, Adson did a bilateral ventral rhizotomy, from the T6 to L2 level in a young woman. Her blood pressure fell from 250/180 to 170/120 mm Hg. A subsequent case series, showed consistently lower blood pressure - truly & spectacularly so. The amazing improvement in blood pressure was accompanied by just-as-impressive side effects. Apart from surgical morbidity and occasional mortality, there was loss of sensation, paralytic ileus, problems with ejaculation, and loss of sweating. And then, a few years later, blood pressure lowering medications started becoming available, such as reserpine in 1949, and in the mid 60's Sir James Black developed propranolol. With the development of calcium channel blockers in the 1970s and captopril in the 1980s there was no looking back. Now A-C-D are established as first line therapy on the basis of safe and effective blood pressure lowering with beneficial effect on cardiovascular outcomes. The hot hypertension debates have moved on to what level of blood pressure lowering (120 or 130 or 140, not 250!) and not on use of surgical procedures and whether blood pressure should be lowered at all.
Renal Denervation Development
However, a minority of patients with hypertension have blood pressure that does not respond to first line agents (typically the A-C-D combination) mentioned above. Though second line agents are aplenty, they have higher rates of adverse events, as well as little data on long-term, hard-outcomes. Hence this area is still considered ripe for testing newer interventions, such as new drugs and novel strategies. One of the most promising entrants here was the development of percutaneous renal denervation, first reported in 2009, from Murray Esler, Markus Schlaich and colleagues from the Baker Institute in Melbourne. The patient they reported had severe resistant hypertension with end organ damage, underwent percutaneous radiofrequency ablation of the sympathetic nerves around both of the renal arteries, resulting in a decrease in kidney, as well as whole body norepinephrine spillover and muscle sympathetic activity. This was accompanied by an impressive 20/17 mm Hg drop in blood pressure - not as much as with dorsolumbar surgical sympathectomy, but impressive nevertheless given that this was on a background of multiple medications.
From Schlaich et al, NEJM 2009: the first case report in the modern era of RDN
The group quickly followed up with two large prospective trials. Symplicity HTN-1 was a single arm prospective trial, which enrolled 50 patients with resistant hypertension, defined as an office systolic BP of 160 or more on 3 or more drugs. It demonstrated a drop of 14/10 mm Hg at one month, which seemed to progressively increase to 27/17 by 6 months of follow up.
From Krum et al, Lancet 2009: Main result of Symplicity HTN-2
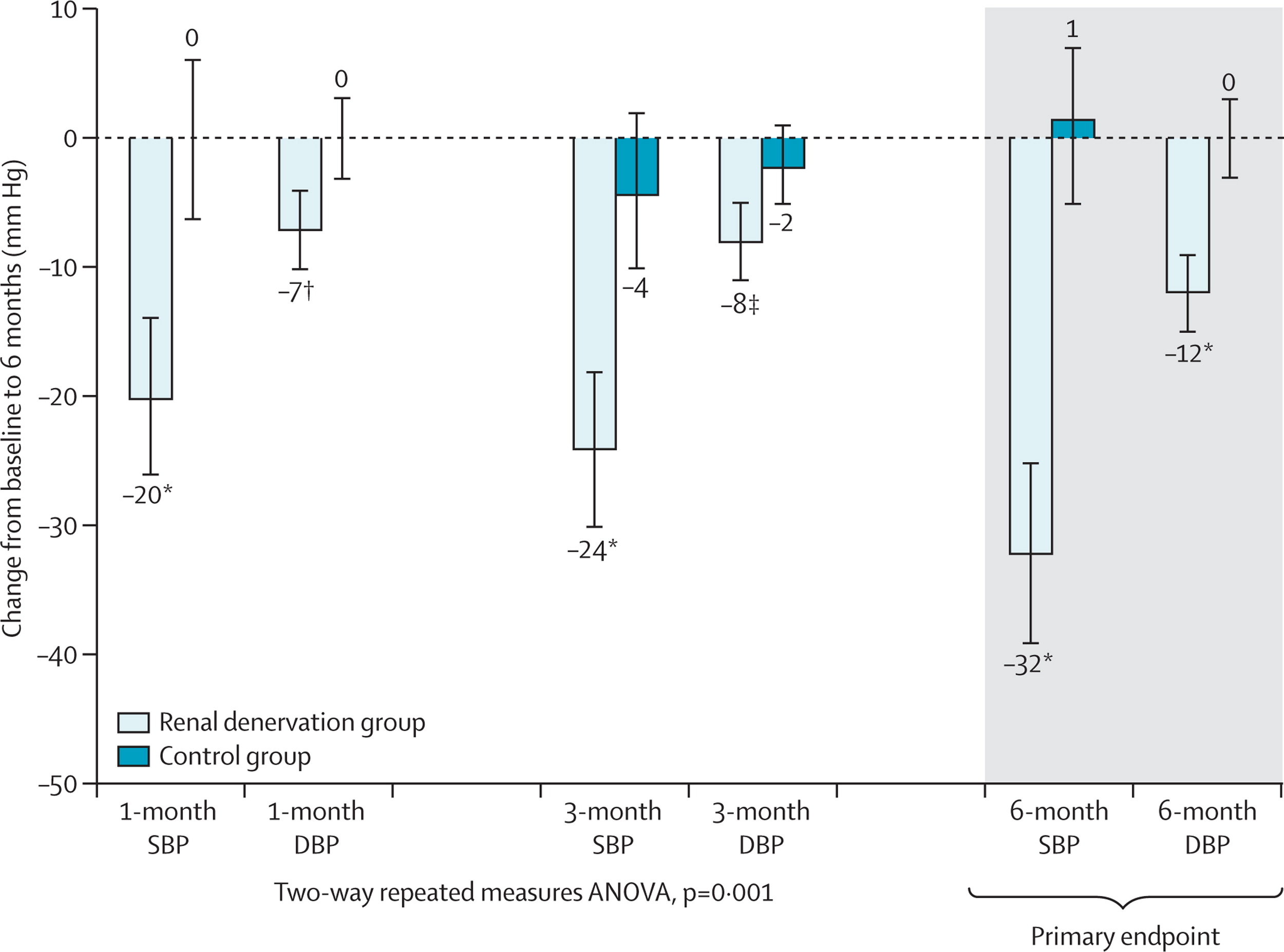
This proof-of-concept trial was followed by the Symplicity HTN-2, this was an open-label, randomized controlled trial in about 100 patients with resistant hypertension. The results showed a decrease in BP of 20/7 at 1 month and 32/12 at 6 months. Renal denervation (RDN) had arrived! CE approval in Europe followed soon after and rates of renal denervation soared. The company that developed RDN (Ardian) was bought by the device behemoth Medtronic for $800 million. Other device companies with different catheters entered the fray. Hypertension was cool again. Until Symplicity HTN-3.
From Esler et al, Lancet 2010: Main results of Symplicity HTN-2
The Crash
Despite CE approval in Europe, RDN was not approved in the US. The FDA insisted on a rigorous trial undertaken in the US. This was Symplicity HTN-3 (S-3). The catheter and technique were the same ones, now from Medtronic. But the trial was different in many salient ways. S-1 and S-2 included patients with an office BP > 160 mm Hg, and the outcomes were office based blood pressure changes. In S-3, the trialists not only required office BP > 160 on 3 or more drugs at maximal dosage, but also 2 weeks of home BP measurements followed by a repeat office visit to measure BP. They all also needed 24-hour ambulatory blood pressure monitoring (ABPM, see #NephMadness post on why this is important), showing daytime systolic BP > 135. This requirement resulted in the trialists enrolling 535 from 1441 potentially eligible patients. Most importantly perhaps, S-3 had a control arm in which the patient underwent renal angiography, with a ‘sham’ RDN procedure. Thus patients were blinded - and so were the trial personnel measuring blood pressure. The results of S-3 came as a shock to the hypertension community. They demonstrated no difference between RDN and control (ie sham RDN) in either office or ABPM. Many explanations were bandied about (see the emergency #NephJC session details and hangout video for more). But RDN was dead, as far as North America and many other places in the world were concerned.
From Bhatt et al, NEJM 2014, Main result of Symplicity HTN-3
Why did S-3 differ from previous RDN trials?
In the immediate aftermath of S-3, some of the key reasons that were thrown around were: heterogeneity of the population (there was a suggestion of less effect in the African-American population versus others) and the completeness of the RDN (the number of ‘notches’ corresponded loosely with the BP lowering). Medtronic developed a newer catheter system (‘Spyral’) which could do radioablation at different points both longitudinally and circumferentially, thus reducing the issue of operator expertise.
Design of the newer SPYRAL catheter system
With respect to the efficacy of actual nerve ablation, it is indeed important to note that there is, yet, no physiological test or biomarker to assess whether the RDN was successful. The norepinephrine spillover or muscle sympathetic nerve activity testing are accurate, but time consuming and only used as research tools. Blood pressure lowering is a physiological test, but it is very noisy.
On the noisiness of blood pressure, one of the fascinating analysis that came from S-3 was from Darrel Francis’s group (yes the same Professor of ‘tweetorial’ fame). A few months before S-3, they predicted that the effect in a sham-controlled trial would be less than in previous trials. S-3, of course, didn’t show just a smaller effect, it reported no effect of RDN compared to sham. So Francis and his smart fellows (James Howard) conducted a further quantification of the three biases that they measured as being important to understand in this area.
Regression to the mean: First described by Francis Galton (‘regression to mediocrity’) this is an important aspect of a messy measurement like blood pressure. Selecting individuals with very high values and repeating another measure (as was done in the early RDN studies) is almost guaranteed to provide lower values - whether patients undergo RDN or not. Repeated measures, to confirm that the elevated blood pressure is truly elevated (say, with 2 weeks of home BP, or with ABPM), help reduce this - and a control group almost solves this. However, in the RDN studies, there was not much of a difference between controlled and uncontrolled studies - and even with unblinded controls who were randomized.
The second important bias then is information bias: in this case measurement bias. Observers who were unblinded taking BP measurements can be, ahem, biased, if they know whether the patient had or did not have RDN. Hence the use of automated measures (such as ABPM) reduces the effect size.
Lastly, blinding - which was a key aspect that was different between the two RCTs (S-2 and S-3). How would this have a different effect compared to unblinded controls? After all, BP - especially with ABPM, is an objective measure? However, knowing that one has had RDN may change behaviour - maybe patients become more adherent to their medication. Perhaps even the elusive placebo effect does truly exist in hypertension.
From Howard et al, Circulation 2016. Relationship between trial design and the reductions in office and ambulatory blood pressures. Each data point represents a trial. The area of the data point is proportional to the trial size. Red diamonds indicate the meta-analyzed estimate of the effect size for each trial design
Apart from these three aspects, it was also noted - from the same group - that variance in BP trials was higher when greater number of blood pressure medications were prescribed.
From Howard et al, J Hypertension 2015. Visit-to-visit variance in blood pressure within individuals increases with the number of medications prescribed. Sample size required for a trial is linearly dependent on visit-to-visit variance, the square of the visit-to-visit SDΔ, because the sample size required for a trial is linearly dependent on this.
Among the recommendations provided to overcome this problem were:
Solution 1: conduct renal denervation trials in patients on no medications (note this is exactly opposite of what was being done so far: testing RDN in patients with resistant hypertension)
Solution 2: continue prior medications, but pause medication for a limited period prior to baseline and final pressure measurements
Solution 3: directly observed antihypertensive therapy with a stable regimen
Based on this, in late 2014, a think tank comprised of representatives from the FDA, along with the NHLBI and the American Society of Hypertension came together and published some guidance for device therapy trials in hypertension, which was followed closely in the three trials we will be discussing at #NephJC this week.
Read so far? Now please go ahead and check out summaries of
Also check out the excellent Visual Abstracts from Angel Ortiz
Lastly, to prepare for the chat, see the summary of all three trials - and some discussion points.
Post by Swapnil Hiremath, Ottawa
Is 130 the new 140? The 2017 AHA/ACC Hypertension guidelines
Top nephrology-related stories of 2017
2017 marks the 8th year of the Top Nephrology Stories post on RFN. Below are links to the last 7 years of the top nephrology stories hosted on RFN.
2017 was a great year in nephrology. Several trials in GN (IgA/Membranous), new hypertension guidelines, multiple studies looking into basic mechanisms of APOL1, sodium homeostasis paradigm shift, and a ton more that didn't make it into the Top 10. 2017 also marks the first year the Top 10 post is published on NephJC. Just like the collaborative nature of NephJC this years Top 10 was written by several members of the NephJC team. No doubt collaboration is key to success and the further maturing of #NephTwitter is a prime example of that. Can't wait to see what 2018 has in store. Here are the winners of this years democratic voting... (%- percent of votes for each study picked for Top Story)
#Infographic
10. Russian Cosmonaut Salt Studies in JCI (12% Tie)-
This study had it all: hermetically sealed Russian cosmonauts, a link to the red planet, big name authors, and a New York Times article telling us that “Everything We Know about Salt May Be Wrong”. So what was it? The authors recognized the unique opportunity to study long term sodium balance in man afforded by two simulated space flights to Mars, one for 105 days and one for 520. What they produced was a 12 page article and 1131 page data supplement that both reinforced some long held concepts of Na/H2O balance and raised a few new ones too. In terms of reinforcement of “known knowns”, the study showed that with a higher dietary salt intake urine concentration rises and so does free water generation. In addition, our ideas about the effects of aldosterone on sodium excretion were well backed up by this data. The novel concepts raised here were truly novel: whilst the kidney plays the main role in coupling water and salt in the long term, sodium excretion may follow a cyclical pattern, and thirst and water ingestion do not seem to be involved in coupling water to salt. Unsurprisingly, this was one of the most chewed over studies of the year. So, this Holiday season, you can either revisit the discussions, opinions and summaries to work your grey matter, or just sit back and watch Matt Damon’s rhythmic control of osmolyte and water.
#VisualAbstract
10. TESTING Trial- Steroids in IgA Nephropathy in JAMA (12% Tie)-
For whatever reason several clinical trials in IgA have hit the presses in the last several years. In 2015, Stop IgA Trial made it all the way to number 3. This year brings another randomized clinical trial looking at steroids in IgA Nephropathy. The TESTING trial was a multicenter, double-blind, placebo-controlled, randomized trial which tested the effect of oral methylprednisolone and clinical outcomes in patients with IgA Nephropathy. IgA Nephropathy is the most common glomerulopathy in the world with highly variable clinical presentations ranging from asymptomatic hematuria, different degrees of proteinuria and glomerulonephritis which could progress to end-stage renal disease. In this trial, 262 patients underwent randomization; 136 to methylprednisolone and 126 to the placebo group. After a median follow up of 2.1 years, the primary outcome (ESKD) occurred in 8 patients in the methylprednisolone group and 20 patients in the placebo group (5.9% vs 15.9%, p = 0.02). Recruitment was discontinued due to serious adverse events (mostly infections) which occurred in 20 patients in the methylprednisolone group and 4 patients in the placebo group. Furthermore, the annual rate of eGFR decline was -1.79 mL/min/1.73/min/1.73 m2 in the methylprednisolone group and -6.95 mL/min/1.73 m2 in the placebo group (p+0.03). The authors concluded that the use of methylprednisolone in patients with IgA nephropathy may have a benefit, however, it is associated with a high rate of adverse events. Important to mention that another trial, STOP IgA showed that the addition of immunosuppression to ongoing comprehensive supportive care, does not add any more benefit. Read the editorial of TESTING trial here. There is an ongoing trial which will evaluate the safety and efficacy of low dose oral methylprednisolone in comparison to placebo.
#VisualAbstract
8. APOL1 Mouse Model Leads to Podocyte Damage and Kidney Disease in Nature Medicine (13%)-
In March NephJC revisited a NephMadness champion, APOL1. Risk variants in APOL1 are common in people of African descent, and there is evidence that trypanosomiasis acted as a selection pressure favoring these risk alleles. This Nature Medicine paper explored the mechanisms of podocytopathy underlying development of chronic kidney disease with APOL1 risk variants. The authors, led by @KSusztak showed that APOL1 risk variant related kidney disease is podocyte specific and relates mechanistically to podocyte autophagy and pyroptotic cell death.
A really neat transgenic mouse model allowed them to tease apart the mechanisms of disease.
This study really thrust the APOL1 field forward, tackled some challenging experiments, and builds a story really nicely from the landmark paper describing the association of APOL1 variants and kidney disease in 2010.
There are plenty of outstanding questions that we explored in the NephJC chat: why is the podocyte so susceptible? Why is HIV an additive risk? How can we target APOL1 podocytopathy?
#VisualAbstract
7. MENTOR Trial Comparing Rituximab versus Cyclosporine in Membranous Nephropathy presented at ASN (14%)-
Coming in at number 7 is the MENTOR Trial which was presented at ASN Kidney Week in New Orleans. Here is the Twitter coverage collated on AJKD blog. All nephrologists know that the evidence base informing treatment of primary nephrotic syndrome is very weak. MENTOR partly rectifies this in a multicenter RCT of Rituximab versus Cyclosporine in biopsy-proven Membranous Nephropathy, the commonest cause of primary nephrotic syndrome in adults.
130 patients were randomized to Rituximab or Ciclosporin for 12 months treatment. Complete or partial remission after 24 months occurred in 62.5% of the Rituximab cohort versus 20.6% of the Cyclosporine cohort (95% CI 2.7-13.2). Patients receiving Cyclosporine had a higher risk of treatment failure at 24 months compared with Rituximab (79.4% versus 37.5%, 95% CI 24.7-55.9). Rituximab took longer to achieve remission than Ciclosporine, but Rituximab exhibited fewer adverse events.
Although this study did not compare Rituximab with the Alkylating agents still recommended by KDIGO, and excluded those with eGFR <40 ml/min, it is important because it highlights the relative safety and non-inferiority of Rituximab over Cyclosporine in Membranous Nephropathy. Only when we have further similar studies will we be able to offer our nephrotic patients proper evidence-based, rather than opinion-based, care.
6. Transplanting Hepatitis C Infected Kidneys into Non-Hepatitis C Recipients (17%)-
Coming in at number 6 is something that could not have been imagined just 5 years ago. One of the greatest stories of 2017 was the use of hepatitis C virus (HCV) infected kidneys in kidney transplantation. Despite critical shortage of organs and longer waiting times, about 500 HCV kidneys are discarded annually. Two bold studies conducted separately by the University of Pennsylvania (Thinker) and Johns Hopkins University (Expander-1) pushed the envelope to utilize these kidneys. These studies tested the safety and efficacy of the transplantation of HCV-infected kidneys into uninfected recipients, followed by antiviral treatment was examined. You heard that right. Findings from Thinker were presented in annual meeting of American Society of Transplantation in 2017 and the results published in a correspondence in NEJM. This was also covered in the AJKD blog post.
The following were the key highlights of the two trials:
- Performed at two separate centers in United States.
- A total of 20 patients received HCV-infected kidneys, with 10 patients enrolled in each center.
- All patients were cured of HCV following treatment with direct-acting antiviral agents.
The THINKER Trial included 10 adult participants between 40 and 65 years old, that had been on the transplant waiting list for fat least 18 months. The average wait time to receive a kidney was 58 days after enrolling in the trial. All 10 patients tested for HCV as soon as 3 days Following the transplant, and received 12-week course of directly acting antiviral elbasvir/grazoprevir (Zepatier) and were cured of the HCV infection. Even though 1 patients had delayed graft function and 1 patient developed FSGS post transplant, median 6-month serum creatinine was 1.1 mg/dL.
This is one of the revolutionary trials in the history of transplantation and gives hope to many patients who have been waiting for long times to receive a kidney transplant. One of the barriers anticipated is the high cost of anti-HCV medications. The drug used in the Thinker trial, Zepatier costs US $55,000, and the other anti HCV medication, Harvoni, costs US $94,000. As the trials are currently being expanded to enroll more patients, it would be interesting to see the long term graft and patients outcomes following transplantation of HCV infected kidneys into HCV-negative recipients along with a cure of HCV infection.
5. AHA/ACC Hypertension Guidelines presented at AHA published in Hypertension (18%)-
The American College of Cardiology and the American Heart Association (ACC/AHA) released the new hypertension guidelines in November. These now replace the JNC (Joint National Committee) guidelines, the last version which were published in 2014 (the JNC-8). JNC-8 was published after the NIH had already announced that they were getting out of the guideline business, and were dogged by controversy, for the delay as well as a minority report that was published disagreeing with the main report. And of course, the SPRINT trial was published a year later, which has changed much about what we know about hypertension targets. The 2017 ACC/AHA incorporate data from SPRINT, and much more, backed by a comprehensive literature review which was released concomitant with the actual guidelines. The one big message is 130/80 as the new target blood pressure. For everyone. It seems to be a very pragmatic decision, eschewing the actual 120 target from SPRINT, but also expanding the 130 target for patient populations not included in SPRINT. As with any guideline, there has been a lot of angst and disagreements - so look forward to a NephJC discussion in the new year. All the guidelines are easily available at guideline hub, with a simpler summary also available here.
4. REPRISE Trial of Tolvaptan in Later Stage ADPKD in NEJM (21%)-
This year at Kidney Week 2017 as a part of the Late-Breaking and High-Impact Trial Session Vicente Torres presented the REPRISE Trial and later that week it was published in NEJM and covered by NephJC. In 2012, the TEMPO 3:4 trial showed a reduction in growth of the total kidney volume and as a secondary outcome showed decreased progression of CKD. As a side note this trial made it to #1 in our “Top Nephrology Stories of 2012”. Tolvaptan was approved for its use in Europe but in America the FDA requested further safety data and wasn’t approved. As you can imagine there was a some disappointment among the nephrology community.
Based on this, the investigators conducted the REPRISE Trial. In the REPRISE Trial they enrolled patients with more severe kidney disease than in TEMPO (GFR 25-65). The primary end-point was change in creatinine-derived eGFR.
The results:
- Loss of eGFR was reduced by tolvaptan from 3.61 ml/min/year to 2.34 ml/min/year (p<0.001). This is 1.27 ml/min/year reduction compared to placebo.
- Rises in the hepatic enzyme Alanine Aminotransferase occurred in 5.6% of Tolvaptan treated patients versus 1.2% of placebo treated patients
These findings will be presented to the FDA in 2018. Will the US have a new drug to treat ADPDK? We will find out soon.
#VisualAbstract
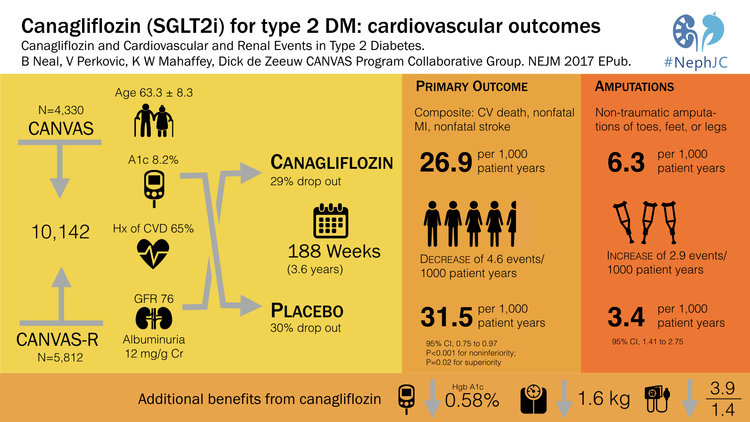
3. CANVAS Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes in NEJM (23%)-
EMPA-Reg was a landmark study that showed that the SGLT2-inhibitor, empagliflozin reduced mortality in type 2 diabetes. This was number 2 in the Top Nephrology Stories of 2015 and then returned in 2016 with number 4 when the FDA gave empagliflozin a license for reducing mortality in type 2 DM. However, a lot of people were on egg shells worried that the empagliflozin-story was just too good to be true. 2017 delivered CANVAS which showed that canagliflozin, another SGLT2i, reduced the composite endpoint of cardiovascular mortality, non-fatal stroke and non-fatal MI. CANVAS validated the findings of EMPA-Reg and solidified SGLT2i as a major breakthrough in the treatment of diabetes. The CANVAS trial was discussed in NephJC and was the subject of grand rounds by Christos Argyropoulos and Joel Topf. CANVAS validated the cardiovascular outcomes of EMPA-REG but also brought a lump of coal. The risk of lower limb amputations with canagliflozin was twice that of patients in the placebo group. The reason for this is unclear and as of now this signal has not been seen with other SGLT2i, but this finding will certainly be on minds of physicians and data safety monitoring boards around the world.
#VisualAbstract
2. PRESERVE Trial of Sodium Bicarb and Acetylcysteine in Preventing Contrast Nephropathy in NEJM (26%)-
This was the long awaited medical reversal, almost two decades in coming. In 2000, the first trial which reported the benefit of acetylcysteine in reducing contrast induced acute kidney injury (CI-AKI) was published. Despite doubts and uncertainties about its mechanism as well as purported protective effect, its use became widespread over the next few years - after all, there were little downsides. In the last few years, more negative trials were published, most notably the ACT trial in 2011, but being in lower risk patients, it was not definitive enough (the KDIGO guidelines still recommended its use). Similarly, in 2004 a trial reported the protection conferred by a bicarbonate based intravenous volume expansion around the time of the contrast exposure. This upstart replaced the previous accepted standard of normal saline. Similar uncertainties about any real difference between bicarb and saline followed. Now in one stroke, the PRESERVE (prevention of serious adverse events following angiography) trial took away both these purported protective pharmacotherapies. Randomizing just over 5000 patients, in this trial funded by the Veterans Affairs, there was absolutely no difference between acetylcysteine and placebo, or bicarb vs plain old saline. Indeed, the trial was stopped early for futility earlier given the absolute lack of difference noted between acetylcysteine and placebo. Read the discussion from NephJC here.
#VisualAbstract
1. SuPAR and APOL1 in Nature Medicine (50%)-
SuPAR is #1, the suPAR enthusiasts come out in force with the heavy voting and it is not a surprise as suPAR has been a consistent story for the last eight years. Last year, it hit the number 1 spot with a basic science paper published in Nature Medicine. In 2011, suPAR was ranked #1 TopNephrology stories with the original Nature Medicine paper describing its potential link to FSGS. In 2015 suPAR landed at number 14 with the NEJM paper linking suPAR to CKD in a cohort of patients at Emory undergoing cardiac catheterization. The current study looked at the association between suPAR, Apolipoprotein L1 (APOL1) risk variants, and eGFR decline in two separate cohorts of individuals of African ancestry. It was found that plasma suPAR levels independently predict renal function decline in individuals with two copies of APOL1 risk variants. They also explored the possibility of a protein–protein interaction between suPAR, APOL1 and αvβ3 integrin and showed that the G1 and G2 risk variants of APOL1, but not the reference protein G0, synergize with suPAR in the activation of αvβ3 integrin on podocyte. This binding causes change in structure and function, allowing disease onset. Using cell models and genetically engineered mice, the authors reproduced kidney disease changes upon expression of APOL1 gene variants, but the disease required the presence of (high level) suPAR.
African Americans have a higher rate of kidney failure than any other group of people. In fact African Americans are four to five times more likely to have kidney failure then Americans of European descent. The discovery of the genetic variants in the APOL1 gene along with the findings of this study may explain a large fraction of this major health disparity.
Thanks to everyone for supporting NephJC over the last year. The growth and participation has been phenomenal. The sky is the limit. We look forward to spirited debates and continued duals between #TeamSwap and #TeamRodby. Continued debate and discourse of scientific literature is what is needed to advance science forward to benefit patients. Happy holidays and new year to all.
The NephJC team
Will PRESERVE preserve the contrast AKI status quo?
Chapter 2- Ulterior Motives
Capter Five: My GIS
Chapter Three: A Precedent for Momentous Change
The #NephJC Pip/Taz + Vancomycin chat wrap up
Storify for both chats
Transcripts
#NephJC posts on #EXTRIP now live at @Ukidney
The #EXTRIP blog posts by the NephJC team are live at Ukidney and a summary of the chat about the intoxications.
#NephJC and #EXTRIP (June 21/22), #EMPA-ReG (June 28/29)
It has been an exciting year for trials in Nephrology. The latest one is EMPAREG, which reported its renal outcomes in the NEJM recently. We will be discussing this trial on June 28th and June 29th.
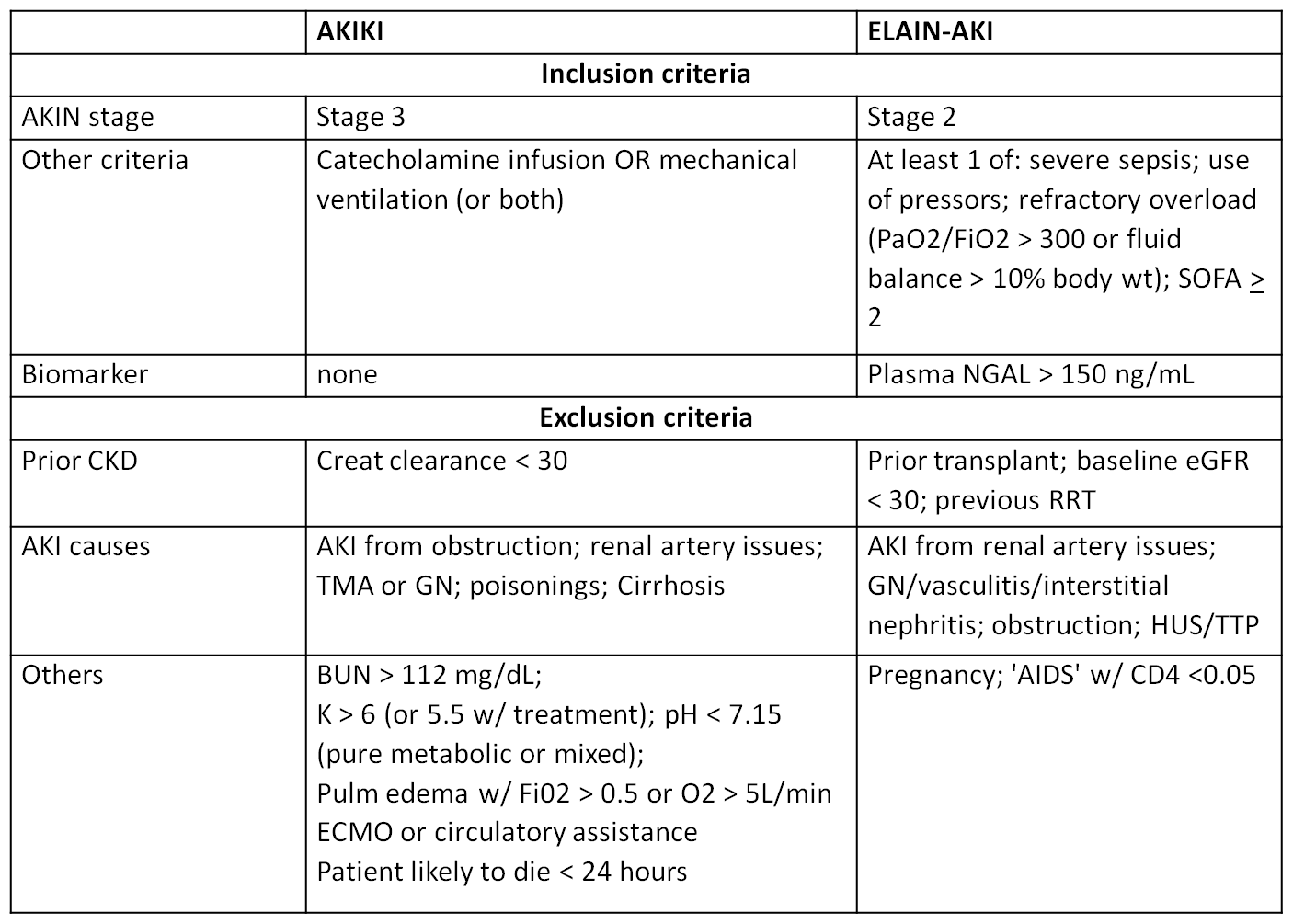
The AKIKI & ELAIN AKI #NephJC wrap up
The AKI timing chats were very ....fast-paced? Lot of great input from the authors, Stephane Gaudry (who made it to both chats) and John Kellum. We had over 80 participants in all
And for those who missed it, check out the great curations via our exemplary storifier, Hector: